Advanced Transition Metal Catalyzed C-H Activation for Scalable 1,2-Benzothiazine Manufacturing
Advanced Transition Metal Catalyzed C-H Activation for Scalable 1,2-Benzothiazine Manufacturing
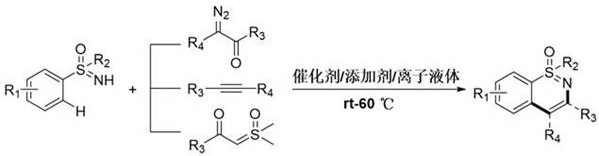
The pharmaceutical and agrochemical industries are constantly seeking more efficient, environmentally benign pathways to access complex heterocyclic scaffolds that serve as the backbone for bioactive molecules. A pivotal advancement in this domain is documented in Chinese Patent CN110105305B, which discloses a novel green synthesis method for 1,2-benzothiazine derivatives. This technology leverages transition metal-catalyzed C-H activation and cyclization reactions, utilizing N1,N3-disubstituted imidazole ionic liquids as the reaction medium. By shifting away from traditional volatile organic solvents and harsh conditions, this methodology addresses critical pain points regarding safety, waste generation, and process scalability. For R&D directors and procurement specialists alike, understanding the nuances of this patent provides a strategic advantage in sourcing high-purity intermediates while aligning with global sustainability goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2-benzothiazine derivatives has relied heavily on methodologies that impose significant operational burdens and environmental hazards. Traditional routes often necessitate the use of stoichiometric amounts of hazardous reagents, strong acids or bases as additives, and elevated temperatures that can compromise the stability of sensitive functional groups. Furthermore, the reliance on volatile organic solvents creates substantial challenges in terms of solvent recovery, worker safety, and regulatory compliance regarding VOC emissions. Many existing catalytic systems suffer from poor turnover numbers and cannot be easily separated from the product mixture, leading to potential heavy metal contamination in the final API intermediate. These factors collectively drive up the cost of goods sold and extend the lead time for process development, making conventional methods less attractive for modern, large-scale manufacturing requirements.
The Novel Approach
In stark contrast, the methodology outlined in patent CN110105305B introduces a paradigm shift by employing a transition metal-catalyzed C-H activation strategy within an ionic liquid medium. This approach operates under remarkably mild conditions, typically ranging from room temperature to 60°C, which drastically reduces energy consumption and thermal stress on the reactants. The use of NH-sulfinimides as starting materials coupled with diverse coupling reagents such as diazo compounds, alkynes, and sulfur ylides allows for exceptional structural diversity. Crucially, the ionic liquid serves not merely as a solvent but as a stabilizing matrix that facilitates the recycling of the catalytic system. This innovation eliminates the need for excessive additives and toxic solvents, thereby streamlining the downstream purification process and enhancing the overall atom economy of the transformation.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation/Cyclization
The core of this synthetic breakthrough lies in the sophisticated interplay between the transition metal catalyst, typically a rhodium(III) complex such as dichloro(pentamethylcyclopentadienyl)rhodium(III) dimer, and the silver additive. The mechanism initiates with the coordination of the sulfinimide substrate to the metal center, followed by the activation of the ortho-C-H bond on the aromatic ring. This C-H activation step is the rate-determining phase where the unique properties of the ionic liquid play a vital role in stabilizing the cationic metal species. Subsequent insertion of the coupling reagent—whether it be a diazo compound forming a carbene intermediate or an alkyne undergoing migratory insertion—leads to the formation of the new carbon-carbon bond. The final cyclization step releases the 1,2-benzothiazine product and regenerates the active catalytic species, ready for the next cycle.
Understanding the impurity profile is critical for R&D teams aiming to replicate this process. The high selectivity of the rhodium catalyst minimizes the formation of regioisomers, a common issue in electrophilic aromatic substitution reactions. The mild thermal window prevents decomposition pathways that often plague high-temperature syntheses. Moreover, the by-product of the reaction, particularly when using diazo compounds, is primarily nitrogen gas, which escapes the reaction mixture effortlessly. This feature significantly simplifies the work-up procedure, as there are no heavy organic by-products to separate via complex chromatography. The ability to tune the electronic properties of the ionic liquid also offers a handle to further suppress side reactions, ensuring that the final product meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize 1,2-Benzothiazine Derivatives Efficiently
Executing this synthesis requires precise control over stoichiometry and reaction parameters to maximize yield and catalyst longevity. The patent specifies a molar ratio of sulfonimide to coupling reagent between 1:1.2 and 1:3.0, ensuring that the limiting reagent is fully consumed while minimizing excess waste. The catalyst loading is kept low, typically between 0.02 and 0.05 equivalents, supported by a silver additive at 0.08 to 0.2 equivalents to facilitate halide abstraction and generate the active cationic catalyst. The reaction concentration is maintained between 0.1 and 0.5 mol/L to balance reaction kinetics with heat dissipation. Following the reaction, a simple extraction with diethyl ether separates the product from the ionic liquid phase, which can then be dried and reused.
- Charge a clean reactor with sulfonimide compounds, coupling reagents (diazo, alkyne, or sulfur ylide), transition metal catalyst, silver additive, and N1,N3-disubstituted imidazole ionic liquid.
- Stir the reaction mixture in an oil bath at a controlled temperature between room temperature and 60°C for approximately 24 hours to ensure complete conversion.
- Upon completion, extract with diethyl ether, purify the organic layer via silica gel column chromatography, and recover the ionic liquid layer for recycling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials and the reduction of dependency on scarce resources. By enabling the recycling of the expensive transition metal catalyst and the ionic liquid solvent, the process decouples production costs from the volatility of precious metal markets. This circular economy approach within the reactor vessel ensures a more predictable cost structure and mitigates the risk of supply disruptions associated with single-use reagents. Furthermore, the elimination of toxic volatile solvents reduces the regulatory burden and insurance costs associated with hazardous material storage and transport.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the recyclability of the reaction medium. In traditional batch processes, the cost of solvent purchase, distillation, and disposal constitutes a significant portion of the operating expense. Here, the ionic liquid layer, once dried under reduced pressure, can be directly reintroduced into the next batch without the addition of fresh catalyst or silver salts. This capability effectively amortizes the cost of these high-value components over numerous production cycles. Additionally, the high atom economy, characterized by the release of nitrogen gas as the only by-product in many variations, means that a larger proportion of the input mass ends up in the final product, reducing the cost per kilogram of the active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain resilience. Operating at temperatures between room temperature and 60°C removes the need for specialized high-pressure or cryogenic equipment, allowing the process to be scaled in standard glass-lined or stainless steel reactors available at most CDMO facilities. The wide substrate scope, accommodating various halogens, alkyl, and aryl groups on the starting materials, ensures that supply chains are not bottlenecked by the availability of highly specialized, exotic precursors. This flexibility allows procurement teams to source generic, commercially available building blocks, thereby reducing lead times and increasing the agility of the supply network in response to market demands.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen safety and environmental challenges, but this methodology is inherently designed for green manufacturing. The non-flammable nature of the ionic liquid solvent eliminates the fire hazards associated with ethers or hydrocarbons, facilitating safer scale-up to multi-ton production levels. From an environmental compliance perspective, the reduction in VOC emissions and the minimization of heavy metal waste align perfectly with increasingly stringent global environmental regulations. This proactive adherence to green chemistry principles future-proofs the manufacturing process against potential regulatory tightening, ensuring uninterrupted production continuity and protecting the brand reputation of the end-user.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using ionic liquids in this synthesis?
A: Ionic liquids provide a unique solvent environment that enhances thermal stability and allows for the recycling of the catalytic system, significantly reducing waste and operational costs compared to volatile organic solvents.
Q: Can the catalyst system be reused effectively?
A: Yes, the patent data indicates that the ionic liquid layer containing the catalyst can be recycled after vacuum drying without the need to add fresh catalyst or silver salts, maintaining high efficiency over multiple cycles.
Q: What represents the main by-product of this reaction?
A: The reaction is designed for high atom economy, with nitrogen gas being the sole by-product in many variations, which simplifies purification and minimizes environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Benzothiazine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in patent CN110105305B for the production of high-value pharmaceutical intermediates. As a premier CDMO partner, we possess the technical expertise to translate these laboratory-scale innovations into robust, commercial-scale processes. Our facilities are equipped to handle complex transition metal catalysis with the utmost precision, ensuring that every batch meets rigorous quality standards. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art infrastructure to deliver consistent results. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and purity of every compound we produce.
We invite global partners to collaborate with us to unlock the full commercial potential of 1,2-benzothiazine derivatives. Whether you require custom synthesis for early-stage drug discovery or large-scale manufacturing for clinical supply, our team is ready to support your objectives. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to reach out for specific COA data and route feasibility assessments to see how our optimized processes can enhance your supply chain efficiency and reduce your overall time to market.
