Scalable Synthesis of Substituted Aryl Ketones via Oxygen-Tolerant Palladium Catalysis
Scalable Synthesis of Substituted Aryl Ketones via Oxygen-Tolerant Palladium Catalysis
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic methodologies that can withstand the rigors of large-scale manufacturing without compromising on yield or purity. Patent CN110818545B introduces a groundbreaking advancement in the field of organic synthesis, specifically addressing the long-standing challenges associated with the alpha-arylation of ketones. This technology utilizes a novel catalytic system based on saturated nitrogen-heterocyclic carbene (NHC) palladium complexes, which uniquely enables efficient coupling reactions even in the presence of oxygen. Unlike traditional methods that demand stringent anaerobic conditions, this innovation allows for operations in oxygen-containing atmospheres ranging from 1% to 40% by volume. This shift represents a paradigm change for process chemists aiming to develop reliable pharmaceutical intermediate supplier capabilities, as it drastically simplifies reactor requirements and operational protocols while maintaining exceptional catalytic activity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of alpha-aryl carbonyl compounds, which are critical motifs in natural products, pharmaceutical intermediates, and light-emitting polymer precursors, has relied heavily on transition metal-catalyzed cross-coupling reactions. However, the industrial implementation of these methods has been severely hindered by the sensitivity of conventional catalysts. Traditional palladium-phosphine systems and even earlier generations of NHC-palladium complexes often exhibit high susceptibility to oxidation and moisture. Consequently, these reactions necessitate the use of expensive inert gas protections, such as nitrogen or argon blanketing, and require rigorously anhydrous solvents and reagents. Furthermore, to drive reactions involving less reactive substrates like aryl chlorides, excessive amounts of precious metal catalysts were often required, sometimes exceeding 1 mol%, which inflates production costs and complicates downstream metal removal processes. The operational complexity of maintaining a strictly oxygen-free environment throughout catalyst preparation, weighing, and reaction execution creates significant bottlenecks for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
The methodology disclosed in CN110818545B overcomes these historical barriers through the strategic design of the ligand architecture. By employing a saturated nitrogen-heterocyclic carbene structure within the palladium complex, the new catalytic system exhibits remarkable stability under oxidative conditions. This innovation allows the arylation of ketones to proceed efficiently in air or oxygen-enriched environments, effectively removing the need for costly inert atmosphere infrastructure. The patent data indicates that this approach is not merely tolerant to oxygen but thrives under these conditions, facilitating the coupling of various aryl halides, including chlorides, bromides, and iodides, with a wide range of ketone substrates. This robustness translates directly into enhanced supply chain reliability, as the process becomes less dependent on specialized equipment and more adaptable to standard chemical manufacturing facilities, thereby supporting the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Saturated NHC-Pd Catalyzed Arylation
The core of this technological breakthrough lies in the specific structural features of the catalyst, designated as Formula (A) in the patent documentation. The catalyst features a palladium center coordinated by a saturated N-heterocyclic carbene ligand. The saturation of the imidazole ring backbone is the critical differentiator; by reducing the unsaturated double bond to a saturated single bond, the carbon-carbon bond length is increased. This structural modification effectively reduces the tension within the five-membered nitrogen-heterocyclic ring. From a mechanistic perspective, this reduction in ring strain enhances the coordination ability of the ligand towards the metal center. The lower rigidity of the saturated NHC ligand allows it to adapt more effectively during the catalytic cycle, maintaining a stable coordination sphere even when exposed to potential oxidants like molecular oxygen. This stability prevents the premature decomposition of the active catalytic species, which is a common failure mode in conventional systems.
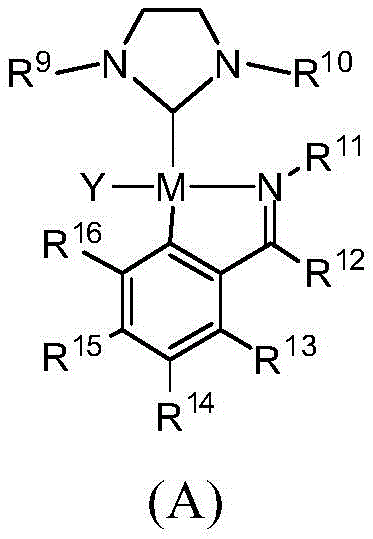
Furthermore, the impurity profile of the resulting substituted aryl ketones is significantly improved due to the high selectivity of this catalytic system. In complex ketone arylation reactions, side reactions such as homocoupling of the aryl halide or over-arylation of the ketone can often plague the process, leading to difficult-to-remove impurities that compromise the quality of high-purity pharmaceutical intermediates. The saturated NHC-Pd catalyst described in this patent demonstrates high turnover numbers and exceptional selectivity, achieving yields of up to 99% with catalyst loadings as low as 0.1 mol%. This high efficiency minimizes the formation of by-products, thereby simplifying the purification workflow. For R&D directors focused on process feasibility, this means a more streamlined path from laboratory bench to pilot plant, with fewer iterations required to meet stringent purity specifications required for regulatory filings.
How to Synthesize Substituted Aryl Ketones Efficiently
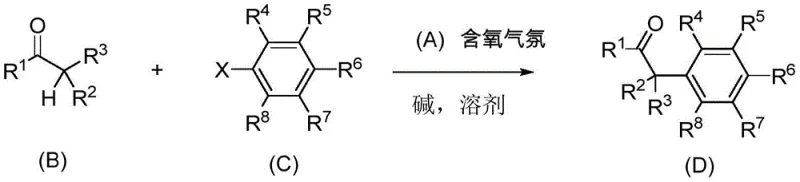
The synthetic protocol outlined in the patent provides a straightforward and highly effective route for generating valuable alpha-aryl ketones. The process begins with the selection of appropriate starting materials, specifically a ketone bearing alpha-hydrogens and an aryl halide or pseudohalide. These substrates are combined in a reaction vessel along with a base, such as sodium tert-butoxide or potassium carbonate, and the specialized saturated NHC-Pd catalyst. The choice of solvent is flexible, encompassing options like 1,4-dioxane, toluene, or THF, allowing for optimization based on solubility and safety profiles. The reaction is then heated, typically between -20°C and 150°C, with a preferred range around 100°C, under an atmosphere containing oxygen. This operational simplicity stands in stark contrast to traditional methods, offering a practical guide for chemists looking to implement this technology. The detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures tailored to different substrate classes.
- Prepare the reaction mixture by combining the ketone substrate, aryl halide, base (e.g., sodium tert-butoxide), and the saturated NHC-Pd catalyst in a suitable solvent like 1,4-dioxane.
- Conduct the reaction under an oxygen-containing atmosphere (1-40% volume ratio) at temperatures ranging from -20°C to 150°C, typically around 100°C for optimal conversion.
- Upon completion, remove the solvent and purify the crude product via column chromatography or crystallization to isolate the high-purity substituted aryl ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this oxygen-tolerant catalytic technology offers profound economic and logistical benefits. The primary advantage stems from the drastic simplification of the reaction environment. By eliminating the absolute requirement for inert gas protection, manufacturers can significantly reduce capital expenditure on specialized gloveboxes, Schlenk lines, and nitrogen generation plants. This reduction in infrastructure dependency lowers the barrier to entry for producing high-value intermediates and allows for greater flexibility in manufacturing site selection. Additionally, the ability to operate in air reduces the consumption of utility gases, contributing to ongoing operational cost savings. The robustness of the catalyst also implies a more resilient supply chain, as the process is less vulnerable to disruptions caused by gas supply shortages or equipment failures related to atmospheric control systems.
- Cost Reduction in Manufacturing: The economic impact of this technology is further amplified by the extremely low catalyst loading required to achieve high conversion rates. Traditional palladium-catalyzed reactions often necessitate high loadings of expensive precious metals to overcome deactivation issues, which not only increases raw material costs but also imposes a heavy burden on waste management and metal scavenging processes. In contrast, this novel system operates efficiently at loadings as low as 0.1 mol%, representing a substantial reduction in precious metal consumption. This efficiency directly translates to lower cost of goods sold (COGS) and reduces the environmental footprint associated with heavy metal waste disposal, aligning with modern green chemistry initiatives and sustainability goals without compromising output.
- Enhanced Supply Chain Reliability: The tolerance of this catalytic system to oxygen and moisture inherently makes the supply chain more robust. Conventional methods that require strictly anhydrous and anaerobic conditions are prone to batch failures if seal integrity is compromised or if reagent quality fluctuates. The forgiving nature of the saturated NHC-Pd catalyst mitigates these risks, ensuring consistent batch-to-batch quality and reliable delivery schedules. Furthermore, the starting materials—aryl halides and simple ketones—are commodity chemicals with well-established global supply networks. The ability to use these readily available feedstocks without demanding ultra-high purity grades for water and oxygen content ensures that production can continue uninterrupted, reducing lead time for high-purity pharmaceutical intermediates and securing continuity of supply for downstream drug manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden complexities, particularly regarding heat transfer and safety in exothermic reactions involving sensitive catalysts. The simplified operational conditions of this arylation method facilitate easier scale-up, as the reaction does not require complex engineering controls to maintain an inert atmosphere in large reactors. This ease of scale-up supports the transition from kilogram to multi-ton production scales with minimal process re-engineering. Moreover, the reduced use of hazardous solvents and the elimination of extensive degassing procedures contribute to a safer working environment and lower emissions. This alignment with environmental compliance standards reduces regulatory hurdles and potential liabilities, making it an attractive option for sustainable chemical manufacturing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented arylation technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on how this method compares to existing industry standards. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this process into their current manufacturing portfolios. The answers highlight the specific advantages in terms of catalyst stability, substrate scope, and operational simplicity that define this innovation.
Q: Why is the saturated NHC ligand superior for industrial arylation?
A: The saturated backbone reduces ring strain and increases ligand flexibility, providing superior stability against oxygen and moisture compared to traditional unsaturated NHC or phosphine ligands.
Q: Can this reaction be performed without inert gas protection?
A: Yes, the patent demonstrates that the catalytic system functions efficiently in oxygen-containing atmospheres (up to 40% O2), eliminating the need for expensive nitrogen or argon blanketing.
Q: What is the typical catalyst loading required for high yields?
A: The method achieves high yields (up to 99%) with extremely low catalyst loadings, potentially as low as 0.1 mol%, significantly reducing precious metal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Aryl Ketones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN110818545B for the production of high-value chemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and purity of every batch. We understand that the consistency of substituted aryl ketones is vital for the efficacy and safety of the final pharmaceutical products, and our manufacturing protocols are designed to meet the highest global regulatory standards.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your specific project needs. Our technical team is prepared to conduct a Customized Cost-Saving Analysis to demonstrate how adopting this oxygen-tolerant catalytic system can optimize your production economics. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your target molecules. By partnering with us, you gain access to a supply chain that prioritizes efficiency, quality, and innovation, ensuring that your development timelines are met with precision and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
