Scalable Synthesis of Chiral Diketone Intermediates via Novel Organocatalysis for Pharmaceutical Applications
Scalable Synthesis of Chiral Diketone Intermediates via Novel Organocatalysis for Pharmaceutical Applications
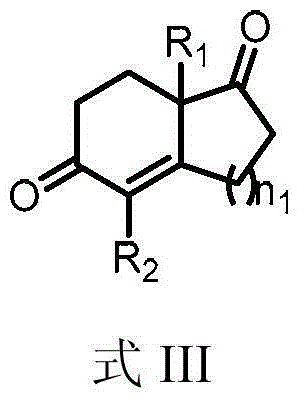
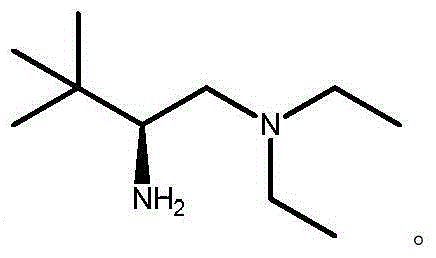
The landscape of asymmetric synthesis for pharmaceutical intermediates is continuously evolving, driven by the demand for higher purity and more sustainable manufacturing processes. A significant breakthrough in this domain is detailed in Chinese patent CN103553931A, which discloses a highly efficient method for synthesizing chiral diketone compounds, specifically targeting the renowned Hajos-Parrish and Wieland-Miescher ketones. These bicyclic enones serve as critical building blocks in the total synthesis of complex natural products, including terpenoids and steroids such as paclitaxel and various hormonal agents. The core innovation lies in the deployment of a novel chiral amine catalyst, represented by Formula I, which operates in conjunction with a dual-acid system to facilitate a robust Robinson annulation. This technology addresses long-standing inefficiencies in traditional organocatalysis, offering a pathway that is not only chemically superior but also commercially viable for a reliable chiral diketone intermediates supplier seeking to optimize their portfolio.
The structural simplicity of the catalyst described in the patent belies its profound impact on reaction efficiency. Unlike earlier generations of organocatalysts that suffered from complex synthetic routes and high loading requirements, this new class of compounds is derived from readily available amino acid precursors, ensuring a stable supply chain. The method involves mixing the catalyst with a strong acid in an organic solvent, removing the solvent, and then engaging the substrate mixture in a reaction that can proceed under solvent-free conditions. This approach drastically reduces the environmental footprint and operational complexity associated with large-scale chemical manufacturing. For R&D teams focused on process intensification, the ability to achieve high enantioselectivity without the burden of excessive solvent waste represents a paradigm shift in how these valuable intermediates are produced.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral bicyclic enones like the Wieland-Miescher ketone has relied heavily on L-proline catalysis, a method established in the early 1970s. While effective for certain substrates like the Hajos-Parrish ketone, the application of L-proline to six-membered ring systems often results in mediocre outcomes, typically yielding enantiomeric excess (ee) values in the range of 70-75%. To achieve the optical purity required for pharmaceutical applications, manufacturers are forced to employ multiple rounds of recrystallization, a process that is not only labor-intensive and time-consuming but also leads to significant material loss. Furthermore, traditional protocols frequently necessitate the use of high-boiling polar aprotic solvents such as dimethylformamide (DMF) or dimethyl sulfoxide (DMSO). These solvents are notoriously difficult to remove completely, posing challenges for downstream processing and raising concerns regarding residual solvent limits in final drug substances. The combination of low catalytic activity, prolonged reaction times requiring heating, and cumbersome purification steps renders these conventional methods economically inefficient and environmentally unsustainable for modern green chemistry standards.
The Novel Approach
The methodology presented in patent CN103553931A offers a compelling solution to these entrenched problems by introducing a structurally simple yet highly effective chiral amine catalyst. This new approach enables a one-step synthesis that bypasses the need for multi-step sequences and extensive purification operations. A key differentiator is the catalyst's ability to function at significantly lower loadings, specifically as low as 2 mol%, compared to the often higher quantities required by previous organocatalysts. The reaction system utilizes a synergistic combination of a strong acid, such as trifluoromethanesulfonic acid, and a weak acid, like m-nitrobenzoic acid, to activate the catalyst and substrates effectively. Remarkably, the process can be conducted under solvent-free conditions after the initial activation step, which aligns perfectly with the principles of green chemistry by minimizing volatile organic compound (VOC) emissions. This streamlined protocol not only accelerates the synthesis cycle but also ensures that high yields and exceptional enantioselectivity are maintained even upon scale-up, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Organocatalytic Robinson Annulation
The success of this synthetic route hinges on the precise mechanistic interplay between the chiral amine catalyst and the dual-acid co-catalyst system. The catalyst, typically a chiral primary amine derived from L-tert-leucine, forms an enamine intermediate with the 1,3-diketone substrate. This enamine species is nucleophilic and attacks the alpha,beta-unsaturated ketone (Michael acceptor) in a stereocontrolled manner. The presence of the strong acid serves to protonate the catalyst or intermediate species, enhancing electrophilicity and stabilizing transition states, while the weak acid acts as a buffer to fine-tune the acidity of the medium, preventing catalyst decomposition or side reactions. This delicate balance is crucial for achieving the high levels of stereoinduction observed, where ee values can reach up to 92% directly from the reaction mixture. The steric bulk of the tert-butyl group on the catalyst backbone plays a pivotal role in shielding one face of the reactive intermediate, thereby directing the incoming nucleophile to the desired trajectory and ensuring the formation of the correct enantiomer.
From an impurity control perspective, the high stereoselectivity of this system inherently limits the formation of unwanted diastereomers and enantiomers, simplifying the impurity profile of the crude product. In traditional proline-catalyzed reactions, the formation of racemic byproducts or the wrong enantiomer often necessitates complex chromatographic separations. In contrast, the novel catalyst system produces a crude product with such high optical purity that simple purification techniques like vacuum distillation or a single recrystallization step are sufficient to achieve pharmaceutical-grade specifications, often exceeding 99% ee. This mechanistic efficiency translates directly into process robustness, as the reaction tolerates a variety of substituents on both the 1,3-diketone and the unsaturated ketone components. Whether introducing allyl, propargyl, or methyl groups, the catalytic cycle remains efficient, demonstrating the versatility of this platform for generating diverse libraries of high-purity steroid precursors and other complex chiral building blocks.
How to Synthesize Chiral Diketone Compounds Efficiently
The practical implementation of this technology involves a straightforward sequence that begins with the preparation of the catalyst itself, followed by its application in the annulation reaction. The catalyst synthesis starts from a protected amino acid derivative, which is coupled with a secondary amine and subsequently reduced to the free amine form. Once the catalyst is secured, the main transformation involves mixing it with the acid additives and substrates. The patent outlines specific molar ratios and conditions that optimize the reaction kinetics and thermodynamic control. For detailed operational parameters, safety considerations, and exact stoichiometric calculations required for GMP manufacturing, please refer to the standardized synthesis guide below.
- Prepare the catalyst by reducing the corresponding amide precursor (Formula II) with lithium aluminum hydride in THF to obtain the chiral amine catalyst (Formula I).
- Mix the catalyst with a strong acid (e.g., triflic acid) in a minimal amount of organic solvent, then remove the solvent to form the active catalytic species.
- Combine the activated catalyst mixture with a weak acid, 1,3-diketone, and alpha,beta-unsaturated ketone, then heat to 60°C to perform the solvent-free Robinson annulation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology presents a multitude of strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the substantial simplification of the manufacturing process, which directly correlates to reduced operational expenditures. By eliminating the need for high-boiling, difficult-to-remove solvents and reducing the number of purification steps, the overall processing time is significantly shortened. This acceleration allows for faster turnover of reactor vessels and increased throughput without the need for additional capital investment in infrastructure. Furthermore, the stability of the catalyst and the robustness of the reaction conditions mean that batch-to-batch variability is minimized, ensuring a consistent supply of quality material that meets rigorous regulatory standards. This reliability is paramount for maintaining uninterrupted production schedules in the fast-paced pharmaceutical industry.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by several key factors, foremost among them being the low catalyst loading requirement. Operating effectively at just 2 mol%, the catalyst consumption is drastically lower than many contemporary organocatalytic systems that require 10 mol% or more. This reduction in precious chiral material usage leads to direct savings in raw material costs. Additionally, the capability to run the reaction under solvent-free conditions after the initial activation phase eliminates the massive costs associated with purchasing, storing, handling, and disposing of large volumes of organic solvents. The simplified workup procedure, which avoids complex chromatography in favor of distillation and crystallization, further reduces labor and utility costs, resulting in a leaner and more profitable manufacturing process.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the accessibility of the starting materials and the simplicity of the catalyst synthesis. The catalyst is derived from L-tert-leucine, a commercially available and abundant amino acid, ensuring that there are no bottlenecks related to exotic or scarce reagents. The synthetic route to the catalyst itself is short and high-yielding, allowing for rapid replenishment of catalytic stocks if needed. Moreover, the reaction's tolerance to scale-up means that production can be easily ramped up to meet surging demand without compromising on quality or yield. This scalability ensures that partners can rely on a steady flow of intermediates, mitigating the risks of stockouts that can derail downstream drug development timelines.
- Scalability and Environmental Compliance: In an era of increasing environmental regulation, the green chemistry attributes of this process offer a significant compliance advantage. The reduction in solvent usage directly lowers the facility's VOC emissions and hazardous waste generation, simplifying the permitting process and reducing waste disposal fees. The use of standard purification techniques like vacuum distillation and recrystallization is inherently easier to scale than column chromatography, which is often impractical for multi-kilogram or ton-scale production. The patent data confirms that both yield and enantioselectivity are maintained upon amplification, proving that the process is truly ready for commercial scale-up of complex chiral building blocks. This alignment with sustainability goals enhances the corporate social responsibility profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is critical for evaluating the feasibility of integrating this route into existing production workflows.
Q: What are the advantages of this catalyst over L-Proline for Wieland-Miescher ketone synthesis?
A: Unlike L-Proline, which often yields moderate enantioselectivity (70-75% ee) for Wieland-Miescher ketones requiring tedious recrystallization, the novel catalyst in CN103553931A achieves significantly higher ee values (up to 92%) and yields in a shorter timeframe, often under solvent-free conditions.
Q: Can this process be scaled up for industrial production?
A: Yes, the patent explicitly demonstrates that the method is suitable for large-scale production. The yield and enantioselectivity are maintained upon scale-up, and the purification methods involve standard industrial techniques like vacuum distillation and recrystallization.
Q: What represents the cost-saving potential of this method?
A: Cost savings are driven by the low catalyst loading (as low as 2 mol%), the elimination of high-boiling polar aprotic solvents like DMF or DMSO, and the simplified one-step reaction protocol which reduces processing time and waste treatment costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Diketone Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic technologies described in patent CN103553931A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory methods into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and enantioselectivity demonstrated in the patent are faithfully reproduced in our facilities. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch of chiral diketone intermediate meets the exacting standards required for global drug registration.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to not just a chemical supplier, but a strategic ally committed to optimizing your supply chain. Please contact us today to request specific COA data for our catalog items or to discuss route feasibility assessments for your custom synthesis needs. Let us help you accelerate your development timeline while maximizing cost efficiency and product quality.
