Advanced Synthesis of 1,1'-Diamino-5,5'-bitetrazole for High-Energy Applications
The landscape of high-nitrogen energetic materials is undergoing a significant transformation, driven by the urgent demand for compounds that offer superior energy density and thermal stability. A pivotal development in this sector is detailed in patent CN112125864B, which discloses a robust and highly efficient synthesis method for 1,1'-diamino-5,5'-bitetrazole. This specific compound represents a critical building block in the architecture of next-generation energetic formulations, yet its historical production has been plagued by low yields and complex purification challenges. The disclosed innovation shifts the paradigm from direct amination strategies, which suffer from poor regioselectivity, to a protective group manipulation approach that guarantees structural fidelity. By utilizing a base-catalyzed hydrolysis of a dicarbamate precursor in a sealed alcoholic environment, this method achieves a remarkable leap in both yield and purity. For R&D directors and procurement specialists alike, this patent signals a move away from wasteful, low-efficiency chemistry toward a streamlined process that is inherently more compatible with the rigorous demands of modern supply chains.
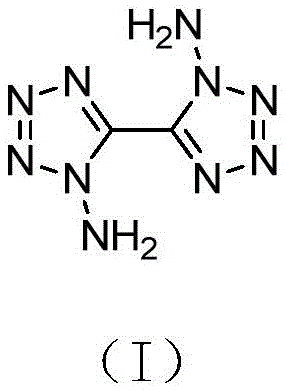
The Limitations of Conventional Methods vs. The Novel Approach
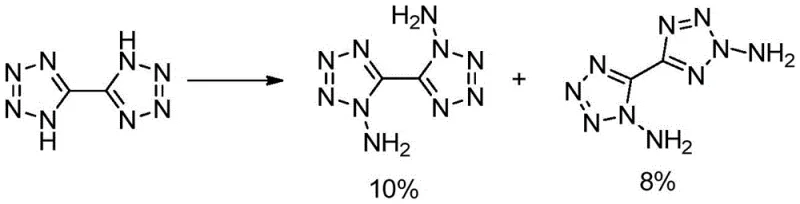
The Limitations of Conventional Methods
Historically, the synthesis of 1,1'-diamino-5,5'-bitetrazole has been a formidable challenge for organic chemists working in the field of energetic materials. The seminal work by Klapötke et al. established a baseline route involving the direct reaction of 5,5'-bitetrazole, but this pathway is fundamentally flawed due to its lack of regiocontrol. As illustrated in the reaction scheme below, the conventional method produces a complex mixture containing not only the desired 1,1'-isomer but also a significant quantity of the 1,2'-isomer byproduct. The yield of the target compound in this traditional route is abysmally low, reported at merely 10%, with an additional 8% lost to the isomeric impurity. This poor selectivity creates a massive bottleneck for scale-up, as separating these structurally similar isomers necessitates labor-intensive and solvent-heavy column chromatography. For any commercial entity, the combination of a 10% yield and the requirement for chromatographic purification renders the process economically unviable for large-scale manufacturing, creating a severe supply constraint for downstream applications.
The Novel Approach
In stark contrast to the stochastic nature of direct amination, the novel approach described in CN112125864B employs a rational design strategy centered on precursor stability and selective deprotection. Instead of attempting to install amino groups onto an unstable tetrazole ring directly, the inventors utilize a 1,1'-dicarbamate-5,5'-bitetrazole intermediate where the nitrogen positions are pre-defined and locked. The innovation lies in the mild yet effective removal of these carbamate protecting groups using common alkali catalysts under sealed heating conditions. This strategic pivot completely bypasses the formation of the unwanted 1,2'-isomer, as the carbon-nitrogen framework is already established in the correct configuration before the final step. The result is a clean reaction profile that delivers the target molecule with exceptional specificity. This methodological shift transforms the synthesis from a low-yield exploration into a predictable, high-efficiency manufacturing process, addressing the core pain points of yield loss and purification complexity that have long hindered the commercialization of high-nitrogen compounds.
Mechanistic Insights into Base-Catalyzed Carbamate Deprotection
The core chemical transformation driving this synthesis is a base-catalyzed hydrolysis (or alcoholysis depending on the specific solvent system) of the carbamate moieties attached to the bitetrazole scaffold. When the 1,1'-dicarbamate precursor is subjected to temperatures between 150°C and 160°C in the presence of a strong base such as sodium hydroxide or potassium hydroxide, the nucleophilic attack on the carbonyl carbon of the carbamate group is significantly accelerated. The high temperature within the sealed vessel ensures that the activation energy barrier for this cleavage is overcome efficiently, leading to the release of the free amine groups and the corresponding alcohol or carbonate byproducts. Crucially, the tetrazole rings themselves remain intact under these specific alkaline conditions, demonstrating the robustness of the heterocyclic core when protected. This mechanistic pathway is distinct from acid-catalyzed deprotections which might risk ring opening or degradation of the sensitive high-nitrogen content. The choice of an alcoholic solvent further facilitates the solubility of both the organic precursor and the inorganic base, creating a homogeneous or semi-homogeneous reaction medium that maximizes contact between reactants and drives the equilibrium toward the desired diamino product.
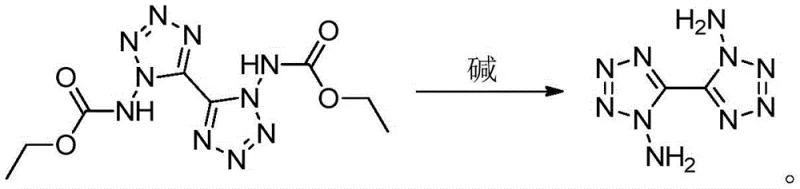
From an impurity control perspective, this mechanism offers a decisive advantage by eliminating the root cause of isomeric contamination. In the conventional route, the ambiguity arises during the bond-forming event where the amino group attacks the tetrazole ring, allowing for multiple attachment points. However, in this novel pathway, the bond formation occurred in a previous, controlled step to create the dicarbamate. The current reaction is purely a cleavage event; it breaks bonds rather than forming new carbon-nitrogen connections that could lead to scrambling. Consequently, the impurity profile is drastically simplified, with the primary contaminants being residual starting material or minor degradation products rather than structural isomers. This inherent selectivity means that the final product purity can exceed 98.6% (as verified by HPLC in the patent examples) simply through standard extraction and drying procedures. For quality control teams, this implies a much more stable and reproducible specification, reducing the analytical burden and ensuring that every batch meets the stringent requirements necessary for energetic material applications without the need for recursive purification loops.
How to Synthesize 1,1'-Diamino-5,5'-bitetrazole Efficiently
The operational simplicity of this new synthesis route makes it particularly attractive for process chemists looking to transfer technology from the bench to the pilot plant. The procedure involves dissolving the 1,1'-dicarbamate-5,5'-bitetrazole precursor in a lower alcohol such as methanol or ethanol, followed by the addition of a stoichiometric amount of an alkali catalyst. The mixture is then charged into a sealed pressure vessel, such as a stainless steel autoclave or a heavy-walled glass tube, and heated to a temperature range of 150°C to 160°C. This sealed environment is critical to maintain the solvent in the liquid phase above its normal boiling point, thereby accelerating the reaction kinetics. After a reaction period of approximately 10 hours, the vessel is cooled, and the product is isolated through a straightforward workup involving water quenching and extraction with ethyl acetate. The detailed standardized operating procedures, including specific molar ratios and safety protocols for handling high-energy intermediates, are outlined in the technical guide below.
- Dissolve the precursor 1,1'-dicarbamate-5,5'-bitetrazole in an alcohol solvent such as methanol or ethanol within a sealed reaction vessel.
- Add a stoichiometric amount of an alkali catalyst, such as sodium hydroxide or potassium hydroxide, to the solution.
- Heat the sealed mixture to 150-160°C for approximately 10 hours, then cool, extract with ethyl acetate, and dry to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from the legacy Klapötke method to this new base-catalyzed process represents a substantial opportunity for cost optimization and risk mitigation. The most immediate impact is the dramatic improvement in mass efficiency; increasing the yield from 10% to roughly 50% means that five times less raw material is required to produce the same amount of finished goods. This five-fold increase in throughput directly translates to a significant reduction in the cost of goods sold (COGS), as the expense of the specialized tetrazole precursors is amortized over a much larger output. Furthermore, the elimination of column chromatography removes a major bottleneck in production scheduling. Chromatography is notoriously difficult to scale, often requiring vast amounts of silica gel and solvents, which creates hazardous waste disposal issues and extends cycle times. By replacing this with a simple extraction and crystallization workflow, the manufacturing timeline is drastically shortened, allowing for faster turnover and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive purification steps and the maximization of raw material utilization. By avoiding the use of silica gel and large volumes of elution solvents required for isomer separation, the operational expenditure on consumables is significantly lowered. Additionally, the higher yield reduces the burden on upstream synthesis stages, meaning that the facility can produce more final product with the same reactor volume and utility consumption. This efficiency gain allows for a more competitive pricing structure for the final energetic intermediate, providing a clear margin advantage in a cost-sensitive market.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by processes that rely on complex, multi-step purifications which are prone to failure or variability. This new method relies on commodity chemicals—such as sodium hydroxide, methanol, and ethyl acetate—which are readily available from multiple global suppliers, reducing the risk of raw material shortages. The robustness of the reaction conditions (sealed vessel heating) is also well-suited for standard chemical manufacturing infrastructure, meaning that production does not require exotic or custom-built equipment. This compatibility with existing assets ensures that supply can be ramped up quickly without long lead times for equipment fabrication, securing a stable supply line for downstream customers.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, the simplification of the workflow offers distinct advantages. The reduction in solvent usage associated with removing chromatography directly lowers the facility's volatile organic compound (VOC) emissions and hazardous waste generation. This aligns with increasingly strict environmental regulations and corporate sustainability goals. Moreover, the scalability of sealed vessel reactions is well-understood in the chemical industry, allowing for a smooth transition from kilogram-scale R&D batches to ton-scale commercial production. The ability to scale without re-engineering the core chemistry de-risks the investment in production capacity, ensuring long-term viability for the supply of this critical high-nitrogen intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN112125864B, providing a factual basis for evaluating the feasibility of adopting this route. Understanding these details is crucial for technical teams assessing the integration of this intermediate into their own formulation pipelines or for procurement officers validating the quality assurances provided by potential suppliers.
Q: What is the primary advantage of this new synthesis method over the Klapötke route?
A: The new method eliminates the formation of the difficult-to-separate 1,2'-isomer, increasing the yield of the target 1,1'-isomer from 10% to approximately 50% while achieving purity greater than 98.6% without column chromatography.
Q: Which catalysts are suitable for this deprotection reaction?
A: The patent specifies that strong alkali catalysts are effective, including inorganic bases like sodium hydroxide (NaOH) and potassium hydroxide (KOH), as well as alkoxides such as sodium methoxide and potassium ethoxide.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes simple reagents and standard sealed vessel equipment (autoclaves) at moderate temperatures (150-160°C), making it highly scalable compared to methods requiring complex purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1'-Diamino-5,5'-bitetrazole Supplier
As the global demand for high-performance energetic materials continues to rise, securing a supply of key intermediates like 1,1'-diamino-5,5'-bitetrazole from a technically competent partner is essential. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in heterocyclic chemistry to deliver solutions that bridge the gap between academic innovation and industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate with stringent purity specifications and maintain rigorous QC labs equipped to verify the absence of critical isomers, guaranteeing that every shipment meets the exacting standards required for energetic applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our advanced synthesis capabilities can improve your bottom line. Please contact us today to request specific COA data for our current batches or to discuss route feasibility assessments for your proprietary projects. Let us be your partner in powering the next generation of high-nitrogen materials.
