Advancing Indole Synthesis: A Novel Nickel-Catalyzed Carbonylative Cyclization for Commercial Scale-Up
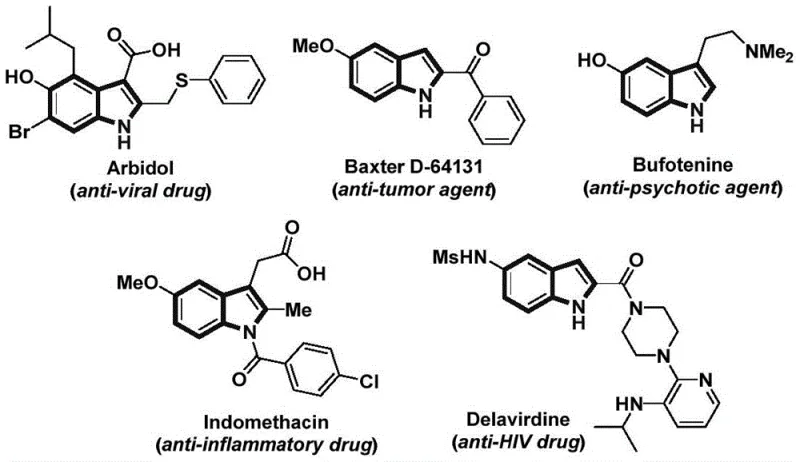
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing privileged scaffolds, among which the indole nucleus stands paramount due to its prevalence in bioactive molecules ranging from antiviral agents like Arbidol to anti-inflammatory drugs like Indomethacin. A significant technological breakthrough in this domain is documented in Chinese Patent CN115286553A, which discloses a highly efficient preparation method for indole compounds via a nickel-catalyzed carbonylative cyclization strategy. This innovation addresses critical bottlenecks in traditional synthetic routes by utilizing readily available starting materials such as 2-alkynylnitrobenzenes and arylboronic acid pinacol esters. The process operates under relatively mild thermal conditions compared to extreme high-pressure alternatives, demonstrating exceptional functional group tolerance and operational simplicity. For R&D directors and procurement specialists, this patent represents a viable pathway to accessing high-purity pharmaceutical intermediates with improved economic feasibility and reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indole core, particularly when involving carbonyl functionalities, has relied heavily on palladium-catalyzed carbonylation reactions or multi-step sequences involving harsh reagents. Traditional carbonylation often necessitates the use of gaseous carbon monoxide, a toxic and hazardous gas that requires specialized high-pressure equipment and rigorous safety protocols, thereby inflating capital expenditure and operational risks. Furthermore, palladium catalysts, while effective, are precious metals subject to volatile market pricing and supply chain constraints, making them less ideal for large-scale commercial manufacturing where cost margins are tight. Many existing methods also suffer from limited substrate scope, failing to accommodate sensitive functional groups or requiring excessive protection-deprotection steps that lower overall atom economy and increase waste generation.
The Novel Approach
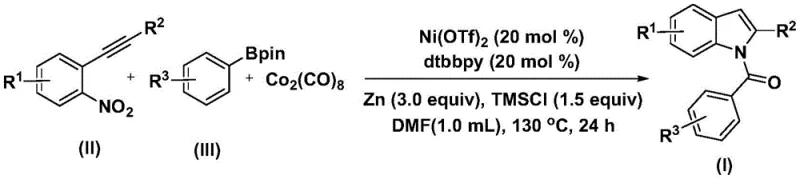
The methodology outlined in CN115286553A offers a transformative alternative by employing a nickel-catalyzed system that utilizes dicobalt octacarbonyl as a solid carbon monoxide surrogate. This strategic shift eliminates the need for handling gaseous CO, significantly enhancing process safety and simplifying reactor requirements. By replacing expensive palladium with earth-abundant nickel, the process inherently drives down raw material costs while maintaining high catalytic efficiency. The reaction proceeds through a tandem sequence involving transmetallation, CO insertion, nitro reduction, and cyclization in a single pot, streamlining the synthesis from multiple steps to a direct one-pot transformation. This consolidation not only reduces solvent consumption and processing time but also minimizes the formation of intermediate impurities, resulting in a cleaner crude profile that facilitates downstream purification.
Mechanistic Insights into Nickel-Catalyzed Carbonylative Cyclization
The mechanistic pathway of this transformation is a sophisticated orchestration of organometallic steps initiated by the activation of the arylboronic acid pinacol ester. It is hypothesized that the nickel catalyst first undergoes transmetallation with the arylboron species to generate an aryl-nickel intermediate. Subsequently, carbon monoxide, released in situ from the thermal decomposition of dicobalt octacarbonyl, inserts into the nickel-carbon bond to form a reactive acyl-nickel species. This acyl intermediate is pivotal as it serves as the electrophilic partner for the subsequent intramolecular cyclization. Concurrently, the nitro group on the 2-alkynylnitrobenzene substrate undergoes reduction, likely facilitated by the zinc reductant and the nickel species, converting it into a nucleophilic amine or hydroxylamine equivalent. This reduced nitrogen species then attacks the acyl-nickel center, followed by reductive elimination to yield an amide intermediate, which finally undergoes cyclization to furnish the target indole scaffold. Understanding this cycle is crucial for optimizing reaction parameters and troubleshooting potential side reactions during scale-up.
From an impurity control perspective, the use of trimethylsilyl chloride (TMSCl) as an additive plays a vital role in scavenging oxygenated byproducts and stabilizing reactive intermediates, thereby suppressing the formation of homocoupling products or incomplete reduction species. The choice of DMF as the solvent is also mechanistically significant, as its high polarity and coordinating ability help stabilize the cationic nickel species and facilitate the solubility of inorganic salts like zinc chloride generated during the reaction. The robustness of this catalytic cycle allows for the tolerance of electron-withdrawing and electron-donating groups alike, ensuring that the electronic nature of the substrates does not drastically alter the reaction kinetics or lead to catalyst deactivation. This mechanistic resilience translates directly to consistent batch-to-batch reproducibility, a key metric for quality assurance in pharmaceutical manufacturing.
How to Synthesize Indole Compounds Efficiently
Implementing this synthesis route requires precise control over stoichiometry and thermal conditions to maximize yield and purity. The protocol involves charging a reaction vessel with nickel trifluoromethanesulfonate, a bipyridine ligand, zinc powder, trimethylsilyl chloride, dicobalt octacarbonyl, the 2-alkynylnitrobenzene substrate, and the arylboronic acid pinacol ester in DMF. The detailed standardized synthesis steps are provided in the guide below.
- Combine nickel catalyst, nitrogen ligand, zinc reductant, additive, carbon monoxide substitute, 2-alkynyl nitrobenzene, and arylboronic acid pinacol ester in an organic solvent.
- Heat the reaction mixture to 130°C and maintain stirring for 24 hours to ensure complete conversion.
- Perform post-treatment including filtration and silica gel mixing, followed by column chromatography purification to isolate the target indole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed protocol offers substantial strategic benefits beyond mere technical feasibility. The shift from precious metal catalysis to base metal catalysis fundamentally alters the cost structure of the synthesis, removing exposure to the volatile pricing of palladium and rhodium. Furthermore, the use of a solid CO source mitigates the regulatory and logistical burdens associated with transporting and storing compressed toxic gases, simplifying site compliance and reducing insurance premiums. The one-pot nature of the reaction reduces the number of unit operations, leading to lower energy consumption and reduced labor hours per kilogram of product. These factors collectively contribute to a more resilient and cost-effective supply chain capable of meeting the demands of global pharmaceutical markets.
- Cost Reduction in Manufacturing: The replacement of palladium catalysts with nickel trifluoromethanesulfonate represents a direct material cost saving, as nickel is orders of magnitude cheaper than precious metals. Additionally, the elimination of gaseous carbon monoxide infrastructure removes the need for specialized high-pressure reactors and gas monitoring systems, resulting in significant capital expenditure avoidance. The high atom economy of the one-pot process minimizes solvent usage and waste disposal costs, further enhancing the overall economic viability of the manufacturing process for high-volume API intermediates.
- Enhanced Supply Chain Reliability: Sourcing nickel salts and dicobalt octacarbonyl is generally more stable and less prone to geopolitical supply disruptions compared to platinum group metals. The starting materials, 2-alkynylnitrobenzenes and arylboronic esters, are commodity chemicals available from multiple global suppliers, ensuring redundancy in the supply base. This diversification reduces the risk of production stoppages due to raw material shortages, allowing for consistent delivery schedules and reliable inventory management for downstream drug manufacturers.
- Scalability and Environmental Compliance: The reaction conditions, operating at atmospheric pressure with a solid CO source, are inherently safer and easier to scale from laboratory to pilot and commercial plants without complex engineering modifications. The process generates less hazardous waste compared to traditional methods involving toxic gases, aligning with increasingly stringent environmental regulations and sustainability goals. The simplified workup procedure, involving filtration and chromatography, reduces the volume of aqueous waste streams, facilitating easier treatment and disposal while lowering the environmental footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity for potential partners and licensees.
Q: What are the key advantages of this nickel-catalyzed method over traditional palladium catalysis?
A: This method utilizes nickel, a base metal, which is significantly more abundant and cost-effective than precious palladium catalysts. Additionally, it employs a solid carbon monoxide substitute (cobalt carbonyl), eliminating the safety hazards and logistical complexities associated with handling toxic gaseous CO.
Q: Does this synthesis method tolerate diverse functional groups?
A: Yes, the patent data demonstrates excellent substrate compatibility. The reaction successfully accommodates various substituents such as halogens (F, Cl, Br), alkyl groups (methyl, tert-butyl), and alkoxy groups (methoxy) on both the nitrobenzene and boronic ester components without compromising yield.
Q: What are the typical reaction conditions required for this transformation?
A: The optimal conditions involve using DMF as the solvent at a temperature of 130°C for a duration of 24 hours. The system requires a nickel catalyst (Ni(OTf)2), a bipyridine ligand, zinc powder as a reductant, and trimethylsilyl chloride as an additive.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed technology for producing high-value indole intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless. Our state-of-the-art facilities are equipped to handle air-sensitive and moisture-sensitive chemistries safely, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this method can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for our indole portfolio and discuss route feasibility assessments for your custom synthesis projects.
