Revolutionizing Optically Active Selenide Production: A Metal-Free Catalytic Breakthrough for Global Supply Chains
Revolutionizing Optically Active Selenide Production: A Metal-Free Catalytic Breakthrough for Global Supply Chains
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more sustainable and efficient synthetic routes for complex molecular architectures. A pivotal advancement in this domain is detailed in Chinese Patent CN102516145A, which discloses a groundbreaking preparation method for optically active selenides. This technology leverages the power of chiral phosphoric acid catalysis to achieve the asymmetric selenofunctionalization of simple alkenes, a transformation that has historically been challenging to control with high stereoselectivity. By utilizing a Brønsted acid catalyst, this method bypasses the traditional reliance on stoichiometric chiral auxiliaries or toxic transition metals, offering a cleaner and more direct pathway to valuable organoselenium intermediates. These intermediates are critical building blocks in the synthesis of bioactive molecules, including antiviral and antitumor agents, making this innovation highly relevant for the pharmaceutical industry. The ability to generate chiral selenonium ions in situ, which are subsequently opened by nucleophiles, represents a significant leap forward in asymmetric catalysis, providing a robust platform for the scalable production of high-value fine chemicals.
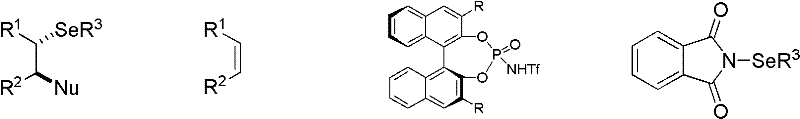
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active organoselenium compounds has been fraught with significant technical and economic hurdles that hinder large-scale adoption. Traditional approaches often rely heavily on the use of stoichiometric amounts of chiral selenium reagents or chiral auxiliaries, which not only increases the atom economy burden but also generates substantial chemical waste that requires costly disposal. Furthermore, many established protocols depend on transition metal catalysts, which introduce the risk of heavy metal contamination in the final product—a critical failure point for pharmaceutical intermediates where strict purity specifications are mandatory. The removal of trace metals often necessitates additional downstream processing steps, such as specialized scavenging or recrystallization, which drastically extends production lead times and inflates manufacturing costs. Additionally, conventional methods frequently suffer from limited substrate scope, struggling to accommodate simple, unfunctionalized alkenes without harsh reaction conditions that can degrade sensitive functional groups. These inefficiencies create a bottleneck in the supply chain, making it difficult for procurement teams to secure reliable sources of high-purity chiral selenides at a competitive price point.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent utilizes a highly efficient organocatalytic system driven by chiral phosphoric acids. This methodology fundamentally shifts the paradigm by employing a catalytic amount of a chiral Brønsted acid to activate the electrophilic selenium source, thereby generating a chiral selenonium ion intermediate with high fidelity. This activation mode allows for the use of simple, commercially available alkenes as starting materials, significantly reducing raw material costs and improving the overall sustainability profile of the synthesis. The reaction proceeds under remarkably mild conditions, typically ranging from -85°C to 25°C, which minimizes energy consumption and reduces the thermal stress on the reaction apparatus. Moreover, the metal-free nature of this catalysis ensures that the final product is free from heavy metal contaminants, simplifying the purification process to a standard column chromatography step. This streamlined workflow not only enhances the operational simplicity for R&D teams but also offers a clear path to commercial scale-up, addressing the critical need for cost reduction in pharmaceutical intermediate manufacturing while maintaining rigorous quality standards.
Mechanistic Insights into Chiral Phosphoric Acid-Catalyzed Selenofunctionalization
The core of this technological breakthrough lies in the unique ability of the chiral phosphoric acid catalyst to orchestrate the stereochemical outcome of the reaction through a well-defined transition state. The catalyst, typically a BINOL-derived phosphoric acid such as the one depicted in Formula III-a, acts as a dual activator, simultaneously coordinating with the electrophilic selenium source and organizing the spatial arrangement of the approaching alkene. This interaction facilitates the formation of a tightly bound chiral selenonium ion, where the chirality of the catalyst is effectively transferred to the substrate. The subsequent nucleophilic attack by species such as benzoic acid or alcohols occurs with high regioselectivity and stereoselectivity, leading to the ring-opening of the selenonium intermediate and the formation of the trans-configured selenide product. The steric bulk of the substituents on the catalyst backbone, such as the 2,4,6-triisopropylphenyl groups, plays a crucial role in shielding one face of the reactive intermediate, thereby enforcing the observed enantioselectivity. Understanding this mechanistic nuance is vital for process chemists aiming to optimize reaction parameters for specific substrates, as slight modifications to the catalyst structure or reaction temperature can fine-tune the enantiomeric excess.
Beyond the primary catalytic cycle, the mechanism also offers inherent advantages regarding impurity control and product purity, which are paramount for regulatory compliance in the pharmaceutical sector. Because the reaction relies on specific non-covalent interactions between the catalyst and the substrates, side reactions such as polymerization or non-selective background reactions are significantly suppressed compared to radical-based or metal-catalyzed alternatives. The use of mild nucleophiles like carboxylic acids further ensures that the functional group tolerance is high, preventing the degradation of sensitive moieties that might be present in complex drug candidates. The resulting impurity profile is generally cleaner, with the major byproducts being easily separable from the desired chiral selenide through standard silica gel chromatography. This predictability in the impurity spectrum allows quality control laboratories to establish robust analytical methods early in the development phase, reducing the risk of batch failures during commercial production. For R&D directors, this level of mechanistic clarity provides confidence in the reproducibility of the process, ensuring that the optical purity remains consistent across different batches and scales of operation.
How to Synthesize Optically Active Selenides Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction conditions and reagent quality to maximize yield and enantioselectivity. The process begins with the precise weighing of the chiral phosphoric acid catalyst, the electrophilic selenium source (typically N-phenylselenyl-phthalimide), and the chosen nucleophile, which are dissolved in an anhydrous solvent such as chloroform or dichloromethane. The alkene substrate is then introduced to the mixture, and the reaction vessel is cooled to the specified temperature, often requiring cryogenic equipment for optimal results with certain cyclic alkenes. Monitoring the reaction progress via TLC or HPLC is essential to determine the optimal quenching time, after which the mixture is treated with a base like triethylamine to neutralize the acid catalyst. The detailed standardized synthesis steps, including specific molar ratios and workup procedures for various substrates like cyclohexene and cyclopentene, are outlined below to guide technical teams in replicating this high-value transformation.
- Prepare the reaction mixture by combining the chiral phosphoric acid catalyst (Formula III), electrophilic selenium source (Formula IV), and nucleophile (NuH) in a suitable solvent such as chloroform or dichloromethane.
- Add the alkene substrate (Formula II) to the mixture and maintain the reaction temperature between -85°C and 25°C, stirring for 12 to 90 hours depending on the specific substrate reactivity.
- Quench the reaction with triethylamine, filter through a short silica plug, and purify the crude product via column chromatography to isolate the optically active selenide with high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this chiral phosphoric acid-catalyzed method presents a compelling value proposition centered on cost efficiency and supply reliability. The elimination of transition metal catalysts removes the necessity for expensive metal scavengers and the associated validation testing for residual metals, which translates directly into substantial cost savings in the manufacturing process. Furthermore, the use of simple alkenes as starting materials leverages widely available commodity chemicals, reducing dependency on specialized, high-cost precursors that are often subject to volatile market pricing and supply disruptions. The mild reaction conditions also contribute to lower energy expenditures and reduced wear on reactor equipment, enhancing the overall economic viability of the production line. From a supply chain perspective, the robustness of the organocatalytic system ensures consistent output quality, minimizing the risk of batch rejections that can delay deliveries to downstream customers. This reliability is crucial for maintaining uninterrupted production schedules for active pharmaceutical ingredients, positioning suppliers who adopt this technology as preferred partners for long-term contracts.
- Cost Reduction in Manufacturing: The transition to a metal-free organocatalytic process fundamentally alters the cost structure of producing optically active selenides by removing several expensive unit operations. Without the need for transition metals, manufacturers avoid the high capital and operational costs associated with metal removal technologies, such as specialized filtration systems or resin treatments. Additionally, the high atom economy of using simple alkenes means that less raw material is wasted, and the overall yield per kilogram of input is improved. The simplified purification protocol, often requiring only a single column chromatography step, reduces solvent consumption and labor hours compared to multi-step recrystallization processes. These cumulative efficiencies result in a significantly lower cost of goods sold (COGS), allowing companies to offer competitive pricing while maintaining healthy profit margins in the fine chemical market.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the accessibility and stability of the reagents required for this synthesis. Chiral phosphoric acids are stable, shelf-stable solids that can be sourced from multiple vendors, mitigating the risk of single-source bottlenecks that often plague specialized catalyst markets. The starting alkenes are bulk commodities with established global supply networks, ensuring that production can continue even during regional logistical challenges. Moreover, the mild nature of the reaction reduces the safety risks associated with high-pressure or high-temperature operations, leading to fewer unplanned shutdowns due to safety incidents. This operational stability ensures a steady flow of high-purity intermediates to clients, fostering trust and strengthening strategic partnerships between chemical suppliers and pharmaceutical manufacturers who demand just-in-time delivery capabilities.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory experiments to multi-ton commercial production is facilitated by the simplicity of the reaction setup and the absence of hazardous metal waste streams. The organocatalytic nature of the reaction aligns perfectly with green chemistry principles, as it generates less toxic waste and reduces the environmental footprint of the manufacturing facility. Regulatory compliance is streamlined because the final product does not require extensive testing for heavy metal residues, accelerating the release of batches for clinical or commercial use. The process is adaptable to continuous flow chemistry setups, which can further enhance throughput and safety profiles for large-scale operations. This scalability ensures that the technology can meet the growing global demand for chiral selenium compounds without compromising on environmental standards or regulatory requirements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric selenofunctionalization technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production portfolios. The versatility of the catalyst system and the breadth of the substrate scope are key factors that distinguish this approach from older methodologies, making it a versatile tool for modern organic synthesis.
Q: What is the maximum enantiomeric excess achieved in this selenide synthesis?
A: According to the patent data, the method achieves enantiomeric excess (ee) values of up to 84%, demonstrating effective stereocontrol for cyclic and acyclic alkene substrates.
Q: Does this process require transition metal catalysts?
A: No, this is a metal-free organocatalytic process utilizing chiral phosphoric acids (Brønsted acids), which eliminates the need for expensive transition metals and complex heavy metal removal steps.
Q: What types of alkenes are compatible with this asymmetric selenofunctionalization?
A: The method shows broad substrate scope, successfully converting simple cyclic alkenes like cyclohexene and cyclopentene, as well as acyclic alkenes such as cis-4-octene, into functionalized selenides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Selenide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this chiral phosphoric acid-catalyzed technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative patent data into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify enantiomeric excess and chemical purity. By leveraging our deep understanding of asymmetric organocatalysis, we can help you navigate the complexities of process optimization and regulatory filing, ensuring a smooth transition from development to market.
We invite you to collaborate with us to explore how this cutting-edge synthesis method can enhance your product portfolio and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. Together, we can build a resilient and efficient supply chain for optically active selenides that drives innovation and growth in the global pharmaceutical industry.
