Scalable Manufacturing of Trifarotene Intermediates via Advanced One-Pot Suzuki Coupling Technology
Scalable Manufacturing of Trifarotene Intermediates via Advanced One-Pot Suzuki Coupling Technology
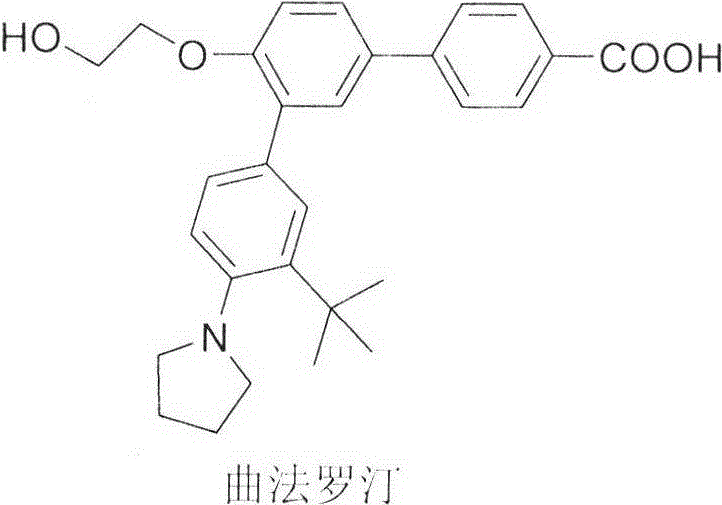
The pharmaceutical industry continuously seeks robust synthetic pathways for complex dermatological agents like Trifarotene, a fourth-generation retinoid approved for treating acne vulgaris. A pivotal advancement in this domain is detailed in Chinese Patent CN113816925A, which discloses a highly efficient preparation method for 3''-tert-butyl-4'-(2-hydroxyethoxy)-4''-pyrrolidin-1-yl[1,1';3',1'']-terphenyl-4-carboxylic acid. This technical breakthrough addresses critical bottlenecks in existing manufacturing protocols by introducing a telescoped, one-pot Suzuki-Miyaura coupling strategy. Unlike traditional multi-step sequences that suffer from low throughput and hazardous reagent usage, this novel approach leverages stable palladium catalysis to achieve a total yield improvement from a mere 5.24% to approximately 61%. For global procurement and R&D teams, this represents a paradigm shift towards safer, more cost-effective, and environmentally sustainable API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Trifarotene intermediates has been plagued by significant operational hazards and inefficiencies, as exemplified by the route disclosed in Patent WO2006066978. The conventional pathway relies heavily on the use of n-butyllithium for the lithiation-borylation of aryl halides, a reagent known for its pyrophoric nature and extreme sensitivity to moisture and air. This necessitates specialized cryogenic equipment and rigorous inert atmosphere controls, driving up capital expenditure and operational complexity. Furthermore, the isolation of arylboronic acid intermediates in these traditional routes is notoriously difficult due to their instability, often leading to decomposition and variable purity. The cumulative effect of these factors results in a dismal overall yield of only 5.24% across the four critical steps of boronization, coupling, and hydrolysis, making the process economically unviable for large-scale commercial supply.
The Novel Approach
In stark contrast, the methodology presented in CN113816925A revolutionizes the synthesis by eliminating the need for hazardous organolithium reagents entirely. The new process initiates with a straightforward hydrolysis of the ester precursor to generate a stable carboxylic acid intermediate, followed by an in-situ borylation of the pyrrolidine-substituted bromide. Crucially, the reaction mixture containing the generated boronate species is not isolated; instead, it is directly subjected to Suzuki coupling with the pre-formed acid intermediate in a single pot. This telescoping strategy not only mitigates the risks associated with handling unstable boronic acids but also streamlines the workflow by removing multiple work-up and purification stages. The result is a dramatic enhancement in process mass intensity and a substantial reduction in solvent waste, aligning perfectly with modern green chemistry principles while delivering superior yields.
Mechanistic Insights into Palladium-Catalyzed One-Pot Suzuki Coupling
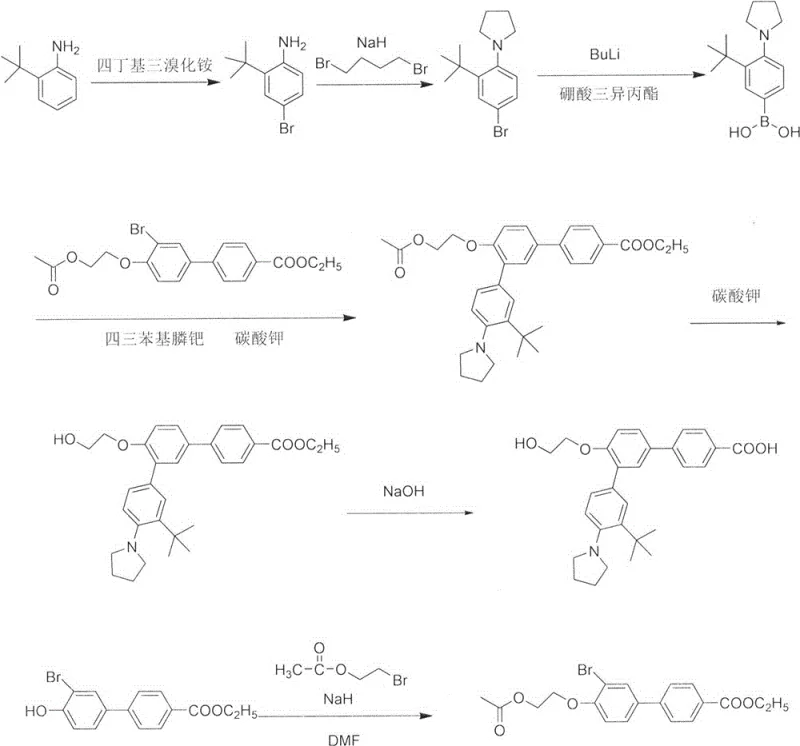
The core of this technological advancement lies in the precise orchestration of palladium-catalyzed cross-coupling reactions under optimized conditions. The mechanism begins with the oxidative addition of the palladium(0) species, generated in situ from palladium acetate and triphenylphosphine, into the carbon-bromine bond of the pyrrolidine-substituted substrate. This is followed by transmetallation with the bis(pinacolato)diboron reagent in the presence of potassium acetate, which acts as a mild base to facilitate the formation of the reactive organoboron intermediate. Unlike harsh bases that might degrade sensitive functional groups, potassium acetate provides a buffered environment that preserves the integrity of the tertiary amine and the steric bulk of the tert-butyl group. The subsequent addition of the carboxylic acid intermediate and a stronger base, potassium phosphate, triggers the second catalytic cycle where the biaryl bond is formed, releasing the final Trifarotene scaffold with high regioselectivity.
Impurity control is another critical aspect where this mechanistic design excels. In previous methods, the coexistence of ester groups and alkaline coupling conditions often led to premature hydrolysis, generating a complex mixture of mono-acid and di-acid byproducts that were difficult to separate. By pre-converting the ester to the free acid (Intermediate 2) in a dedicated step (S1) prior to the coupling event, the new process ensures that the substrate entering the Suzuki reaction is chemically uniform. This pre-emptive stabilization prevents the formation of hydrolysis-related impurities during the high-temperature coupling phase. Furthermore, the use of specific phosphine ligands like triphenylphosphine helps suppress homocoupling side reactions of the aryl halide, ensuring that the final crude product possesses a purity profile suitable for direct recrystallization, thereby simplifying downstream processing.
How to Synthesize Trifarotene Efficiently
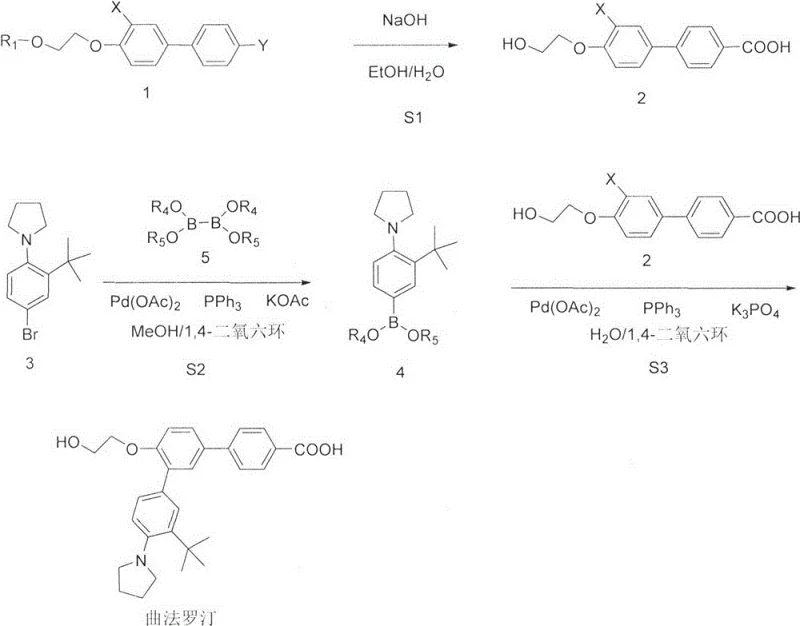
The implementation of this synthesis route requires careful attention to reagent stoichiometry and thermal management to maximize the benefits of the one-pot design. The process begins with the hydrolysis of the ethyl ester precursor using sodium hydroxide in ethanol, followed by acidification to isolate the free acid. Subsequently, the borylation of the bromo-aniline derivative is conducted in a mixed solvent system of 1,4-dioxane and methanol. Once the boronate formation is complete, the reaction mixture is cooled, and the pre-prepared acid intermediate is introduced along with aqueous potassium phosphate. The detailed standardized operating procedures, including specific temperature ramps, addition rates, and quenching protocols required to replicate the 61% yield safely, are outlined in the technical guide below.
- Hydrolyze the protected ester precursor (Compound 1) using sodium hydroxide in ethanol to generate the free acid intermediate (Intermediate 2).
- Perform palladium-catalyzed borylation on the bromo-substituted aniline derivative (Compound 3) using bis(pinacolato)diboron and potassium acetate to form the boronate species (Intermediate 4).
- Without isolating Intermediate 4, add Intermediate 2, potassium phosphate, and additional palladium catalyst directly to the reaction vessel to effect Suzuki coupling and yield Trifarotene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers profound strategic advantages beyond simple yield metrics. The elimination of n-butyllithium removes a major safety bottleneck, allowing production to occur in standard stainless steel reactors rather than requiring specialized glass-lined or Hastelloy vessels designed for pyrophoric materials. This reduction in equipment specificity lowers the barrier to entry for contract manufacturing organizations (CMOs), increasing the pool of qualified suppliers and enhancing supply chain resilience. Moreover, the telescoping of the borylation and coupling steps significantly reduces the total batch time and solvent consumption, leading to a drastic simplification of the manufacturing footprint and a corresponding decrease in utility costs per kilogram of output.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the massive improvement in overall process yield, which has been elevated from roughly 5% to over 60%. This eleven-fold increase in efficiency means that significantly less raw material is required to produce the same amount of active pharmaceutical ingredient, directly lowering the cost of goods sold (COGS). Additionally, by avoiding the isolation and purification of the unstable boronic acid intermediate, the process saves on chromatography media, filtration aids, and drying energy. The use of commodity chemicals like palladium acetate and triphenylphosphine, which can often be recovered or used at low loadings, further optimizes the catalyst cost profile compared to exotic proprietary ligands.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on hazardous reagents that have strict transportation and storage regulations. By replacing n-butyllithium with stable solid reagents like bis(pinacolato)diboron and potassium salts, the logistics of raw material sourcing become far more predictable and less prone to regulatory delays. The robustness of the chemistry also implies a wider operating window for reaction parameters, reducing the risk of batch failures due to minor deviations in temperature or moisture content. This reliability translates into more consistent lead times for customers and a lower risk of stockouts for critical dermatology medications.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is inherently greener. The reduction in solvent usage and the avoidance of hazardous lithiation byproducts simplify waste stream treatment, making it easier for manufacturing sites to comply with increasingly stringent environmental regulations. The process generates fewer organic waste streams and avoids the generation of lithium salts, which can be challenging to dispose of in large quantities. This alignment with green chemistry principles not only reduces disposal costs but also enhances the sustainability profile of the final product, a key factor for pharmaceutical companies aiming to meet corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented Trifarotene synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy processes in terms of safety, yield, and purity.
Q: How does the new one-pot method improve yield compared to prior art?
A: The novel process described in CN113816925A eliminates the isolation of unstable arylboronic acids and avoids the use of hazardous n-butyllithium. By telescoping the borylation and coupling steps, the total yield increases dramatically from approximately 5.24% in conventional routes to over 61%, significantly reducing material waste.
Q: What are the safety advantages of avoiding n-butyllithium in Trifarotene production?
A: Conventional methods rely on n-butyllithium for lithiation-borylation, which poses severe fire risks and requires strict moisture-free conditions. The new method utilizes mild palladium catalysis with stable boron reagents, drastically lowering operational hazards and simplifying equipment requirements for large-scale manufacturing.
Q: Does the direct coupling of the free acid intermediate cause side reactions?
A: Prior art often suffered from ester hydrolysis during alkaline coupling conditions, leading to complex impurity profiles. This optimized protocol pre-hydrolyzes the ester to the free acid (Intermediate 2) before coupling, ensuring chemical compatibility and preventing transesterification or partial hydrolysis by-products during the final C-C bond formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifarotene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex dermatological APIs like Trifarotene depends on mastering intricate synthetic challenges. Our technical team has extensively analyzed the route disclosed in CN113816925A and possesses the expertise to adapt this one-pot Suzuki coupling methodology for industrial-scale production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields observed in the laboratory are faithfully reproduced in our manufacturing facilities. Our state-of-the-art rigorous QC labs and adherence to stringent purity specifications guarantee that every batch of Trifarotene intermediate meets the exacting standards required for global regulatory filings.
We invite pharmaceutical partners to leverage our technical capabilities to optimize their supply chains for this high-value retinoid. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential reductions in COGS achievable through this advanced manufacturing route. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how our scalable solutions can secure your long-term supply of high-quality Trifarotene intermediates.
