Revolutionizing Planar Chiral Metallocene Production via Palladium-Catalyzed C-H Activation for Commercial Scale-Up
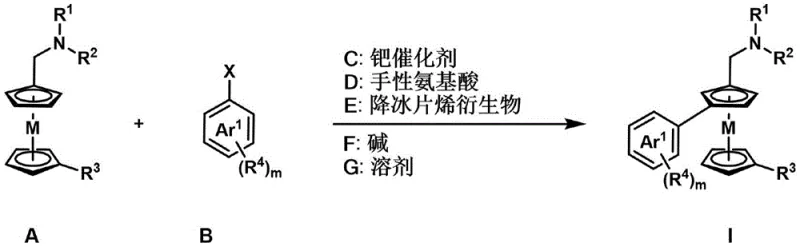
The landscape of asymmetric synthesis is constantly evolving, driven by the relentless demand for high-purity chiral building blocks in the pharmaceutical and agrochemical sectors. A significant breakthrough in this domain is documented in Chinese Patent CN114409714A, which discloses a highly efficient method for synthesizing 1,3-disubstituted planar chiral metallocene compounds. These structures are pivotal as ligands in asymmetric catalysis and as key intermediates in drug discovery, yet their synthesis has historically been plagued by low yields and poor stereocontrol. The patented technology leverages a sophisticated palladium-catalyzed C-H activation strategy, utilizing simple N,N-alkylaminomethyl ferrocene or ruthenium precursors alongside aryl halides. By integrating a chiral amino acid ligand and a norbornene derivative mediator, this process achieves direct functionalization at the meta-position relative to the directing group, a transformation that is notoriously difficult to achieve with high regio- and enantioselectivity. This innovation represents a paradigm shift for manufacturers seeking a reliable pharmaceutical intermediate supplier capable of delivering complex chiral scaffolds with consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,3-disubstituted planar chiral ferrocenes has relied on laborious multi-step sequences that are ill-suited for modern cost reduction in fine chemical manufacturing. Traditional strategies often involve diastereoselective ortho-lithiation using chiral auxiliaries, which requires cryogenic temperatures and strictly anhydrous conditions, posing significant safety and infrastructure burdens on production facilities. Alternatively, chemists have employed chiral resolution of racemates, a fundamentally inefficient approach that theoretically caps the maximum yield at 50% unless dynamic kinetic resolution is successfully implemented, which adds further complexity. Other methods necessitate the installation and subsequent removal of temporary blocking groups to force substitution at the desired position, resulting in excessive waste generation and extended processing times. These legacy techniques suffer from poor atom economy, reliance on hazardous reagents like n-butyllithium, and limited substrate scope, making them economically unviable for the commercial scale-up of complex polymer additives or API intermediates required by global supply chains.
The Novel Approach
In stark contrast, the methodology outlined in patent CN114409714A introduces a streamlined catalytic cycle that bypasses these historical bottlenecks. The process utilizes readily available aryl iodides or bromides and simple aminomethyl metallocenes as starting materials, reacting them under mild thermal conditions ranging from 25°C to 100°C. The core innovation lies in the synergistic combination of a palladium catalyst, a chiral amino acid, and a norbornene derivative, which facilitates a Catellani-type reaction pathway. This allows for the direct installation of the aryl group at the 3-position of the metallocene ring with exceptional precision. The reaction tolerates a broad spectrum of functional groups, including esters, nitriles, halogens, and nitro groups, without the need for protecting group manipulation. This robustness translates directly into reduced lead time for high-purity pharmaceutical intermediates, as the synthetic route is shortened and purification protocols are simplified. The ability to operate in common organic solvents like DMSO or DMAc further enhances the practicality of this method for industrial adoption.
Mechanistic Insights into Pd-Catalyzed C-H Activation and Norbornene Mediation
The success of this synthesis hinges on a delicate interplay between the palladium center and the norbornene mediator, a mechanism often referred to as the Catellani reaction. Initially, the palladium catalyst undergoes oxidative addition with the aryl halide substrate. Subsequently, the norbornene derivative inserts into the palladium-aryl bond, forming a key alkyl-palladium species. This transient species is crucial as it directs the palladium center to activate the specific C-H bond at the 3-position of the metallocene ring, overcoming the inherent electronic and steric biases that typically favor 1,2-substitution. The chiral amino acid ligand coordinates to the metal center during this critical C-H cleavage step, creating a chiral environment that dictates the facial selectivity of the reaction. This ensures that the resulting planar chirality is established with high fidelity. Following C-H activation, reductive elimination releases the final 1,3-disubstituted product and regenerates the active palladium catalyst, allowing the cycle to continue. This mechanistic elegance avoids the formation of stable organolithium intermediates, thereby minimizing side reactions such as protonation or Wurtz-type coupling that often plague traditional methods.
From an impurity control perspective, this catalytic system offers distinct advantages. The use of a chiral amino acid ligand, rather than a stoichiometric chiral auxiliary, means that the source of chirality is catalytic and can be optimized independently of the substrate structure. This reduces the risk of diastereomeric impurities that are difficult to separate. Furthermore, the mild reaction conditions prevent the decomposition of sensitive functional groups on the aryl halide or the metallocene core, which is a common issue in high-temperature or strongly basic traditional protocols. The high tolerance for diverse substituents implies that the impurity profile is cleaner, with fewer byproducts arising from functional group incompatibility. For quality control teams, this predictability is invaluable, as it ensures that the final product meets stringent purity specifications with minimal downstream processing, thereby enhancing the overall reliability of the supply chain for critical chiral materials.
How to Synthesize 1,3-Disubstituted Planar Chiral Metallocenes Efficiently
Implementing this synthesis requires careful attention to the selection of catalysts and additives to maximize yield and enantioselectivity. The patent details a robust protocol where N,N-dimethylaminomethylferrocene reacts with various aryl halides in the presence of palladium acetate and a specific chiral amino acid derivative. The reaction is typically conducted in a mixture of polar aprotic solvents such as DMSO and DMAc, using potassium carbonate as the base. Maintaining an inert atmosphere, preferably argon, is recommended to prevent catalyst oxidation, although the system shows some tolerance to air. The detailed standardized synthesis steps see the guide below.
- Combine N,N-alkylaminomethyl ferrocene/ruthenium and aryl halide with a palladium catalyst, chiral amino acid, norbornene derivative, and base in an organic solvent.
- Stir the reaction mixture at temperatures between 25°C and 100°C under inert gas protection for 1 to 48 hours.
- Quench the reaction, extract with organic solvents, and purify the crude product via column chromatography to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers transformative benefits that extend beyond mere technical feasibility. The shift from stoichiometric organometallic reagents to a catalytic system fundamentally alters the cost structure of production. By eliminating the need for expensive, hazardous, and temperature-sensitive reagents like n-butyllithium, the process significantly reduces raw material costs and associated safety compliance expenditures. The use of commercially available aryl halides and simple ferrocene derivatives ensures a stable and diversified supply base, mitigating the risk of shortages that often affect specialized chiral reagents. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by multinational pharmaceutical clients.
- Cost Reduction in Manufacturing: The transition to a catalytic process inherently lowers the cost of goods sold by reducing reagent consumption. Unlike traditional methods that require stoichiometric amounts of chiral auxiliaries or blocking groups, this method uses catalytic quantities of palladium and chiral ligands. This drastic reduction in material usage, combined with the elimination of cryogenic cooling requirements, leads to substantial energy savings and lower utility costs. Furthermore, the high yields and excellent enantioselectivity minimize the loss of valuable starting materials and reduce the burden on purification resources, resulting in a more economically efficient manufacturing process that enhances overall profit margins without compromising quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as aryl bromides, iodides, and basic ferrocene derivatives ensures that the supply chain is resilient against market volatility. These starting materials are produced by multiple vendors globally, preventing single-source dependency. The robustness of the reaction conditions, which tolerate a wide range of functional groups and do not require ultra-dry or ultra-cold environments, simplifies logistics and storage requirements. This flexibility allows for faster turnaround times and more reliable delivery schedules, ensuring that downstream customers receive their critical intermediates without delay, thereby strengthening the partnership between the manufacturer and the end-user.
- Scalability and Environmental Compliance: The protocol has been validated at the gram scale with consistent results, demonstrating its readiness for kilogram and tonne-scale production. The simplified workup procedure, involving standard extraction and column chromatography, is easily adaptable to large-scale industrial equipment. Additionally, the avoidance of toxic organolithium reagents and the reduction of waste through higher atom economy align with green chemistry principles. This not only reduces the environmental footprint of the manufacturing process but also simplifies regulatory compliance regarding hazardous waste disposal, making it a sustainable choice for long-term production of specialty chemicals and advanced materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process capabilities and limitations for potential partners and technical stakeholders.
Q: What is the primary advantage of this synthesis method over traditional lithiation strategies?
A: Unlike traditional methods requiring cryogenic conditions and stoichiometric organometallic reagents, this palladium-catalyzed approach operates under mild thermal conditions (25°C-100°C) using commercially available starting materials, significantly simplifying the operational complexity and safety profile.
Q: What level of enantioselectivity can be achieved with this protocol?
A: The method demonstrates exceptional stereocontrol, consistently achieving enantiomeric excess (ee) values greater than 99% across a wide range of substrates, eliminating the need for downstream chiral resolution steps.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly demonstrates gram-scale synthesis with maintained high yield and purity, indicating robust scalability for commercial production of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Disubstituted Planar Chiral Metallocenes Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics and advanced materials. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement complex catalytic systems, such as the palladium-norbornene cooperative catalysis described in CN114409714A, positions us as a strategic partner for companies seeking to secure their supply of difficult-to-make chiral building blocks.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can accelerate your development timeline and enhance your competitive edge in the global market.
