Advanced Palladium-Catalyzed Synthesis of 2-Vinyl Indoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds with high efficiency and minimal environmental impact. Patent CN109134343B introduces a transformative approach to the alkenylation of the indole C-2 position, a critical structural motif found in numerous bioactive molecules and agrochemical agents. This technology leverages a palladium-catalyzed coupling between geminal dibromostyrene derivatives and hydrazones to generate 2-vinyl indoles in a single operational step. By circumventing the need for harsh cryogenic conditions and unstable organolithium reagents typical of legacy syntheses, this innovation represents a significant leap forward for any reliable pharmaceutical intermediate supplier aiming to optimize their production pipelines. The method not only achieves exceptional conversion rates but also ensures that the physical and chemical properties of the resulting products remain stable across varying temperature environments, a crucial factor for downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the indole core at the C-2 position has been fraught with significant synthetic challenges that hinder large-scale adoption. Traditional pathways often rely on the introduction of ketone groups followed by reduction, or the use of highly reactive n-butyllithium to install isopropanolate groups under trifluoroacetic acid conditions. These conventional routes are inherently problematic due to their multi-step nature, which drastically reduces overall throughput and increases the accumulation of waste. Furthermore, the requirement for stringent reaction conditions, such as extremely low temperatures to manage the reactivity of organolithium species, imposes heavy energy burdens and safety risks on manufacturing facilities. The poor atom economy associated with these methods, coupled with the difficulty in removing specific regio-inductive groups, results in lower yields and higher purification costs, making them economically unviable for the cost reduction in API manufacturing that modern supply chains demand.
The Novel Approach
In stark contrast, the methodology disclosed in CN109134343B offers a streamlined, one-pot solution that elegantly bypasses these historical bottlenecks. By utilizing geminal dibromostyrene and hydrazone derivatives as primary building blocks, the reaction proceeds efficiently under the influence of a palladium catalyst and a mild base like lithium tert-butoxide. This novel approach eliminates the necessity for regio-inductive groups, thereby simplifying the molecular design and reducing the number of synthetic operations required. The process operates at a moderate temperature range of 100-115°C, which is far more accessible and safer than cryogenic alternatives. Additionally, the ability to recycle the transition metal catalyst from the inorganic phase post-reaction adds a layer of sustainability and economic efficiency that is rarely seen in traditional cross-coupling protocols, positioning this technology as a superior choice for the commercial scale-up of complex heterocycles.
Mechanistic Insights into Pd-Catalyzed C-2 Alkenylation
The core of this technological breakthrough lies in the sophisticated palladium catalytic cycle that facilitates the formation of the carbon-carbon bond at the indole C-2 position. The mechanism initiates with the oxidative addition of the palladium catalyst into the carbon-bromine bond of the geminal dibromostyrene substrate, generating a reactive organopalladium intermediate. This species subsequently coordinates with the hydrazone derivative, setting the stage for a migratory insertion or similar coupling event that constructs the vinyl framework. The presence of the base is critical in facilitating the elimination steps necessary to restore aromaticity and finalize the 2-vinyl indole structure. Understanding this mechanistic pathway is vital for R&D teams, as it highlights the precise role of each reagent and allows for fine-tuning of reaction parameters to maximize selectivity and minimize side reactions.
From an impurity control perspective, this catalytic system exhibits remarkable specificity, largely due to the inherent stability of the intermediates formed during the cycle. The reaction conditions are optimized to suppress competing pathways, such as homocoupling of the styrene derivative or decomposition of the hydrazone, which are common pitfalls in less refined systems. The patent data indicates that by-products are minimal and can be easily removed via conventional purification methods like column chromatography or crystallization. This high level of chemoselectivity ensures that the final product meets the rigorous purity standards required for high-purity indole derivatives used in sensitive biological applications. The robustness of the catalyst system also means that variations in substrate electronic properties, such as electron-withdrawing or donating groups on the aromatic rings, are well-tolerated without compromising the integrity of the final molecule.
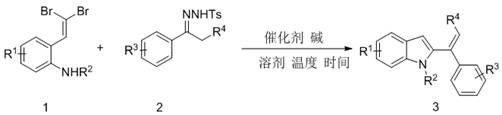
How to Synthesize 2-Vinyl Indole Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to replicate the high yields reported in the patent literature. The process is designed to be operationally simple, involving the sequential addition of substrates, catalyst, and base into a suitable solvent system before heating. For research and development teams looking to adopt this methodology, the following guide outlines the fundamental procedural steps derived directly from the exemplary embodiments. It is essential to maintain the specified molar ratios, particularly the excess of hydrazone and base, to drive the reaction to completion and ensure the efficient consumption of the valuable dibromostyrene starting material.
- Combine geminal dibromostyrene derivative and hydrazone derivative in a reaction vessel with a palladium catalyst such as PdCl2(PPh3)2.
- Add lithium tert-butoxide as the base and dioxane as the solvent, ensuring a molar ratio of approximately 1: 2 for substrates and 3 equivalents of base.
- Heat the mixture to 110°C for 1 hour, then perform standard aqueous workup and column chromatography to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The shift away from hazardous and expensive reagents like n-butyllithium translates directly into a safer working environment and reduced expenditure on specialized containment and handling equipment. Moreover, the simplification of the workflow from a multi-step sequence to a single-pot reaction significantly compresses the manufacturing timeline, allowing for faster turnaround times and improved responsiveness to market demands. The ability to recover and reuse the palladium catalyst further mitigates the impact of fluctuating precious metal prices, providing a more predictable cost structure for long-term production planning.
- Cost Reduction in Manufacturing: The elimination of cryogenic cooling requirements and the reduction in synthetic steps lead to substantial energy savings and lower labor costs per kilogram of product. By avoiding the use of transient protecting groups and complex workup procedures associated with older methods, the overall material throughput is improved, resulting in significant cost optimization without the need for capital-intensive new infrastructure.
- Enhanced Supply Chain Reliability: The starting materials, specifically geminal dibromostyrene and various hydrazones, are industrially available commodities with stable supply lines, reducing the risk of raw material shortages. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in environmental controls, ensuring consistent batch-to-bquality and reliable delivery schedules for downstream clients.
- Scalability and Environmental Compliance: The one-step nature of the reaction facilitates easier scale-up from laboratory to pilot and commercial scales, as there are fewer unit operations to manage and optimize. Furthermore, the generation of fewer by-products and the potential for catalyst recycling align with green chemistry principles, simplifying waste treatment processes and ensuring compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkenylation technology. These insights are derived from the detailed specifications and experimental data provided in the patent documentation, aimed at clarifying the operational parameters and potential advantages for industrial partners. Understanding these nuances is key to evaluating the fit of this technology within existing manufacturing portfolios.
Q: What are the advantages of this method over traditional n-butyllithium routes?
A: Unlike traditional methods requiring hazardous n-butyllithium and cryogenic conditions, this Pd-catalyzed process operates at moderate temperatures (110°C) with safer, commercially available reagents, significantly improving operational safety and atom economy.
Q: Is the palladium catalyst recoverable in this process?
A: Yes, the patent explicitly states that the transition metal catalyst can be recovered and reused from the inorganic phase after extraction, which supports cost-effective and environmentally compliant manufacturing practices.
Q: What is the substrate scope for R groups on the indole ring?
A: The method demonstrates broad tolerance, accommodating various substituents including H, F, Cl, Br, methoxy, cyano, and nitro groups on the aromatic rings, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Vinyl Indole Supplier
As the global demand for complex heterocyclic intermediates continues to rise, partnering with an experienced CDMO is essential for navigating the complexities of process development and commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and purity of every batch, guaranteeing that the 2-vinyl indole derivatives you receive meet the highest industry standards.
We invite you to engage with our technical procurement team to discuss how this innovative palladium-catalyzed route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain.
