Scalable Glucose-Mediated Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly those bearing fluorinated motifs which are critical for metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in patent CN113880781A, which discloses a novel method for synthesizing 3-trifluoromethyl-substituted 1,2,4-triazole compounds using glucose as a sustainable carbon source. This approach represents a paradigm shift from traditional petrochemical-dependent routes to biomass-utilizing strategies, offering a compelling value proposition for manufacturers of active pharmaceutical ingredients (APIs) and advanced intermediates. The 1,2,4-triazole scaffold is ubiquitous in medicinal chemistry, serving as the core structure for blockbuster drugs such as Sitagliptin and various antifungal and anticancer agents, as illustrated by the diverse bioactive molecules shown below.
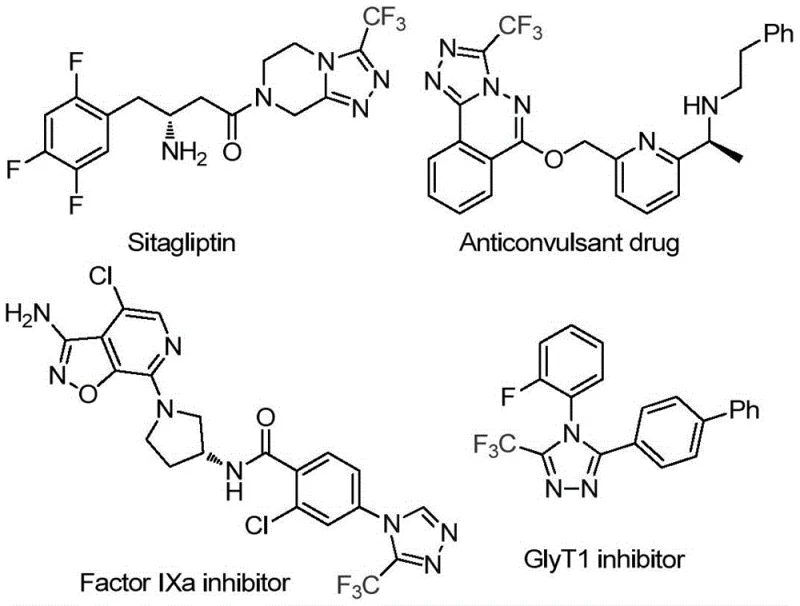
The strategic importance of this technology lies in its ability to merge high reaction efficiency with operational simplicity. By leveraging the natural abundance of glucose, the process addresses both economic and environmental pressures facing the modern chemical supply chain. For R&D directors and process chemists, this method offers a versatile platform for generating libraries of fluorinated heterocycles without the burden of exotic reagents or hazardous conditions. The patent explicitly highlights the mild reaction parameters and the ease of post-processing, which are critical factors when transitioning from laboratory discovery to commercial production. As we delve deeper into the technical specifics, it becomes evident that this glucose-mediated cascade reaction is not merely an academic curiosity but a viable industrial solution for the reliable production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to 3-trifluoromethyl-1,2,4-triazoles often rely on the condensation of amidrazones with carboxylic acid derivatives or the cyclization of hydrazones with nitriles, processes that frequently suffer from significant drawbacks. These conventional methods typically necessitate the use of expensive, moisture-sensitive reagents and often require harsh reaction conditions, such as high temperatures or strong bases, which can lead to poor atom economy and the generation of substantial chemical waste. Furthermore, many existing protocols demand strictly anhydrous and anaerobic environments to prevent side reactions or reagent decomposition, imposing heavy infrastructure costs on manufacturing facilities. The reliance on petrochemical-derived aldehydes or specialized building blocks also introduces supply chain volatility, as these precursors can be subject to market fluctuations and availability issues. Additionally, the purification of products from these complex reaction mixtures often involves tedious work-up procedures, including multiple extractions and recrystallizations, which further erodes overall yield and increases production time.
The Novel Approach
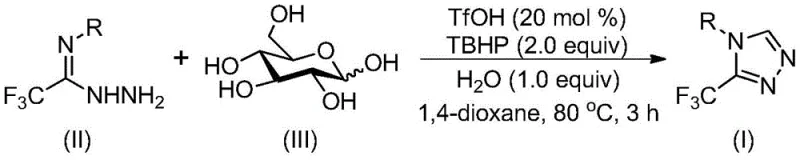
In stark contrast, the methodology described in patent CN113880781A utilizes a trifluoromethanesulfonic acid (TfOH) catalyzed cascade reaction that transforms inexpensive glucose directly into the requisite aldehyde intermediate in situ. This innovative strategy bypasses the need for isolated aldehyde precursors, effectively streamlining the synthetic sequence into a single pot operation. The reaction proceeds under remarkably mild conditions, typically between 70°C and 90°C, and notably tolerates the presence of water, eliminating the need for costly drying agents or inert atmosphere gloveboxes. The use of tert-butyl hydroperoxide (TBHP) as a terminal oxidant facilitates the final aromatization step efficiently, ensuring high conversion rates without the formation of toxic heavy metal byproducts. This approach not only simplifies the operational workflow but also enhances safety profiles by avoiding pyrophoric reagents. The general reaction scheme, depicting the transformation of trifluoroethylimide hydrazide and glucose into the target triazole, underscores the elegance and efficiency of this biomass-driven synthesis.
Mechanistic Insights into TfOH-Catalyzed Cascade Cyclization
The mechanistic pathway of this transformation is a sophisticated example of acid-catalyzed biomass valorization coupled with heterocyclic construction. Initially, the strong Brønsted acid catalyst, trifluoromethanesulfonic acid, promotes the cleavage of the glucose ring structure to generate a reactive aldehyde species. This in situ generated aldehyde immediately undergoes a condensation reaction with the trifluoroethylimide hydrazide to form a hydrazone intermediate. This step is crucial as it avoids the isolation of unstable aldehyde intermediates, thereby minimizing decomposition pathways. Following hydrazone formation, an intramolecular nucleophilic addition occurs, driving the cyclization process to form the dihydro-triazole ring system. The final step involves an oxidative aromatization mediated by TBHP, which removes two hydrogen atoms to establish the fully aromatic 1,2,4-triazole core. This cascade sequence is highly efficient because each step is thermodynamically driven forward, preventing the accumulation of partially reacted intermediates that could complicate purification.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in ensuring high product quality. Because the reaction does not require extreme thermal stress or aggressive basic conditions, the formation of degradation byproducts or polymerization tars is significantly suppressed. The specificity of the TfOH catalyst ensures that the glucose cleavage is directed towards the desired aldehyde functionality rather than random fragmentation, which enhances the regioselectivity of the subsequent cyclization. Furthermore, the compatibility of the system with aqueous media helps to solubilize polar byproducts and salts, facilitating their removal during the work-up phase. For R&D teams focused on impurity profiling, this mechanism suggests a cleaner crude reaction profile, which translates to reduced burden on downstream purification units like preparative HPLC or extensive recrystallization campaigns. The ability to tune the R-group on the hydrazide allows for the precise engineering of the electronic properties of the final triazole, enabling the synthesis of diverse analogues for structure-activity relationship (SAR) studies.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for scalability and ease of handling, making it accessible for both laboratory-scale optimization and pilot plant operations. The protocol involves charging a reactor with the hydrazide substrate, glucose, catalytic TfOH, and the oxidant in a suitable solvent such as 1,4-dioxane. The detailed standardized synthesis steps, including specific molar ratios and quenching procedures, are outlined in the guide below to ensure reproducibility and safety compliance.
- Combine trifluoroethylimide hydrazide, glucose, trifluoromethanesulfonic acid catalyst, and tert-butyl hydroperoxide oxidant in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to a temperature range of 70°C to 90°C and maintain stirring for a duration of 2 to 4 hours to ensure complete conversion.
- Upon completion, perform post-treatment involving filtration and silica gel column chromatography to isolate the pure 3-trifluoromethyl-substituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this glucose-based synthesis route offers tangible benefits that extend beyond simple yield metrics. The primary advantage lies in the drastic simplification of the raw material portfolio; replacing specialized, high-cost aldehydes with commodity-grade glucose creates a buffer against market volatility and price spikes associated with petrochemical feedstocks. This shift not only stabilizes the cost of goods sold (COGS) but also diversifies the supplier base, as glucose is globally sourced from multiple agricultural regions, thereby mitigating geopolitical supply risks. Moreover, the elimination of sensitive reagents that require cold chain logistics or specialized storage containers reduces warehousing costs and insurance premiums. The operational simplicity of the process, which foregoes the need for inert gas blanketing and rigorous moisture control, allows for the utilization of standard stainless steel reactors rather than specialized glass-lined or Hastelloy vessels, representing a significant capital expenditure saving for manufacturing sites.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the replacement of expensive synthetic building blocks with low-cost biomass. By utilizing glucose, a widely available and inexpensive sugar, the direct material costs are significantly lowered compared to traditional routes that rely on functionalized aryl aldehydes. Additionally, the catalytic nature of the trifluoromethanesulfonic acid means that only small quantities are needed to drive the reaction, further reducing reagent expenses. The high atom economy of the cascade reaction minimizes waste disposal costs, as fewer byproducts are generated per kilogram of product. This comprehensive reduction in input costs allows for a more competitive pricing structure for the final pharmaceutical intermediate, enhancing margin potential for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-hazardous starting materials that are not subject to strict regulatory controls or transportation restrictions. Unlike certain fluorinated reagents that may face shipping limitations, glucose and the hydrazide precursors are generally classified as safe for transport, simplifying logistics and reducing lead times for raw material delivery. The tolerance of the reaction to water and air means that production schedules are less likely to be disrupted by equipment failures related to drying systems or inert gas supply shortages. This reliability ensures consistent output volumes, allowing procurement teams to commit to longer-term supply agreements with confidence. The ability to source raw materials from multiple global vendors prevents single-source bottlenecks, ensuring continuity of supply even during regional disruptions.
- Scalability and Environmental Compliance: From a sustainability and scale-up perspective, this process aligns perfectly with modern green chemistry mandates and corporate ESG goals. The use of a renewable carbon source like glucose reduces the carbon footprint of the manufacturing process, appealing to environmentally conscious stakeholders and regulators. The reaction conditions are mild enough to be safely scaled from gram-level laboratory batches to multi-ton commercial production without exothermic runaway risks, facilitating a smoother technology transfer. The absence of heavy metal catalysts eliminates the need for complex and costly metal scavenging steps to meet strict residual metal specifications in pharmaceutical products. This streamlined workflow reduces the overall environmental load of the facility, lowering wastewater treatment costs and simplifying regulatory compliance reporting for environmental agencies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this glucose-mediated triazole synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of this technology. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of using glucose in this triazole synthesis?
A: Using glucose as a biomass-derived carbon source significantly reduces raw material costs compared to traditional synthetic aldehydes. It eliminates the need for complex pre-functionalization steps, simplifies the supply chain by utilizing a widely available commodity chemical, and aligns with green chemistry principles by leveraging renewable feedstocks.
Q: Does this method require strict anhydrous or anaerobic conditions?
A: No, one of the distinct operational advantages of this patented process is that it does not require stringent anhydrous or oxygen-free environments. The reaction tolerates the presence of water, which is actually added as an additive to improve efficiency, thereby reducing equipment complexity and operational overhead for manufacturing teams.
Q: What is the substrate scope for the R-group in this synthesis?
A: The method demonstrates broad substrate tolerance, accommodating various substituted aryl groups including those with methyl, methoxy, methylthio, halogen, or trifluoromethyl substituents at ortho, meta, or para positions. It also supports phenethyl groups, allowing for the diverse functionalization required in modern drug discovery and development pipelines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biomass-derived synthetic routes in the production of high-value pharmaceutical intermediates. Our team of expert process chemists has extensively evaluated the glucose-mediated synthesis of 3-trifluoromethyl-1,2,4-triazoles and confirmed its viability for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of trifluoromethyl-triazole intermediate delivered meets the highest industry standards for potency and impurity profiles.
We invite global partners to collaborate with us to leverage this cost-effective and sustainable technology for their drug development programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data for our triazole intermediates and to discuss route feasibility assessments for your custom synthesis projects. Let us help you optimize your supply chain and reduce your time-to-market with our advanced manufacturing capabilities.
