Advanced Four-Step Synthesis of TPCA-1 Inhibitor for Scalable Pharmaceutical Production
Advanced Four-Step Synthesis of TPCA-1 Inhibitor for Scalable Pharmaceutical Production
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing potent kinase inhibitors, particularly for treating inflammatory diseases like rheumatoid arthritis. A significant breakthrough in this domain is detailed in Chinese patent CN109438416B, which discloses a novel method for synthesizing the thiophene-containing inhibitor TPCA-1 (CAS: 507475-17-4). This compound acts as a selective IKK-2 inhibitor, playing a crucial role in regulating pro-inflammatory molecules via the NF-kB pathway. The patented technology represents a strategic shift from traditional multi-step syntheses to a streamlined four-step process. By utilizing thiourea and 2-(4-fluorophenyl)cyclopropane-1,1-dimethyl dicarboxylate as起始 materials, the method leverages a sequence of ring expansion, oxidation, addition, and amidation reactions. This approach not only simplifies the operational complexity but also addresses critical supply chain pain points associated with precursor availability and reaction safety, positioning it as a highly viable route for reliable pharmaceutical intermediate supplier networks aiming for high-purity output.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of TPCA-1 was fraught with significant technical and logistical challenges that hindered efficient commercial scale-up of complex pharmaceutical intermediates. The conventional methodology typically involved a five-step reaction sequence, which inherently increased the cumulative loss of material and extended the overall production timeline. A major drawback of the traditional route was the reliance on 2,5-dihydroxy-1,4-dithiane as a starting material. This reagent is notorious for its extremely foul odor, posing severe occupational health and safety risks in a manufacturing environment and complicating waste management protocols. Furthermore, the reaction conditions in the legacy process were often relatively harsh, requiring stringent controls that drove up energy consumption and equipment maintenance costs. The complexity of the five-step pathway also meant a higher probability of generating difficult-to-remove impurities, thereby increasing the burden on downstream purification processes and negatively impacting the final cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
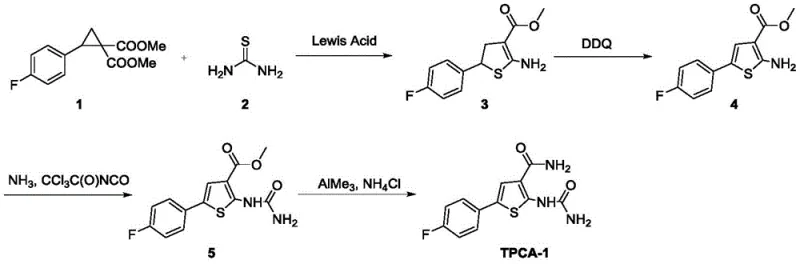
In stark contrast, the novel approach outlined in the patent introduces a robust four-step synthetic strategy that effectively circumvents the drawbacks of the legacy methods. The process initiates with the condensation of thiourea and a fluorophenyl-substituted cyclopropane derivative, catalyzed by a Lewis acid. This ring expansion reaction constructs the thiophene core directly, eliminating the need for the malodorous dithiane precursor entirely. The subsequent steps involve a mild oxidation using DDQ to aromatize the ring, followed by a urea installation via trichloroacetyl isocyanate, and finally, an amidation step using trimethylaluminum. This streamlined pathway significantly shortens the synthetic timeline and operates under much milder conditions, with temperatures generally ranging from ambient to 90°C. By reducing the step count and utilizing stable, commercially available reagents, this method offers a superior alternative for reducing lead time for high-purity pharmaceutical intermediates while maintaining excellent control over the impurity profile.
Mechanistic Insights into Yb(OTf)3-Catalyzed Ring Expansion and Functionalization
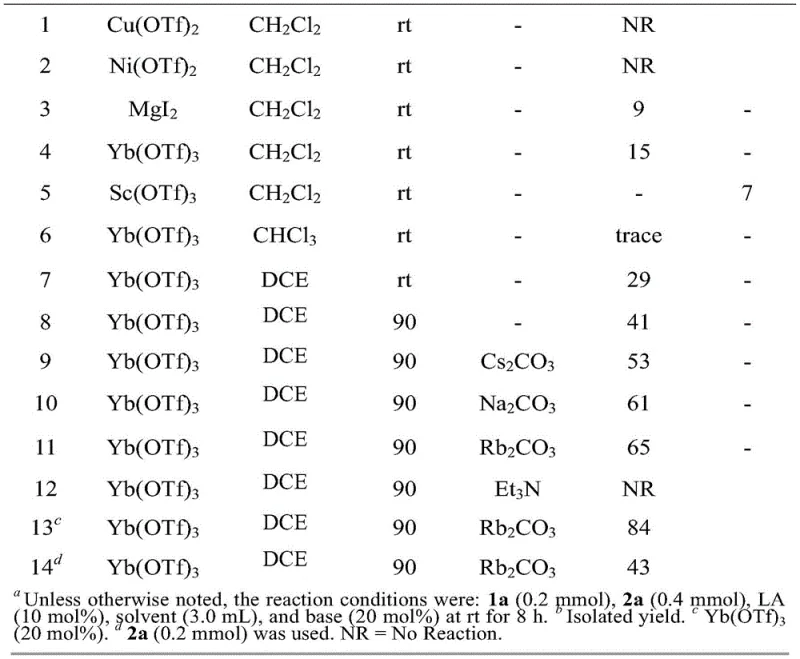
The cornerstone of this synthetic innovation lies in the first step: the Lewis acid-catalyzed ring expansion of the cyclopropane derivative. The patent identifies Ytterbium triflate (Yb(OTf)3) as the optimal catalyst, functioning as a potent Lewis acid that activates the cyclopropane ring towards nucleophilic attack by thiourea. This activation facilitates the cleavage of the strained three-membered ring and the subsequent formation of the five-membered dihydrothiophene structure. The choice of base is equally critical; screening revealed that Rubidium carbonate (Rb2CO3) outperforms other bases like cesium or sodium carbonate, likely due to its specific solubility profile and basicity in 1,2-dichloroethane (DCE). The reaction proceeds efficiently at 90°C, yielding the amino-dihydrothiophene ester with high selectivity. Following this, the oxidation step employs DDQ (2,3-Dichloro-5,6-dicyano-1,4-benzoquinone) to dehydrogenate the dihydrothiophene intermediate. This oxidative aromatization is clean and effective, converting the saturated ring into the fully aromatic thiophene system required for the biological activity of the final inhibitor without over-oxidizing sensitive functional groups.
The latter half of the synthesis focuses on installing the urea and amide functionalities with precision. The urea group is introduced by reacting the amino-thiophene ester with trichloroacetyl isocyanate, which acts as an activated isocyanate source. This addition reaction occurs smoothly at low temperatures (-10°C to -30°C initially), preventing side reactions, followed by ammonolysis to complete the urea formation. The final transformation involves converting the methyl ester to the primary carboxamide. Instead of traditional hydrolysis followed by coupling, the patent utilizes a direct amidation strategy using trimethylaluminum (AlMe3) and ammonium chloride. This reagent combination generates an aluminum-amide species in situ that attacks the ester carbonyl directly. This method is particularly advantageous for sterically hindered or sensitive substrates, ensuring high conversion rates to the final TPCA-1 product. The mechanistic elegance of this route ensures that impurities are minimized at each stage, facilitating easier purification and higher overall purity.
How to Synthesize TPCA-1 Efficiently
The synthesis of TPCA-1 via this patented route requires careful attention to reaction parameters, particularly in the initial ring expansion step where catalyst loading and base selection dictate success. The process begins with the mixing of the cyclopropane starting material and thiourea in a solvent such as dichloroethane, followed by the addition of the Yb(OTf)3 catalyst and Rb2CO3 base. Heating this mixture to 90°C for approximately 8 hours drives the ring expansion to completion. Subsequent steps involve standard workup procedures including extraction and column chromatography to isolate intermediates, although in a continuous flow or telescoped process, these could be further optimized. The detailed standardized synthetic steps, including precise stoichiometric ratios, quenching protocols, and purification methods for each of the four stages, are provided in the guide below to ensure reproducibility and safety in a laboratory or pilot plant setting.
- Perform Lewis acid catalyzed ring expansion of dimethyl 2-(4-fluorophenyl)cyclopropane-1,1-dicarboxylate with thiourea using Yb(OTf)3 and Rb2CO3 in DCE at 90°C.
- Oxidize the resulting dihydrothiophene intermediate using DDQ in dichloromethane to form the aromatic thiophene ester.
- React the thiophene ester with trichloroacetyl isocyanate followed by ammonolysis to install the ureido group.
- Convert the methyl ester to the primary carboxamide using trimethylaluminum and ammonium chloride in toluene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits beyond mere chemical efficiency. The elimination of 2,5-dihydroxy-1,4-dithiane removes a significant logistical and safety hurdle, as sourcing and handling malodorous sulfur compounds often require specialized containment facilities and incur higher disposal costs. By switching to thiourea and cyclopropane derivatives, the supply chain becomes more robust, relying on commodity chemicals that are widely available from multiple global vendors. This diversification of raw material sources mitigates the risk of supply disruptions and stabilizes pricing volatility. Furthermore, the reduction from five steps to four steps inherently improves the overall mass balance of the process. Fewer unit operations mean less solvent consumption, reduced energy usage for heating and cooling, and lower labor hours per kilogram of product, all of which contribute to a leaner cost structure without compromising quality.
- Cost Reduction in Manufacturing: The economic impact of this new route is driven primarily by the simplification of the process flow and the removal of expensive or difficult-to-handle reagents. By avoiding the use of the foul-smelling dithiane, manufacturers save significantly on environmental compliance costs and specialized waste treatment fees. Additionally, the high yields reported in the patent examples for each step suggest a superior atom economy compared to older methods. The use of Yb(OTf)3, while a specialized catalyst, is employed in catalytic amounts (10-20 mol%), and its effectiveness allows for milder reaction conditions that reduce energy overheads. The direct amidation using trimethylaluminum also bypasses the need for separate hydrolysis and activation steps, further consolidating the process and reducing the consumption of coupling reagents and solvents, leading to substantial cost savings in the final production budget.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical API intermediates is paramount for downstream drug formulation. This synthesis method enhances reliability by utilizing starting materials that are chemically stable and easy to store, such as thiourea and the fluorophenyl cyclopropane ester. Unlike the sensitive and odorous precursors of the past, these materials have longer shelf lives and do not require exotic storage conditions. The robustness of the reaction conditions, particularly the tolerance of the Lewis acid catalyst system, means that the process is less prone to batch failures due to minor fluctuations in temperature or moisture. This consistency ensures a steady output of high-quality intermediate, allowing supply chain planners to maintain tighter inventory controls and meet delivery schedules with greater confidence, ultimately supporting the continuous manufacturing needs of pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden bottlenecks, but this route is designed with scalability in mind. The reactions utilize common organic solvents like dichloroethane, dichloromethane, and toluene, which are standard in industrial settings and have well-established recovery and recycling protocols. The absence of extreme temperatures or high-pressure requirements simplifies the engineering controls needed for the reactors. From an environmental perspective, the shorter synthetic path generates less chemical waste per unit of product. The avoidance of heavy metal catalysts in later steps (using aluminum instead) and the efficient use of oxidants like DDQ contribute to a greener process profile. This alignment with green chemistry principles facilitates easier regulatory approval and supports the sustainability goals of modern chemical enterprises, making it an attractive option for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of TPCA-1 as described in the patent literature. These answers are derived directly from the experimental data and technical disclosures to provide clarity on the feasibility and advantages of this method. Understanding these details is essential for R&D teams evaluating process transfer and for procurement specialists assessing the viability of this route for long-term sourcing strategies. The information covers catalyst selection, reaction conditions, and the comparative benefits over traditional synthesis methods.
Q: What are the key advantages of the new TPCA-1 synthesis route over conventional methods?
A: The new route described in patent CN109438416B reduces the synthesis from five steps to four steps and eliminates the use of foul-smelling 2,5-dihydroxy-1,4-dithiane. It utilizes readily available starting materials like thiourea and achieves high yields under milder reaction conditions.
Q: Which catalyst system provides the optimal yield for the ring expansion step?
A: Optimization studies indicate that Ytterbium triflate (Yb(OTf)3) combined with Rubidium carbonate (Rb2CO3) in 1,2-dichloroethane (DCE) at 90°C provides the highest yield for the initial ring expansion reaction.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is designed for scalability. It avoids harsh conditions, uses common organic solvents like DCE and toluene, and employs robust reagents like DDQ and trimethylaluminum, making it feasible for cost reduction in pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable TPCA-1 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value pharmaceutical intermediates like TPCA-1. Our technical team has thoroughly analyzed the patented methodology and possesses the expertise to implement this four-step process with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of TPCA-1 meets the exacting standards required for clinical and commercial applications. We are committed to delivering consistent quality and reliability, leveraging our deep understanding of Lewis acid catalysis and heterocyclic chemistry to serve your project needs effectively.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Whether you require pilot-scale quantities for clinical trials or metric tons for commercial launch, NINGBO INNO PHARMCHEM is prepared to be your strategic partner in delivering high-purity TPCA-1 with unmatched speed and efficiency.
