Advanced Photocatalytic Synthesis of Difluoroalkyl Intermediates for Pharmaceutical Applications
The pharmaceutical and agrochemical industries continuously seek robust methodologies to enhance the metabolic stability and oral bioavailability of bioactive molecules. Patent CN116375611A introduces a groundbreaking approach for synthesizing difluoroalkyl compounds directly from aliphatic amines, addressing a critical gap in organic synthesis. The selective introduction of difluoromethylene (CF2) groups is paramount in modern medicinal chemistry, as evidenced by high-profile drugs like Lubiprostone and Ivosidenib, which rely on these motifs for efficacy. This patent discloses a method that operates under nitrogen protection, utilizing a fluorinating reagent and a catalyst in a reaction solvent, driven by illumination to achieve the transformation efficiently.
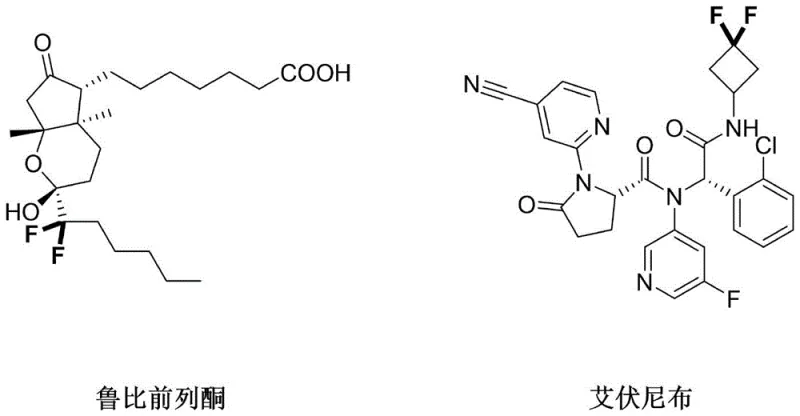
The significance of this technology lies in its ability to functionalize complex substrates late in the synthesis sequence, avoiding the need for pre-functionalized starting materials. By leveraging visible light irradiation, the process activates the C(sp3)-H bonds on aliphatic alkyl chains, a transformation that has historically been challenging due to the high inertness of these bonds. This innovation provides a streamlined pathway for R&D teams aiming to incorporate fluorine atoms into lead compounds without compromising structural integrity or generating excessive hazardous byproducts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for introducing difluoroalkyl groups often suffer from significant drawbacks that hinder their application in large-scale manufacturing. Conventional methods frequently require harsh reaction conditions, such as extreme temperatures or the use of highly reactive and dangerous reagents that pose safety risks in a production environment. Furthermore, many existing protocols necessitate pre-functionalization of the substrate, adding extra synthetic steps that increase overall cost and reduce atom economy. The direct difluorination of aliphatic chains has been particularly elusive, with prior art often limited to specific activated positions like alpha-carbonyls or heterobenzylic sites, leaving simple alkyl chains difficult to modify efficiently.
The Novel Approach
In contrast, the method described in CN116375611A offers a mild and environmentally friendly alternative that overcomes these historical barriers. By employing a photocatalytic system with anthraquinone or its derivatives, the reaction proceeds smoothly at temperatures ranging from 0°C to 50°C, with a preferred range of 25°C to 35°C. This gentle thermal profile minimizes thermal degradation of sensitive functional groups, ensuring higher purity profiles for the final intermediates. The use of visible light sources, such as blue, white, or purple light, drives the reaction mechanism without the need for expensive transition metal catalysts, significantly simplifying the downstream purification process and reducing the burden of heavy metal removal.

Mechanistic Insights into Anthraquinone-Catalyzed Photofluorination
The core of this innovative synthesis lies in the photocatalytic cycle mediated by anthraquinone derivatives. Upon irradiation with visible light, the anthraquinone catalyst absorbs photons and transitions to an excited state, enabling it to interact with the fluorinating reagent, such as Selectfluor or NFSI. This interaction generates reactive fluorine radicals or cationic species capable of abstracting hydrogen atoms from the inert C(sp3)-H bonds of the aliphatic amine substrate. The resulting carbon-centered radical then undergoes further fluorination to install the second fluorine atom, completing the difluorination sequence. This radical mechanism is highly selective and operates under mild conditions, preserving the stereochemistry and integrity of other sensitive moieties within the molecule.
Impurity control is a critical aspect of this mechanism, as the mild reaction conditions inherently suppress side reactions that typically occur at elevated temperatures. The use of specific solvent systems, such as mixtures of 2,2,2-trifluoroethanol with acetonitrile or ethyl acetate, further stabilizes the reactive intermediates and enhances the solubility of the fluorinating agents. By optimizing the molar ratios of the amine, fluorinating reagent, and catalyst, typically around 1:2.5-3.5:0.01-0.05, the process maximizes conversion while minimizing the formation of over-fluorinated or decomposed byproducts. This precise control over the reaction environment ensures a clean impurity profile, which is essential for meeting the stringent quality standards required for pharmaceutical intermediates.
How to Synthesize Difluoroalkyl Compounds Efficiently
To implement this synthesis effectively, operators must adhere to strict procedural guidelines regarding atmosphere control and light exposure. The reaction is initiated by combining the aliphatic amine, fluorinating reagent, and anthraquinone catalyst in a mixed solvent system under a nitrogen atmosphere to prevent oxidative side reactions. Detailed standardized synthesis steps follow below to ensure reproducibility and safety during scale-up operations.
- Prepare the reaction mixture by adding the aliphatic amine substrate, fluorinating reagent (Selectfluor or NFSI), and anthraquinone catalyst into a suitable solvent system under nitrogen protection.
- Irradiate the reaction mixture with a visible light source (blue, white, or purple light) while stirring at a controlled temperature between 0°C and 50°C.
- Upon completion, concentrate the reaction solution under reduced pressure and purify the crude product via chromatographic column separation to isolate the target difluoroalkyl compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology presents substantial opportunities for optimizing manufacturing costs and securing reliable supply lines. The elimination of expensive transition metal catalysts removes the need for costly scavenging resins and extensive purification steps typically required to meet residual metal specifications in API production. Additionally, the operation at near-ambient temperatures drastically reduces energy consumption associated with heating and cooling cycles, contributing to a lower carbon footprint and reduced utility costs per kilogram of product manufactured.
- Cost Reduction in Manufacturing: The process utilizes readily available and inexpensive anthraquinone catalysts alongside common fluorinating reagents, avoiding the need for proprietary or exotic materials that drive up raw material costs. The simplified workup procedure, involving concentration and chromatography, reduces solvent usage and labor hours compared to multi-step traditional sequences. These factors combine to deliver significant cost savings in difluoroalkyl compound manufacturing without compromising on quality or yield.
- Enhanced Supply Chain Reliability: By relying on stable and commercially available starting materials like aliphatic amines and Selectfluor, the risk of supply chain disruptions due to raw material scarcity is minimized. The robustness of the reaction conditions allows for flexible production scheduling, as the process is less sensitive to minor fluctuations in environmental parameters. This reliability ensures consistent delivery timelines for high-purity difluoroalkyl intermediates, supporting uninterrupted downstream drug development and commercialization efforts.
- Scalability and Environmental Compliance: The method is designed with scalability in mind, utilizing standard photochemical reactors that can be adapted for larger batch sizes. The reduction in hazardous waste generation, achieved by avoiding pre-functionalization steps and harsh reagents, aligns with increasingly strict environmental regulations. This compliance facilitates smoother regulatory approvals and reduces the costs associated with waste disposal, making the process sustainable for long-term commercial production of complex pharmaceutical additives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to assist R&D and technical procurement teams in evaluating feasibility.
Q: What are the key advantages of this photocatalytic method over traditional difluorination techniques?
A: This method utilizes mild reaction conditions (0-50°C) and visible light irradiation, avoiding the harsh conditions and pre-functionalization steps often required in conventional strategies, thereby reducing hazardous waste generation.
Q: Which fluorinating reagents are compatible with this synthesis protocol?
A: The protocol is versatile and supports the use of common fluorinating reagents such as Selectfluor (1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane bis(tetrafluoroborate) salt) and NFSI (N-fluorobisbenzenesulfonamide).
Q: Is this process scalable for commercial production of pharmaceutical intermediates?
A: Yes, the use of inexpensive anthraquinone catalysts and simple workup procedures involving concentration and chromatography makes this method highly suitable for scaling up to meet commercial supply chain demands.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoroalkyl Compounds Supplier
As the demand for fluorinated intermediates continues to grow in the development of next-generation therapeutics, partnering with an experienced CDMO is essential for successful commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from laboratory discovery to full-scale manufacturing. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of difluoroalkyl compounds meets the highest industry standards for safety and efficacy.
We invite you to contact our technical procurement team to discuss your specific requirements for fluorinated intermediates. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how this photocatalytic technology can optimize your budget. We are ready to provide specific COA data and route feasibility assessments to support your supply chain strategy and accelerate your time to market.
