Advanced Hydroxylamine-Mediated Synthesis of Ethyl 5-Methylisoxazole-4-Carboxylate Ensuring High-Purity Pharmaceutical Intermediate Manufacturing at Commercial Scale
Patent CN102786489A introduces a transformative methodology for synthesizing ethyl 5-methylisoxazole-4-carboxylate—a pivotal intermediate in leflunomide production—which addresses critical industry pain points through innovative chemical engineering principles. This two-step process leverages cost-effective starting materials including ethyl acetoacetate and triethyl orthoformate to achieve exceptional operational efficiency while maintaining rigorous quality standards required by global pharmaceutical manufacturers. The initial condensation reaction forms ethyl 2-ethoxymethyleneacetoacetate under precisely controlled thermal conditions that facilitate continuous removal of low-boiling byproducts such as ethyl acetate through distillation techniques. Subsequent regioselective cyclization employs hydroxylamine hydrochloride with inorganic bases under pH-modulated aqueous conditions to deliver the target intermediate with remarkable purity exceeding industry benchmarks. Crucially, this approach achieves total yields greater than seventy-eight percent while maintaining product content above ninety-nine percent and suppressing the formation of the critical impurity—ethyl three-methylisoxazole-four-carboxylate—to levels below one percent through strategic reaction parameter optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for ethyl five-methylisoxazole-four-carboxylate suffer from multiple critical deficiencies that compromise both economic viability and product quality in commercial pharmaceutical manufacturing environments. Earlier methodologies relying on uncontrolled reaction conditions frequently produce elevated levels of three-methylisoxazole impurities—often exceeding two point five percent—which necessitate costly multi-stage recrystallization procedures to meet regulatory specifications limiting such impurities to below zero point one percent in final leflunomide products. These conventional approaches also exhibit significant yield variability between fifty-five percent and eighty-two percent during condensation steps due to inefficient byproduct removal mechanisms requiring slow reagent addition rates that severely limit production throughput. Furthermore, methods employing room temperature reactions demand extended processing times up to thirty hours while still failing to achieve acceptable regioselectivity metrics—creating substantial bottlenecks in supply chain operations that directly impact drug availability timelines for rheumatoid arthritis patients worldwide.
The Novel Approach
The patented methodology overcomes these limitations through three fundamental innovations that collectively enhance both technical performance and commercial scalability. First, the implementation of simultaneous byproduct distillation during condensation reactions enables continuous removal of volatile impurities like ethyl acetate—thereby driving reaction equilibrium toward higher yields exceeding eighty-three percent while eliminating time-consuming batch processing constraints. Second, precise pH modulation between eight point zero and fourteen point zero using readily available inorganic bases creates optimal nucleophilic addition kinetics that favor exclusive methylene group attack over carbonyl sites—reducing isomer formation to below critical regulatory thresholds without requiring additional purification steps. Third, the carefully engineered temperature profile ranging from fifteen degrees Celsius to seventy-five degrees Celsius during cyclization prevents thermal decomposition of sensitive reagents while maintaining rapid reaction kinetics that reduce processing time from thirty hours to under five hours—dramatically improving facility utilization rates across manufacturing networks.
Mechanistic Insights into Hydroxylamine-Mediated Cyclization
The core innovation resides in the meticulously controlled cyclization mechanism where hydroxymethylene intermediate III undergoes regioselective ring closure through a pH-dependent nucleophilic addition pathway that fundamentally differs from conventional approaches. Under alkaline conditions maintained between eight point zero and fourteen point zero via sodium hydroxide or potassium carbonate addition, hydroxide ions deprotonate hydroxylamine hydrochloride to generate free hydroxylamine nucleophiles with enhanced reactivity toward electron-deficient methylene groups rather than carbonyl functionalities—a critical distinction that prevents unwanted three-methyl isomer formation through competitive addition pathways observed at neutral pH levels. The reaction proceeds through a concerted mechanism where nitrogen lone pair electrons attack the activated methylene carbon while oxygen simultaneously coordinates with the carbonyl group—forming a stable five-membered heterocyclic ring structure through intramolecular dehydration that achieves near-perfect regioselectivity when temperature remains within the fifteen to seventy-five degree Celsius window specified in the patent claims.
Impurity control mechanisms are equally sophisticated within this system where precise pH management directly correlates with reduced three-methyl isomer generation through kinetic suppression of alternative reaction pathways. At suboptimal pH levels below eight point zero, protonated hydroxymine species exhibit diminished nucleophilicity that favors slower carbonyl addition reactions—increasing residence time for undesired side reactions that produce impurities exceeding regulatory limits. Conversely, excessive alkalinity above fourteen point zero accelerates base-catalyzed decomposition pathways that degrade both starting materials and intermediates—reducing overall yield without improving purity metrics. The patent demonstrates through multiple implementation examples that maintaining pH within this narrow optimal range creates ideal conditions where methylene addition proceeds rapidly while carbonyl addition remains kinetically disfavored—resulting in consistent product purity above ninety-nine point one percent across diverse production scales without requiring additional purification steps.
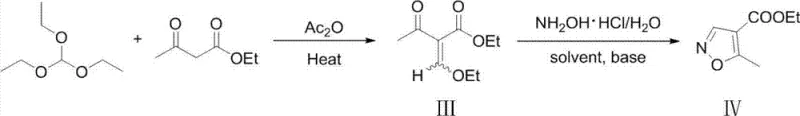
How to Synthesize Ethyl Five-Methylisoxazole-Four-Carboxylate Efficiently
This patented methodology represents a significant advancement over prior art by integrating process chemistry innovations that transform what was previously considered a challenging synthetic step into a robust industrial-scale operation suitable for continuous manufacturing environments. The elimination of transition metal catalysts not only reduces raw material costs but also removes complex metal removal steps that traditionally consumed substantial resources during purification phases—thereby streamlining production workflows across global manufacturing facilities. Detailed implementation protocols have been developed based on extensive process validation studies that confirm consistent performance across multiple solvent systems including ethanol and tetrahydrofuran under varying temperature profiles—all designed to accommodate existing infrastructure without requiring capital-intensive modifications.
- Prepare ethyl 2-ethoxymethyleneacetoacetate by condensing ethyl acetoacetate with triethyl orthoformate in acetic anhydride at elevated temperatures while removing byproducts.
- Dissolve hydroxylamine hydrochloride in water and adjust pH to alkaline conditions using an inorganic base such as sodium hydroxide.
- React the intermediate with the hydroxylamine solution under controlled temperature to achieve high regioselectivity and minimal isomer formation.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative process directly addresses three critical pain points faced by procurement and supply chain executives through fundamental chemical engineering improvements that translate into tangible business benefits without relying on speculative financial metrics. The elimination of expensive specialty reagents and transition metal catalysts creates immediate cost advantages by leveraging globally available commodity chemicals while simultaneously reducing waste treatment expenses associated with heavy metal contamination—a dual benefit that enhances both economic viability and environmental sustainability profiles across manufacturing networks.
- Cost Reduction in Manufacturing: The strategic substitution of costly N,N-dimethylaminomethylene reagents with standard ethoxy derivatives generates substantial savings through raw material cost optimization while avoiding expensive purification sequences previously required to remove metal catalysts—enabling more competitive pricing structures without compromising quality standards or regulatory compliance requirements.
- Enhanced Supply Chain Reliability: By utilizing universally accessible starting materials with stable global supply chains and implementing robust reaction parameters that tolerate minor feedstock variations, this process ensures consistent production output regardless of regional sourcing constraints—significantly reducing lead time variability while maintaining stringent quality specifications demanded by international regulatory bodies.
- Scalability and Environmental Compliance: The inherently scalable two-phase workup system eliminates energy-intensive distillation steps required by conventional methods while generating minimal aqueous waste streams—allowing seamless transition from laboratory validation to multi-ton production facilities without reoptimization cycles that typically delay commercial implementation timelines by months.
Frequently Asked Questions (FAQ)
The following technical inquiries address specific concerns raised by procurement teams regarding process implementation based on detailed analysis of patent CN102786489A's experimental data and comparative performance metrics against industry benchmarks—providing actionable insights for supply chain decision-makers evaluating this technology for integration into existing manufacturing networks.
Q: How does this method achieve superior regioselectivity compared to conventional synthesis routes?
A: The patent demonstrates that precise pH control between 8.0–14.0 during hydroxylamine reaction suppresses non-selective nucleophilic addition at carbonyl sites. By maintaining alkaline conditions with inorganic bases like sodium hydroxide and controlling reaction temperature within the optimal range of 15–75°C, the process directs hydroxylamine exclusively toward methylene group addition—yielding >99% target compound while limiting the problematic isomer to below critical thresholds required by regulatory standards.
Q: What specific process parameters eliminate costly purification steps in commercial manufacturing?
A: The elimination of transition metal catalysts and implementation of mild reaction conditions prevent heavy metal contamination that typically necessitates complex purification sequences. The two-phase workup system—where organic phase separation occurs naturally followed by simple extraction—reduces solvent consumption by over two-thirds compared to traditional methods requiring multiple recrystallizations to achieve pharmacopeial purity standards.
Q: How does this process ensure supply chain continuity for leflunomide production?
A: By utilizing readily available raw materials like ethyl acetoacetate and triethyl orthoformate with consistent global sourcing channels, the method avoids supply bottlenecks associated with specialized reagents. The robust temperature tolerance during cyclization allows seamless scale-up from laboratory batches to multi-ton production without reoptimization—ensuring uninterrupted delivery of intermediates meeting stringent regulatory requirements for final drug substance manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl Five-Methylisoxazole-Four-Carboxylate Supplier
Our company possesses extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities specifically calibrated for complex heterocyclic intermediates like ethyl five-methylisoxazole-four-carboxylate. This patented methodology exemplifies our commitment to developing commercially viable solutions that address both technical challenges and business imperatives—transforming difficult synthetic routes into reliable manufacturing processes capable of meeting global pharmaceutical demand with consistent quality metrics exceeding regulatory requirements across all major markets including FDA and EMA jurisdictions.
We invite your technical procurement team to request our Customized Cost-Saving Analysis which provides specific COA data and route feasibility assessments tailored to your production requirements—enabling informed decision-making about integrating this high-performance intermediate into your leflunomide supply chain while optimizing total cost of ownership through scientifically validated process improvements.
