Advanced One-Pot Synthesis of Azithromycin Impurity F for High-Purity QC Standards
The pharmaceutical industry's relentless pursuit of quality control in antibiotic manufacturing has brought significant attention to the precise characterization of impurities. Patent CN108727445B introduces a groundbreaking synthetic methodology for Azithromycin Impurity F, a critical reference standard required for the quality assurance of second-generation macrolide antibiotics. This technical disclosure addresses a long-standing bottleneck in the supply chain of analytical standards by shifting the synthetic starting point from scarce intermediates to the abundant bulk drug itself. By leveraging a tandem demethylation-formylation strategy, the invention enables the production of high-purity impurity standards essential for HPLC calibration and regulatory compliance. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and economically viable sourcing of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Azithromycin Impurity F relied heavily on the availability of Azithromycin Impurity I as the primary starting material, a dependency that introduced severe inefficiencies into the supply chain. Existing literature, such as CN104829662 and US2007/43214, describes routes where Impurity I undergoes formylation using reagents like methyl acetic anhydride or ethyl formate under basic conditions. However, the precursor Impurity I itself is notoriously difficult to synthesize in high yields, with reported efficiencies hovering around 55%, creating a compounding loss of material before the final step even begins. Furthermore, the reliance on freshly prepared methyl acetic anhydride poses logistical challenges due to its instability and tendency to decompose, necessitating immediate usage and complicating inventory management. These conventional pathways are not only cost-prohibitive due to the multi-step nature but also generate inconsistent batch qualities that fail to meet the rigorous purity demands of modern pharmacopoeia standards.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes Azithromycin directly as the raw material, effectively bypassing the isolation of the low-yield intermediate Impurity I. This one-pot transformation integrates the demethylation and formylation steps into a single streamlined operation, drastically simplifying the workflow and enhancing overall atom economy. The process employs a robust solvent system of methanol and water, which ensures the homogeneous dissolution of both the organic substrate and the inorganic base, thereby facilitating a more uniform reaction environment. By utilizing stable and commercially available ethyl formate as the formylating agent, the method eliminates the safety and stability risks associated with anhydride reagents. This strategic pivot from intermediate-based to API-based synthesis fundamentally alters the cost structure, making the production of this critical impurity standard significantly more accessible for large-scale quality control laboratories.
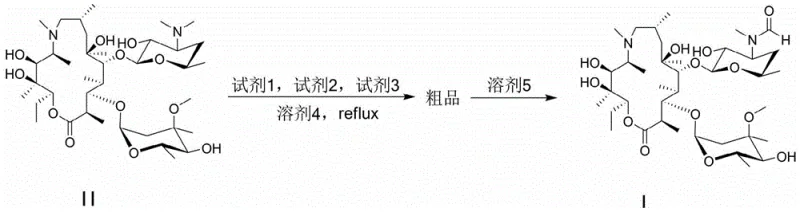
Mechanistic Insights into Iodine-Mediated Demethylation and Formylation
The core chemical transformation relies on a sophisticated interplay between oxidative demethylation and nucleophilic substitution, mediated by molecular iodine and potassium carbonate. In the initial phase, iodine acts as a mild oxidizing agent in the presence of the base to cleave the N-methyl group on the desosamine sugar moiety of the azithromycin macrocycle. This demethylation generates a secondary amine intermediate in situ, which is highly reactive and immediately available for the subsequent acylation step. The presence of water in the methanol solvent system is crucial, as it aids in the solubilization of potassium carbonate, ensuring that the basic conditions are maintained throughout the reflux period to drive the equilibrium forward. The reaction temperature is carefully optimized to 80°C, providing sufficient thermal energy to overcome the activation barrier for the N-demethylation without degrading the sensitive macrolide backbone.
Following the generation of the secondary amine, ethyl formate serves as the electrophilic source for the formyl group. The nucleophilic attack by the newly formed amine on the carbonyl carbon of ethyl formate results in the formation of the N-formyl derivative, which is the defining structural feature of Impurity F. The use of ethyl formate is particularly advantageous because it is less prone to hydrolysis compared to other acylating agents under these specific biphasic-like conditions, ensuring high conversion rates. The stoichiometry is tightly controlled, with a molar ratio of azithromycin to iodine maintained between 1:1.8 and 1:2.2 to ensure complete demethylation while minimizing side reactions such as over-oxidation. This precise control over reaction parameters allows for the suppression of unwanted byproducts, resulting in a crude product that is already of high quality before the final purification step.
How to Synthesize Azithromycin Impurity F Efficiently
The operational simplicity of this synthesis makes it highly attractive for both laboratory-scale standard preparation and potential commercial manufacturing. The process begins by dissolving azithromycin in a methanol-water mixture, followed by the sequential addition of iodine, potassium carbonate, and ethyl formate. The mixture is then heated to reflux for a duration of 4 to 6 hours, allowing the tandem reaction to proceed to completion. Upon cooling, the solvent is recovered via vacuum distillation, and the residue is washed with water to remove inorganic salts, yielding a coarse solid. This crude material is then subjected to a recrystallization process using acetone, which selectively precipitates the target impurity while leaving residual starting materials and minor byproducts in the solution.
- Dissolve Azithromycin in a methanol-water (4: 1) solvent system and add iodine, potassium carbonate, and ethyl formate.
- Heat the mixture to reflux at 80°C for 4-6 hours to facilitate demethylation and formylation.
- Recover solvent via vacuum distillation, wash with water, filter to obtain crude product, and recrystallize using acetone to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic benefits that extend beyond mere technical feasibility. The shift to using Azithromycin as the starting material leverages an existing, high-volume supply chain, thereby insulating the production of the impurity standard from the volatility associated with niche intermediate markets. This vertical integration of the synthesis pathway ensures a more predictable lead time and reduces the risk of supply disruptions that often plague multi-step processes relying on specialized precursors. Additionally, the elimination of unstable reagents like methyl acetic anhydride simplifies warehouse management and reduces the costs associated with hazardous material handling and disposal. The overall process intensification, characterized by fewer unit operations and higher yields, translates directly into a lower cost of goods sold, enabling more competitive pricing for high-purity reference standards.
- Cost Reduction in Manufacturing: The economic advantage of this method is primarily driven by the substitution of expensive, low-yield intermediates with readily available Azithromycin bulk drug. By consolidating two reaction steps into a single pot, the process significantly reduces labor hours, energy consumption, and solvent usage, leading to substantial operational expenditure savings. Furthermore, the high recovery rate of the solvent through distillation allows for recycling, further diminishing the environmental footprint and raw material costs. The use of stable ethyl formate eliminates the waste and cost associated with the frequent preparation of fresh reagents, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: Sourcing Azithromycin is far more reliable than sourcing specialized impurities like Impurity I, as the former is produced on a kiloton scale globally. This abundance ensures that production schedules for the impurity standard can be maintained consistently without being held hostage by the lead times of upstream intermediate suppliers. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require exotic catalysts or cryogenic temperatures, further enhances the reliability of the supply chain. This stability is crucial for pharmaceutical companies that require a steady stream of certified reference materials for ongoing batch release testing and regulatory filings.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind, utilizing common solvents like methanol, water, and acetone that are easily managed in large-scale reactors. The reduction in the number of synthetic steps inherently lowers the E-factor (environmental factor) of the process by minimizing waste generation per kilogram of product. The aqueous workup and simple filtration steps reduce the complexity of downstream processing, making it easier to comply with increasingly stringent environmental regulations regarding solvent emissions and waste disposal. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of Azithromycin Impurity F. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the method's superiority over traditional routes. Understanding these details is essential for quality control managers and technical buyers who need to validate the suitability of this impurity standard for their specific analytical methods.
Q: Why is starting from Azithromycin superior to using Impurity I?
A: Starting directly from Azithromycin bypasses the isolation of Impurity I, which historically suffers from low yields (around 55%). This one-pot approach significantly improves overall process efficiency and reduces raw material costs.
Q: What purity levels can be achieved with this synthesis method?
A: The patented method utilizes a specific acetone recrystallization step that consistently yields Azithromycin Impurity F with a purity exceeding 99.5%, meeting stringent requirements for reference standards.
Q: Is the formylation reagent stable for industrial storage?
A: Yes, unlike methyl acetic anhydride used in older methods which requires fresh preparation due to instability, this process uses ethyl formate, which is chemically stable, safe to store, and highly efficient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azithromycin Impurity F Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity impurity standards play in ensuring the safety and efficacy of life-saving antibiotics. Our team of expert chemists has thoroughly analyzed the patented one-pot synthesis route and is fully equipped to replicate and optimize this process for commercial scale-up. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of Azithromycin Impurity F. Our state-of-the-art facilities are supported by rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch meets or exceeds the >99.5% purity threshold required for pharmacopoeial compliance.
We invite pharmaceutical manufacturers and quality control laboratories to collaborate with us to secure their supply of this essential reference material. By leveraging our technical expertise and manufacturing capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your quality control operations remain uninterrupted and compliant with global regulatory standards.
