Revolutionizing N-Heterocycle Synthesis: A Metal-Free Alkylation Strategy for Commercial Scale-Up
Revolutionizing N-Heterocycle Synthesis: A Metal-Free Alkylation Strategy for Commercial Scale-Up
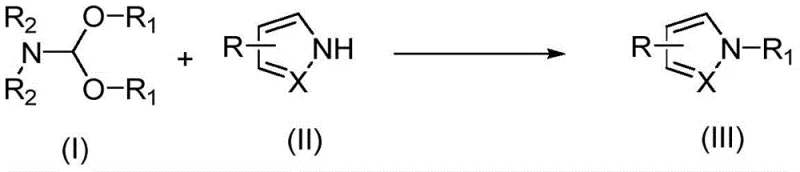
The landscape of organic synthesis is constantly evolving, driven by the need for greener, more efficient, and cost-effective methodologies. A significant breakthrough in this domain is detailed in patent CN108503583B, which discloses a novel alkylation method for nitrogen-containing hydrogen compounds. This technology represents a paradigm shift from traditional, harsh alkylation protocols to a metal-free, mild, and highly efficient process. By utilizing N,N-dimethylformamide dialkyl acetals as alkylating agents, this method enables the direct N-alkylation of N-H heterocyclic compounds without the necessity for transition metal catalysts or strong inorganic bases. For R&D directors and procurement managers in the pharmaceutical and agrochemical sectors, this innovation offers a compelling value proposition: it drastically simplifies the synthetic route, reduces environmental burden, and ensures high purity of the final intermediates, which is critical for downstream API manufacturing.
The core of this invention lies in the unique reactivity of N,N-dimethylformamide dialkyl acetals. These compounds serve a dual purpose, acting not only as the source of the alkyl group but also providing a weakly alkaline environment that facilitates the reaction. This eliminates the generation of stoichiometric amounts of inorganic salt by-products, a common pain point in traditional nucleophilic substitution reactions. The process operates under mild thermal conditions, typically between 50°C and 120°C, with optimal results often observed around 90°C. Such温和 conditions preserve sensitive functional groups on the heterocyclic scaffold, thereby expanding the substrate scope significantly. This technological advancement positions suppliers who adopt this methodology as reliable pharmaceutical intermediate suppliers capable of delivering complex molecules with superior quality profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alkylation of nitrogen atoms in heterocyclic compounds has relied heavily on the use of alkyl halides, sulfates, or sulfonates. These traditional reagents, while effective, come with substantial drawbacks that impact both the economic and environmental viability of the process. The use of alkyl halides typically necessitates the presence of strong bases such as sodium carbonate, n-butyllithium, or sodium tert-butoxide to generate the reactive nitrogen anion intermediate. These conditions are often harsh, requiring low temperatures and strictly anhydrous environments, which increases operational complexity and energy consumption. Furthermore, the reaction generates stoichiometric amounts of inorganic salts as by-products, creating a significant waste disposal challenge and complicating the purification process. The presence of these salts can trap the product, leading to lower yields and requiring extensive washing steps that consume large volumes of water and organic solvents.
Additionally, alternative alkylating agents like dimethyl sulfate or diethyl sulfate, although reactive under milder conditions, pose severe safety and health risks due to their high toxicity and carcinogenicity. Handling these reagents requires specialized equipment and rigorous safety protocols, which inevitably drives up the cost of production. For procurement managers, the reliance on such hazardous materials introduces supply chain risks and regulatory hurdles. The cumulative effect of these limitations is a process that is difficult to scale, expensive to operate, and environmentally unsustainable. In an industry increasingly focused on green chemistry principles, these conventional methods are becoming obsolete, creating a pressing demand for innovative alternatives that can deliver cost reduction in API manufacturing without compromising on safety or quality.
The Novel Approach
The methodology described in patent CN108503583B offers a transformative solution to these entrenched problems. By employing N,N-dimethylformamide dialkyl acetals as the alkylating agent, the reaction proceeds smoothly without the need for any external metal catalysts or strong inorganic bases. The acetal itself possesses weak basicity, which is sufficient to promote the alkylation of the N-H bond while avoiding the aggressive conditions associated with traditional bases. This metal-free nature is a critical advantage, particularly for pharmaceutical applications where residual heavy metals must be kept below strict ppm limits. Eliminating the metal catalyst removes the need for expensive and time-consuming metal scavenging steps, thereby streamlining the downstream processing. The reaction conditions are remarkably mild, typically conducted in polar solvents like DMF at temperatures around 90°C, which are easily achievable in standard industrial reactors.
Moreover, the workup procedure is exceptionally simple. Since no inorganic salts are generated, the product can often be isolated through straightforward extraction or even crystallization, significantly reducing solvent usage and waste generation. The patent data indicates that this method maintains high reaction efficiency even upon scale-up, making it highly suitable for large-scale industrial production. The broad substrate tolerance means that a single optimized protocol can be applied to a diverse range of heterocycles, from simple imidazoles to complex fused ring systems like benzimidazoles and quinazolinones. This versatility allows manufacturers to consolidate their synthetic platforms, reducing the need for multiple specialized processes. For supply chain heads, this translates to enhanced reliability and reduced lead times for high-purity intermediates, as the process is robust, reproducible, and less prone to failure due to sensitive reaction parameters.
Mechanistic Insights into Metal-Free N-Alkylation
Understanding the mechanistic underpinnings of this reaction is essential for R&D teams looking to implement this technology effectively. The reaction initiates with the interaction between the N-H heterocyclic compound and the N,N-dimethylformamide dialkyl acetal. Unlike traditional SN2 reactions where a discrete anion attacks an electrophile, this process likely involves a concerted or semi-concerted mechanism facilitated by the weak basicity of the acetal oxygen or the dimethylamino group. The acetal acts as an electrophilic alkylating agent, where the alkyl group attached to the oxygen is transferred to the nitrogen atom of the heterocycle. The leaving group in this transformation is a hemiaminal ether or an amide derivative, which subsequently decomposes into DMF and the corresponding alcohol. This decomposition is thermodynamically favorable and drives the reaction forward. The absence of strong bases prevents side reactions such as elimination or over-alkylation, which are common pitfalls in traditional methods. This selectivity is crucial for maintaining the integrity of complex molecular architectures often found in drug candidates.
From an impurity control perspective, the metal-free nature of this catalysis is paramount. In conventional transition-metal catalyzed couplings, trace metals can coordinate with product molecules or catalyze degradation pathways during storage. By completely removing metals from the equation, the impurity profile of the resulting intermediate is significantly cleaner. The primary by-products are DMF and alcohols, both of which are volatile and easily removed during the concentration or drying stages. This simplicity in the impurity profile facilitates easier purification, often allowing for high-purity products (>99%) to be obtained with minimal effort. For quality control laboratories, this means faster release times and lower analytical burdens. The robustness of the mechanism also implies that minor variations in reagent quality or reaction temperature do not drastically affect the outcome, providing a wide operating window that is ideal for manufacturing environments where perfect control is sometimes challenging to maintain.
How to Synthesize N-Alkylated Heterocycles Efficiently
Implementing this synthesis route requires attention to specific operational parameters to maximize yield and purity. The general procedure involves dissolving the N-H heterocyclic substrate in a suitable polar organic solvent, with DMF being the preferred choice due to its ability to dissolve both reactants and stabilize the transition state. The N,N-dimethylformamide dialkyl acetal is then added, typically in a molar excess (around 4 equivalents) to drive the reaction to completion. The mixture is heated, with optimization studies suggesting that 90°C provides the best balance between reaction rate and yield. Reaction times are generally short, often completing within 4 hours, which enhances throughput. Following the reaction, the workup involves a simple aqueous extraction to remove the polar by-products, followed by solvent removal and purification if necessary. The detailed standardized synthesis steps for specific derivatives are outlined in the guide below.
- Dissolve the N-H containing heterocyclic compound in a polar organic solvent such as DMF.
- Add N,N-dimethylformamide dialkyl acetal dropwise to the solution at a molar ratio of approximately 4: 1.
- Heat the reaction mixture to 90°C for 4 hours, then perform aqueous workup and extraction to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free alkylation technology offers tangible strategic benefits that go beyond mere technical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Traditional methods often require the sourcing of hazardous alkylating agents like dimethyl sulfate or expensive organometallic bases, which can be subject to strict regulatory controls and supply volatility. In contrast, N,N-dimethylformamide dialkyl acetals are derived from DMF, a commodity chemical with a stable and abundant global supply. This shift reduces the risk of supply disruptions and stabilizes raw material costs. Furthermore, the elimination of heavy metal catalysts removes a significant cost center associated with catalyst recovery and validation. The simplified workup procedure also means less consumption of solvents and utilities, directly contributing to cost reduction in API manufacturing. These factors combine to create a more resilient and cost-efficient supply chain.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the elimination of expensive and hazardous reagents. By removing the need for strong inorganic bases and transition metal catalysts, the process avoids the costs associated with purchasing these high-value materials and the subsequent waste treatment. Traditional methods generate large quantities of inorganic salt waste, the disposal of which is becoming increasingly expensive due to tightening environmental regulations. This new method produces organic by-products that are easier to handle and potentially recyclable. Additionally, the high yields reported in the patent (often exceeding 90%) mean that less raw material is wasted, improving the overall atom economy. The simplified purification process reduces the demand for chromatography media and excessive solvent volumes, further lowering the operational expenditure. These cumulative savings allow for a more competitive pricing structure for the final intermediates.
- Enhanced Supply Chain Reliability: Reliability is the cornerstone of a robust supply chain, and this technology enhances it by reducing process complexity. The mild reaction conditions and broad substrate tolerance mean that the process is less sensitive to minor fluctuations in temperature or reagent quality, reducing the likelihood of batch failures. The use of stable, non-hazardous reagents simplifies logistics and storage requirements, removing the need for specialized containment facilities required for toxic sulfates or pyrophoric bases. This ease of handling accelerates the turnover time from raw material receipt to finished goods. For supply chain heads, this translates to shorter lead times and the ability to respond more agilely to market demands. The scalability of the process ensures that supply can be ramped up quickly to meet surges in demand without the need for extensive process re-engineering or new equipment investments.
- Scalability and Environmental Compliance: As the pharmaceutical industry moves towards greener manufacturing practices, environmental compliance is no longer optional but a license to operate. This metal-free alkylation method aligns perfectly with green chemistry principles by minimizing waste and avoiding toxic substances. The absence of heavy metals simplifies the regulatory filing process for new drug applications, as there is no need to demonstrate the removal of trace metals to extremely low levels. The process generates less hazardous waste, reducing the environmental footprint of the manufacturing site. Scalability is inherently built into the design; the patent explicitly notes that reaction efficiency remains high upon amplification. This means that moving from kilogram to ton-scale production does not require fundamental changes to the chemistry, ensuring a smooth transition from pilot plant to commercial manufacturing. This scalability ensures long-term supply continuity for key intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this alkylation technology. These answers are derived directly from the experimental data and specifications provided in patent CN108503583B. They are intended to clarify the scope, safety, and operational feasibility of the method for potential partners and stakeholders. Understanding these details is crucial for evaluating the fit of this technology within your existing manufacturing portfolio.
Q: What are the advantages of using DMF acetals over traditional alkyl halides?
A: Unlike traditional alkyl halides which require strong bases and generate inorganic salt waste, DMF acetals act as both the alkyl source and a weak base, eliminating the need for metal catalysts and simplifying purification.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the reaction efficiency remains high after amplification, making it suitable for large-scale industrial production due to mild conditions and simple workup.
Q: What types of substrates are compatible with this alkylation method?
A: The method exhibits high functional group tolerance and is applicable to a wide range of N-H heterocycles including imidazoles, benzimidazoles, pyrazoles, indoles, triazoles, and various lactams.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Heterocyclic Intermediate Supplier
The technological potential of metal-free N-alkylation is immense, offering a pathway to cleaner, cheaper, and faster production of vital pharmaceutical building blocks. At NINGBO INNO PHARMCHEM, we pride ourselves on being at the forefront of adopting such innovative synthetic methodologies. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art rigorous QC labs that ensure every batch meets stringent purity specifications, regardless of the complexity of the molecule. We understand that in the fast-paced world of drug development, consistency and quality are non-negotiable. Our team of expert chemists is ready to adapt this patented technology to your specific target molecules, ensuring a seamless transfer from lab bench to commercial reactor.
We invite you to explore how this advanced alkylation strategy can optimize your supply chain and reduce your overall manufacturing costs. Whether you are looking for a reliable source of complex intermediates or need assistance in process development, our technical procurement team is here to support you. Please contact us today to request a Customized Cost-Saving Analysis tailored to your project needs. We are eager to provide you with specific COA data and route feasibility assessments that demonstrate the tangible benefits of partnering with us. Let us help you accelerate your journey from discovery to market with our superior chemical solutions.
