Advanced Synthetic Route for Alpha-Alkynyl Ethers: Scaling High-Purity Pharmaceutical Intermediates
Introduction: A Breakthrough in Heterocyclic Synthesis
The landscape of medicinal chemistry is constantly evolving, driven by the need for more efficient and sustainable pathways to access bioactive scaffolds. Among these, α-alkynyl-substituted ether compounds represent a critical structural motif, found in over 20% of the top 200 small-molecule drugs. Recognizing this demand, recent intellectual property developments, specifically patent CN107556269B, have unveiled a robust and environmentally friendly methodology for constructing these valuable cores. This technology addresses long-standing challenges in organic synthesis by replacing harsh conditions and expensive reagents with a mild, radical-mediated approach. For industry leaders seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this synthetic route is paramount for securing future supply chains. The method not only streamlines the construction of the ether linkage but also ensures high functional group tolerance, making it applicable to a diverse range of drug candidates.
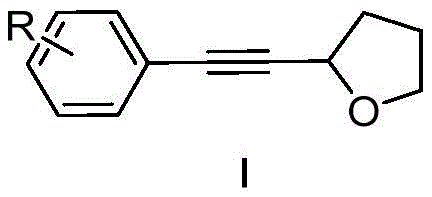
The significance of this patent lies in its ability to utilize simple alkynyl esters as alkynylation reagents, a strategy that markedly simplifies the precursor synthesis compared to historical methods. By leveraging tetrahydrofuran (THF) both as a solvent and a reactant, the process achieves a high degree of atom economy. This dual role of THF reduces waste generation and lowers the overall material costs associated with large-scale manufacturing. Furthermore, the reaction proceeds under nitrogen protection at room temperature, eliminating the energy-intensive heating steps required by previous methodologies. For procurement managers focused on cost reduction in API manufacturing, this shift towards milder conditions translates directly into lower operational expenditures and enhanced safety profiles within the production facility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of α-alkynyl-substituted ethers has been plagued by significant technical hurdles that impede efficient commercialization. Early approaches, such as those reported by Fuchs in 1996, relied on alkynyl trifluoromethylsulfonyl reagents which are not only expensive but also pose handling difficulties due to their reactivity. Subsequent methods introduced by Anderson in 2011 utilized palladium catalysts for the synthesis of 2-alkynyl oxocycles; however, these routes often suffered from complex substrate requirements and the necessity for rigorous removal of heavy metal residues. The presence of palladium in the final active pharmaceutical ingredient (API) is strictly regulated, necessitating additional purification steps that drive up costs and extend lead times. Moreover, other metal-free alternatives reported by Wang in 2014 required reaction temperatures as high as 150°C, creating substantial safety risks and limiting the compatibility with thermally sensitive functional groups commonly found in advanced drug intermediates.
The Novel Approach
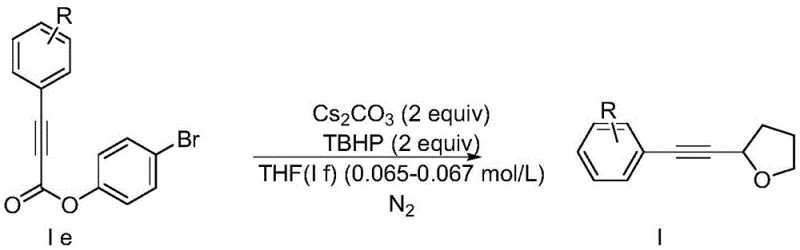
In stark contrast to these legacy techniques, the methodology disclosed in CN107556269B introduces a paradigm shift through a three-step sequence that prioritizes simplicity and green chemistry principles. The core innovation lies in the final cyclization step, where a substituted phenylpropiolic acid p-bromophenyl ester undergoes a radical addition-elimination reaction with THF. This transformation is facilitated by tert-butyl hydroperoxide (TBHP) and cesium carbonate, avoiding the use of transition metals entirely in the ring-forming stage. The mild conditions (20-30°C) ensure that delicate molecular architectures remain intact, thereby expanding the scope of accessible chemical space for R&D teams. By employing readily available alkynyl esters instead of complex hypervalent iodine reagents, the supply chain becomes more resilient and less susceptible to raw material shortages. This novel approach effectively decouples the complexity of the synthesis from the cost of goods, offering a scalable solution for producing high-value heterocycles.
Mechanistic Insights into Radical-Mediated Alkynylation
To fully appreciate the technical superiority of this route, one must delve into the mechanistic underpinnings of the radical alkynylation step. The reaction initiates with the homolytic cleavage of tert-butyl hydroperoxide (TBHP), likely promoted by the basic environment provided by cesium carbonate. This generates tert-butoxyl radicals which subsequently abstract a hydrogen atom from the alpha-position of the tetrahydrofuran solvent. The resulting alpha-oxy radical is nucleophilic in character and attacks the electron-deficient triple bond of the alkynyl ester moiety. This addition forms a vinyl radical intermediate, which then undergoes a fragmentation process to eliminate the p-bromophenoxy radical, restoring the triple bond and closing the ether ring. This elegant cascade avoids the formation of stable byproducts that typically complicate purification. The use of cesium carbonate is critical here, as it acts as a base to neutralize acidic byproducts and may also assist in the activation of the peroxide species, ensuring a steady flux of radicals throughout the 8 to 12-hour reaction window.
From an impurity control perspective, this mechanism offers distinct advantages over ionic pathways. Since the reaction proceeds via discrete radical intermediates rather than charged species, there is minimal risk of competing nucleophilic substitutions or eliminations that often plague ionic chemistry. The specificity of the hydrogen abstraction from THF ensures that the ether ring is formed regioselectively at the 2-position. Furthermore, the absence of heavy metal catalysts in this key step means that the impurity profile is devoid of toxic metal traces, a crucial factor for regulatory compliance in pharmaceutical manufacturing. The robustness of this radical process allows for excellent functional group tolerance, accommodating substituents such as methyl, chloro, and biphenyl groups on the aromatic ring without significant degradation in yield. This predictability is invaluable for process chemists aiming to lock down a synthesis route for clinical trial materials.
How to Synthesize 2-(Phenylethynyl)tetrahydrofuran Efficiently
The practical implementation of this technology involves a streamlined three-step protocol that can be adapted for both laboratory and pilot-scale operations. The process begins with the preparation of the alkynyl acid precursor, followed by activation as a bromophenyl ester, and concludes with the radical cyclization. Each step has been optimized to maximize yield while minimizing waste, utilizing standard reagents that are globally sourced. The detailed standardized synthesis steps, including precise stoichiometry and workup procedures, are outlined below to guide technical teams in replicating this high-efficiency route.
- Perform a palladium-catalyzed coupling between propynoic acid and substituted iodobenzene using DBU in DMSO to generate substituted phenylpropiolic acid.
- Conduct an esterification reaction between the resulting phenylpropiolic acid and p-bromophenol using DCC and DMAP in dichloromethane.
- Execute a radical alkynylation reaction using the bromophenyl ester, tetrahydrofuran, cesium carbonate, and TBHP under nitrogen protection to yield the final ether product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology presents a compelling value proposition centered on cost efficiency and operational reliability. The elimination of expensive transition metal catalysts in the final cyclization step removes the need for specialized scavenging resins or complex filtration processes, directly reducing the cost of goods sold (COGS). Additionally, the use of tetrahydrofuran as both solvent and reactant simplifies the bill of materials, reducing inventory complexity and storage requirements. The mild reaction temperatures further contribute to energy savings, as no heating infrastructure is required for the key transformation step. These factors collectively enhance the economic viability of producing alpha-alkynyl ethers at a commercial scale, making it an attractive option for long-term supply agreements.
- Cost Reduction in Manufacturing: The strategic replacement of hypervalent iodine reagents and palladium catalysts with inexpensive organic peroxides and cesium salts drives a substantial decrease in raw material costs. By avoiding the use of precious metals in the final step, the process eliminates the financial burden associated with metal recovery and validation testing for residual metals. This simplification of the chemical workflow allows for a more lean manufacturing model, where resources are allocated towards yield optimization rather than impurity management. Consequently, the overall production cost is significantly lowered, providing a competitive edge in the pricing of fine chemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as propynoic acid, iodobenzene derivatives, and THF ensures a stable and diversified supply base. Unlike specialized reagents that may be subject to single-source bottlenecks, these starting materials are produced by multiple global vendors, mitigating the risk of supply disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain. This resilience is critical for maintaining continuous production schedules and meeting the just-in-time delivery expectations of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The green chemistry attributes of this method align perfectly with modern environmental, social, and governance (ESG) goals. The absence of heavy metals and the use of ambient temperature conditions reduce the environmental footprint of the manufacturing process. Waste generation is minimized through the dual use of THF and the high atom economy of the radical coupling. This facilitates easier regulatory approval and reduces the costs associated with waste disposal and treatment. The process is inherently scalable, allowing for seamless transition from kilogram-level development to multi-ton commercial production without the need for major equipment modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these alpha-alkynyl ether compounds. The answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production pipelines.
Q: What are the primary advantages of this synthesis method over traditional palladium-catalyzed etherification?
A: Unlike traditional methods that often require harsh conditions (up to 150°C) or complex hypervalent iodine reagents, this patented route operates at room temperature (20-30°C) in the final step. It eliminates the need for heavy metal catalysts in the cyclization stage, significantly simplifying purification and reducing environmental impact.
Q: Is this process scalable for commercial production of pharmaceutical intermediates?
A: Yes, the process utilizes readily available starting materials like propynoic acid and tetrahydrofuran, which also serves as the solvent. The mild reaction conditions and absence of expensive transition metals in the final step make it highly suitable for commercial scale-up of complex heterocycles.
Q: What is the expected purity and yield profile for these alpha-alkynyl ethers?
A: Experimental data indicates isolated yields ranging from 51% to 75% depending on the substituent. The use of standard column chromatography purification ensures high-purity alpha-alkynyl ethers suitable for downstream medicinal chemistry applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Alkynyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development lifecycle. Our team of expert process chemists has thoroughly analyzed the synthetic route described in CN107556269B and is fully prepared to execute this methodology at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alpha-alkynyl ether delivered meets the highest industry standards. We are committed to being a partner that not only supplies chemicals but also provides technical solutions that accelerate your time to market.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to build a more resilient and cost-effective supply chain for your next-generation pharmaceutical products.
