Advanced Catalytic Synthesis of Benzopyrone Derivatives for Scalable Pharmaceutical Manufacturing
Introduction to Green Catalytic Synthesis Technologies
The pharmaceutical industry is currently undergoing a significant paradigm shift towards greener, more sustainable synthetic methodologies, driven by the dual pressures of regulatory compliance and economic efficiency. A pivotal development in this domain is documented in patent CN111875591B, which discloses a novel method for the catalytic synthesis of drug intermediates, specifically 3-benzo[d]imidazole benzopyrone derivatives. These compounds represent a critical structural scaffold in modern medicinal chemistry, exhibiting potent biological activities including Rho kinase inhibition, which is relevant for treating cardiovascular diseases and cancer. The traditional synthesis of such complex heterocycles often involves multi-step sequences with harsh conditions, leading to poor atom economy and substantial environmental burdens. However, the technology described in this patent introduces a streamlined, one-pot multicomponent reaction strategy that utilizes a specialized basic ionic liquid catalyst. This approach not only addresses the longstanding issues of catalyst recovery and high loading requirements but also significantly enhances the overall yield and purity profile of the final active pharmaceutical ingredient (API) intermediate, positioning it as a highly attractive route for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 3-benzo[d]imidazole benzopyrone derivatives was predominantly achieved through conventional multistep pathways or less efficient one-pot methods utilizing traditional organic bases. For instance, existing literature describes the use of pyridine as a catalyst in ethanol solvent, which, while functional, suffers from several critical drawbacks that hinder industrial viability. Primarily, pyridine exhibits relatively low catalytic activity, necessitating high loading amounts typically around 10 mol% relative to the substrate, which increases raw material costs and complicates downstream purification due to the difficulty in removing residual amine odors and impurities. Furthermore, both the pyridine catalyst and the ethanol solvent are volatile and difficult to recover efficiently, leading to significant solvent waste and environmental pollution. The selectivity of these traditional systems is often suboptimal, resulting in the formation of various side products that lower the overall yield, which historically ranges broadly from 48% to 89%. Additionally, the reaction times required for these conventional methods are often prolonged, reducing the throughput capacity of manufacturing facilities and increasing energy consumption, thereby creating a bottleneck for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent leverages a sophisticated basic ionic liquid catalyst that fundamentally alters the reaction kinetics and thermodynamics. This new methodology employs a one-pot condensation of substituted benzaldehyde, 2-cyanomethyl benzimidazole, and 5,5-dimethyl-1,3-cyclohexanedione, facilitated by the unique properties of the ionic liquid. ![General reaction scheme for the synthesis of 3-benzo[d]imidazole benzopyrone derivatives using basic ionic liquid catalyst](/insights/img/benzopyrone-derivative-ionic-liquid-pharma-supplier-20260307103605-02.webp) The use of this advanced catalyst allows for a dramatic reduction in catalyst loading to merely 4-6%, effectively halving the requirement compared to pyridine-based systems. Moreover, the reaction is conducted in an isopropanol aqueous solution, a solvent system that offers superior safety and environmental profiles compared to pure organic solvents. The process is remarkably efficient, with reaction times shortened to between 26 and 48 minutes under reflux conditions, yet it consistently delivers yields exceeding 90% for many substrates, with purities often surpassing 98% as determined by HPLC. This represents a substantial leap forward in process intensification, enabling manufacturers to achieve higher throughput with significantly reduced waste generation, aligning perfectly with the goals of cost reduction in API manufacturing and sustainable chemical production.
The use of this advanced catalyst allows for a dramatic reduction in catalyst loading to merely 4-6%, effectively halving the requirement compared to pyridine-based systems. Moreover, the reaction is conducted in an isopropanol aqueous solution, a solvent system that offers superior safety and environmental profiles compared to pure organic solvents. The process is remarkably efficient, with reaction times shortened to between 26 and 48 minutes under reflux conditions, yet it consistently delivers yields exceeding 90% for many substrates, with purities often surpassing 98% as determined by HPLC. This represents a substantial leap forward in process intensification, enabling manufacturers to achieve higher throughput with significantly reduced waste generation, aligning perfectly with the goals of cost reduction in API manufacturing and sustainable chemical production.
Mechanistic Insights into Basic Ionic Liquid Catalysis
The exceptional performance of this synthetic route can be attributed to the unique molecular architecture of the basic ionic liquid catalyst, which functions through a dual-activation mechanism. 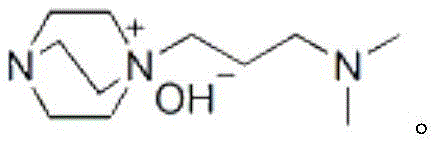 Unlike simple organic bases that rely on a single mode of activation, this ionic liquid possesses both Brønsted basic sites and Lewis basic sites within its structure. This bifunctionality allows the catalyst to simultaneously activate multiple reaction partners: the basic sites can deprotonate the active methylene group of the cyclohexanedione to generate a nucleophilic enolate, while concurrently activating the carbonyl group of the aldehyde through hydrogen bonding or electrostatic interactions. This synergistic activation lowers the energy barrier for the Knoevenagel condensation and subsequent cyclization steps, facilitating a rapid and smooth transformation even at relatively mild temperatures. The stability of the ionic liquid structure ensures that it remains intact throughout the vigorous reflux conditions, preventing decomposition and maintaining consistent catalytic activity over multiple cycles, which is a critical factor for maintaining batch-to-batch consistency in large-scale operations.
Unlike simple organic bases that rely on a single mode of activation, this ionic liquid possesses both Brønsted basic sites and Lewis basic sites within its structure. This bifunctionality allows the catalyst to simultaneously activate multiple reaction partners: the basic sites can deprotonate the active methylene group of the cyclohexanedione to generate a nucleophilic enolate, while concurrently activating the carbonyl group of the aldehyde through hydrogen bonding or electrostatic interactions. This synergistic activation lowers the energy barrier for the Knoevenagel condensation and subsequent cyclization steps, facilitating a rapid and smooth transformation even at relatively mild temperatures. The stability of the ionic liquid structure ensures that it remains intact throughout the vigorous reflux conditions, preventing decomposition and maintaining consistent catalytic activity over multiple cycles, which is a critical factor for maintaining batch-to-batch consistency in large-scale operations.
Furthermore, the choice of the isopropanol-water solvent system plays a pivotal role in the mechanistic pathway and product isolation. The specific volume percentage of isopropanol, optimized between 84% and 91%, creates a solvent environment with tailored polarity that maximizes the solubility of reactants during the high-temperature reflux phase while minimizing the solubility of the product upon cooling. This phenomenon effectively turns the reaction solvent into a recrystallization medium, driving the equilibrium towards product formation through precipitation. As the product crystallizes out of the solution, it is inherently protected from further side reactions or degradation, which contributes to the observed high purity levels. The presence of water in the mixture also enhances the polarity of the medium, which helps to stabilize the charged intermediates formed during the ionic liquid-catalyzed process, thereby accelerating the reaction rate. This intricate balance of solubility and reactivity eliminates the need for separate crystallization steps or chromatographic purification, streamlining the entire workflow and reducing the potential for impurity introduction during workup.
How to Synthesize 3-Benzo[d]imidazole Benzopyrone Derivatives Efficiently
Implementing this advanced synthesis protocol requires precise control over reaction parameters to fully realize the benefits of the ionic liquid catalytic system. The process begins with the preparation of the reaction vessel, typically a three-neck flask equipped with a magnetic stirrer, thermometer, and spherical condenser to ensure efficient mixing and temperature control. The reactants—substituted benzaldehyde, 2-cyanomethyl benzimidazole, and 5,5-dimethyl-1,3-cyclohexanedione—are introduced into the isopropanol aqueous solution at room temperature to ensure homogeneous mixing before the catalyst is added. This order of addition is crucial to prevent localized high concentrations of the catalyst which could lead to exothermic spikes. Once the catalyst is introduced, the mixture is heated to reflux, and the progress is monitored via thin-layer chromatography (TLC) to confirm the disappearance of the starting material spot, ensuring complete conversion before proceeding to isolation.
- Prepare the reaction mixture by combining substituted benzaldehyde, 2-cyanomethyl benzimidazole, and 5,5-dimethyl-1,3-cyclohexanedione in an isopropanol aqueous solution (84-91% v/v) within a three-neck flask equipped with reflux condenser.
- Add the basic ionic liquid catalyst (4-6 mol%) to the mixture at room temperature, then heat to reflux for 26-48 minutes until TLC indicates complete consumption of starting materials.
- Cool the reaction to room temperature to induce precipitation, filter the solid product, wash with isopropanol aqueous solution, and dry under vacuum; the filtrate can be recycled for subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers compelling strategic advantages that extend far beyond simple yield improvements. The transition from volatile, non-recyclable catalysts like pyridine to a stable, recyclable basic ionic liquid system fundamentally reshapes the cost structure of production. By eliminating the need for expensive and hazardous solvent recovery distillation columns typically required for ethanol/pyridine mixtures, the process significantly reduces energy consumption and capital expenditure on waste treatment infrastructure. The ability to recycle the catalytic system directly from the filtrate for multiple consecutive batches without complex regeneration steps translates into substantial cost savings on raw materials, as the effective cost of the catalyst per kilogram of product is drastically lowered. This efficiency gain is particularly valuable in the context of fluctuating raw material markets, providing a buffer against price volatility and ensuring more predictable budgeting for long-term projects.
- Cost Reduction in Manufacturing: The implementation of this ionic liquid catalytic system drives down manufacturing costs through multiple mechanisms, primarily by reducing the catalyst loading requirement by nearly half compared to traditional methods. This reduction in catalyst quantity, combined with the elimination of costly purification steps such as column chromatography, results in a leaner production process with lower labor and material inputs. The simplified workup procedure, which relies on simple filtration and washing rather than complex extraction and distillation sequences, further reduces the operational time and utility costs associated with each batch. Consequently, the overall cost of goods sold (COGS) for these high-value intermediates is significantly optimized, allowing for more competitive pricing strategies in the global marketplace without compromising on quality standards.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this synthetic route ensures greater reliability and continuity of supply. The use of commercially available and stable starting materials, coupled with a catalyst that can be reused multiple times, minimizes the risk of production delays caused by raw material shortages or catalyst supply chain disruptions. The shortened reaction time of under one hour allows for faster turnover of manufacturing equipment, increasing the overall capacity of the production facility to meet sudden surges in demand. Furthermore, the high selectivity of the reaction reduces the formation of difficult-to-remove impurities, ensuring that the final product consistently meets stringent quality specifications, thereby reducing the risk of batch rejection and the associated supply chain bottlenecks that can arise from quality failures.
- Scalability and Environmental Compliance: The environmental profile of this process makes it exceptionally well-suited for scaling up to industrial volumes while maintaining compliance with increasingly strict environmental regulations. The replacement of toxic organic solvents with a safer isopropanol-water mixture reduces the facility's volatile organic compound (VOC) emissions and lowers the hazard classification of the waste streams generated. The recyclability of the catalyst and solvent system dramatically reduces the volume of liquid waste requiring disposal, aligning with green chemistry principles and corporate sustainability goals. This reduced environmental footprint not only mitigates regulatory risks but also enhances the brand reputation of the manufacturer as a responsible partner, which is an increasingly important criterion for multinational pharmaceutical companies when selecting suppliers for their critical API intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalytic technology, derived directly from the experimental data and comparative analysis presented in the patent documentation. These insights are intended to clarify the operational benefits and feasibility of adopting this method for large-scale production, helping stakeholders make informed decisions about integrating this technology into their existing manufacturing frameworks.
Q: What are the primary advantages of using basic ionic liquids over traditional pyridine catalysts?
A: Basic ionic liquids offer dual active sites (Brønsted and Lewis basicity) which significantly enhance catalytic activity, allowing for lower catalyst loading (4-6% vs 10%) and shorter reaction times. Furthermore, their non-volatile nature enables efficient recycling of the catalytic system, drastically reducing waste and operational costs compared to volatile organic bases like pyridine.
Q: How does the isopropanol-water solvent system contribute to product purity?
A: The specific isopropanol-water mixture (84-91% isopropanol) serves a dual function as both the reaction medium and a recrystallization solvent. Upon cooling, the product precipitates directly from the reaction mixture with high purity (>98%), eliminating the need for complex chromatographic purification and simplifying the isolation process.
Q: Is the catalytic system reusable for industrial scale-up?
A: Yes, the patent data demonstrates that the filtrate containing the ionic liquid and solvent can be reused for at least 4 cycles with only negligible decreases in yield and purity. This recyclability is a critical factor for sustainable industrial scale-up and cost reduction in large-volume manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Benzo[d]imidazole Benzopyrone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic synthesis methods described in patent CN111875591B and have integrated similar green chemistry principles into our own R&D and production capabilities. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of 3-benzo[d]imidazole benzopyrone derivatives we produce. We understand that in the pharmaceutical sector, consistency and quality are paramount, and our commitment to process optimization ensures that we deliver intermediates that meet the highest global regulatory standards, supporting our clients in accelerating their drug development timelines.
We invite potential partners to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project requirements. By leveraging our expertise in ionic liquid catalysis and multicomponent reactions, we can offer a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to our optimized processes can improve your bottom line. We encourage you to contact us today to request specific COA data for our catalog of benzopyrone intermediates and to receive detailed route feasibility assessments for your custom synthesis projects, ensuring a partnership built on transparency, technical excellence, and mutual growth.
