Scalable Non-Metal Catalytic Hydrogenation for High-Purity 3-Substituted Chromanones
Scalable Non-Metal Catalytic Hydrogenation for High-Purity 3-Substituted Chromanones
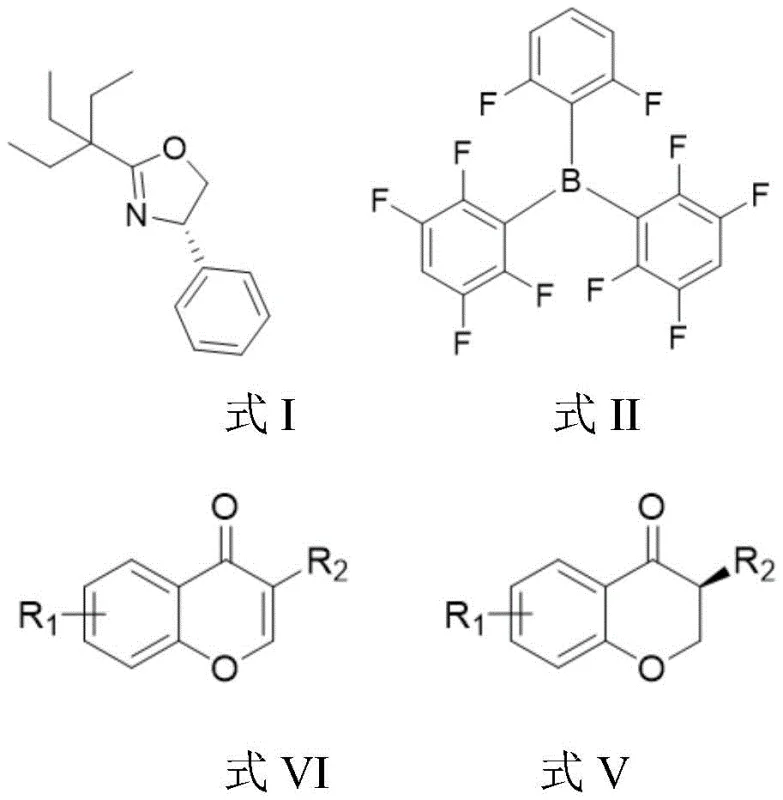
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high stereocontrol with environmental sustainability and cost efficiency. A significant breakthrough in this domain is documented in Chinese Patent CN112898259A, which discloses a novel method for preparing 3-position substituted chromanone compounds through non-metallic catalytic hydrogenation. This technology represents a paradigm shift from traditional transition metal-catalyzed processes, utilizing a unique frustrated Lewis pair (FLP) system composed of a chiral oxazoline and an achiral borane. By leveraging molecular hydrogen as the sole hydrogen source, this method achieves exceptional enantioselectivity with ee values reaching up to 95% and yields as high as 98%. For R&D directors and procurement specialists, this patent offers a compelling alternative for synthesizing key pharmaceutical intermediates, particularly those requiring strict control over metal impurities and chiral integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 3-substituted chromanones, which serve as critical scaffolds for bioactive molecules like dihydrodaidzein, has relied heavily on chiral metal catalysts. While effective, these conventional pathways introduce significant downstream challenges, primarily concerning metal residue contamination. The presence of toxic heavy metals such as palladium, rhodium, or ruthenium necessitates rigorous and costly purification protocols to meet stringent regulatory standards for active pharmaceutical ingredients (APIs). Furthermore, the sensitivity of many metal catalysts to air and moisture often requires inert atmosphere handling and specialized equipment, complicating the operational workflow. These factors collectively inflate the cost of goods sold (COGS) and extend lead times, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology outlined in the patent circumvents these issues by employing a metal-free catalytic system. The core innovation lies in the synergistic interaction between a chiral oxazoline Lewis base (Formula I) and an achiral borane Lewis acid (Formula II). This combination creates a sterically hindered environment capable of activating molecular hydrogen without the need for transition metals. The reaction proceeds under remarkably mild conditions, typically at 30°C to 40°C and a hydrogen pressure of 40 bar, using mesitylene as the solvent. This approach not only eliminates the risk of metal contamination but also simplifies the reaction setup, allowing for easier handling and potentially lower capital expenditure on reactor infrastructure. The versatility of this system is demonstrated across a broad substrate scope, accommodating various substituents on the benzene ring and at the 3-position.
Mechanistic Insights into Non-Metal Catalytic Hydrogenation
The efficacy of this synthesis relies on the precise engineering of the catalytic species. The chiral oxazoline, depicted in Formula I, acts as the stereochemical director, imparting chirality to the final product through its rigid structural framework. When paired with the bulky, electron-deficient borane species shown in Formula II, they form a Frustrated Lewis Pair (FLP). In this state, the steric bulk prevents the formation of a classical Lewis adduct, leaving both the Lewis acid and base sites available to cooperatively activate the hydrogen molecule. This heterolytic cleavage of H2 generates a proton and a hydride equivalent that are transferred to the substrate in a highly organized transition state. The result is the asymmetric reduction of the carbon-carbon double bond in the chromone ring, yielding the saturated chromanone with high optical purity.
Impurity control is inherently superior in this metal-free regime. Without transition metals, there is no risk of generating metal-associated byproducts or requiring scavenger resins for purification. The primary impurities are likely to be unreacted starting materials or minor diastereomers, which are readily separated via standard silica gel column chromatography using petroleum ether and dichloromethane mixtures. The robustness of the catalyst system allows for consistent performance across different batches, ensuring a stable impurity profile. This predictability is crucial for regulatory filings, where defining and controlling the impurity landscape is a mandatory requirement for API approval. The ability to achieve up to 98% yield further minimizes waste generation, aligning with green chemistry principles.
How to Synthesize 3-Substituted Chromanone Efficiently
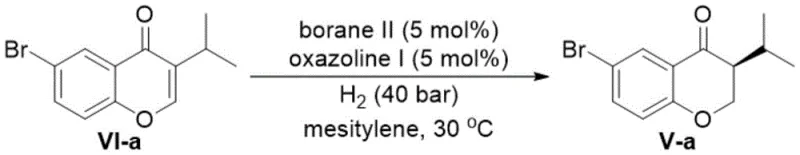
Implementing this synthesis requires careful attention to the preparation of the catalytic components and the reaction environment. The process begins with the independent synthesis of the chiral oxazoline and the fluorinated borane, followed by their combination with the chromone substrate in an autoclave. The reaction is conducted under a positive pressure of hydrogen, ensuring a constant supply of the reducing agent throughout the 24 to 72-hour reaction window. Post-reaction workup involves simple concentration and purification, avoiding complex aqueous extractions often needed to remove metal salts. The following visual guide illustrates a representative example of this transformation, highlighting the specific conditions and substrate compatibility.
- Prepare the catalytic system by mixing chiral oxazoline (Formula I) and achiral borane (Formula II) in mesitylene solvent.
- Add the 3-substituted chromone substrate (Formula VI) to the reaction mixture under nitrogen protection.
- Transfer to an autoclave, pressurize with hydrogen to 40 bar, and stir at 30°C for 24-72 hours to obtain the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this non-metal catalytic technology presents a strategic opportunity to optimize costs and mitigate supply risks. The elimination of precious metal catalysts removes a volatile cost component from the bill of materials, as prices for metals like palladium and rhodium can fluctuate wildly based on geopolitical factors. Moreover, the simplified purification process reduces the consumption of auxiliary materials such as metal scavengers and specialized filtration media. This streamlining of the manufacturing process translates directly into a more predictable and potentially lower cost structure for the final intermediate. The reliance on commercially available and stable organic reagents further enhances supply security, reducing dependence on single-source metal catalyst suppliers.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts significantly lowers the raw material costs associated with the hydrogenation step. Additionally, the absence of metal residues eliminates the need for costly and time-consuming metal removal processes, such as treatment with activated carbon or silica-supported scavengers, thereby reducing overall processing time and consumable usage. The high yields reported, often exceeding 95%, minimize material loss and maximize the throughput of the manufacturing facility, contributing to substantial economic efficiency.
- Enhanced Supply Chain Reliability: By utilizing organic catalysts that can be synthesized from readily available precursors, the supply chain becomes more resilient to disruptions common in the mining and refining sectors of precious metals. The mild reaction conditions (30°C) reduce the energy demand compared to high-temperature metal-catalyzed processes, lowering utility costs and minimizing the thermal stress on equipment. This operational simplicity facilitates easier technology transfer between manufacturing sites, ensuring consistent quality and availability of the intermediate regardless of the production location.
- Scalability and Environmental Compliance: The metal-free nature of this process simplifies waste management and disposal, as there are no heavy metal contaminants to treat in the effluent streams. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The process is inherently scalable, as demonstrated by the use of standard autoclaves and common organic solvents like mesitylene. The robustness of the catalyst system supports long campaign runs, enabling the commercial scale-up of complex pharmaceutical intermediates without the need for specialized high-pressure or high-temperature reactors typically required for less active metal catalysts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this non-metal catalytic hydrogenation technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering a clear understanding of the process capabilities and limitations for potential adopters.
Q: How does this non-metal method compare to traditional metal catalysis regarding purity?
A: Unlike traditional methods using transition metals which risk heavy metal contamination, this patented non-metal approach eliminates metal residues entirely, ensuring higher purity suitable for sensitive pharmaceutical applications without complex purification steps.
Q: What are the typical enantiomeric excess (ee) values achieved with this process?
A: The process demonstrates excellent stereoselectivity, achieving enantiomeric excess (ee) values as high as 95% for various substrates, such as 6-bromo-3-isopropyl chromanone, ensuring high optical purity.
Q: Is this catalytic system scalable for industrial production?
A: Yes, the reaction operates under mild conditions (30°C, 40 bar H2) using stable organic catalysts, making it highly amenable to commercial scale-up without the safety hazards associated with high-temperature metal catalysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Substituted Chromanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free catalytic technologies in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112898259A can be successfully translated into industrial reality. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise measurement of enantiomeric excess and the confirmation of zero metal residues, guaranteeing that every batch meets the highest global standards.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free approach can optimize your budget. Please contact our technical procurement team today to request specific COA data for our chiral chromanone portfolio and to discuss route feasibility assessments for your target molecules. Let us help you secure a sustainable and cost-effective supply chain for your critical pharmaceutical intermediates.
