Scalable Synthesis of Geminal Diboron Compounds via Selective Olefin 1,1-Diboronation
The landscape of organoboron chemistry is undergoing a significant transformation driven by the urgent need for more sustainable and cost-effective synthetic methodologies. Patent CN111217844A introduces a groundbreaking approach for the preparation of geminal diboron compounds through the selective 1,1-diboronation of olefins. This technology addresses critical bottlenecks in the synthesis of versatile building blocks used extensively in cross-coupling reactions and medicinal chemistry. By leveraging a cyclopentadienyl pre-transition metal catalyst system, specifically zirconocene dichloride, the process achieves high atom economy without the necessity for expensive noble metals or stoichiometric hydrogen acceptors. This innovation represents a paradigm shift for manufacturers seeking a reliable pharmaceutical intermediate supplier capable of delivering complex organoboron structures with enhanced efficiency.
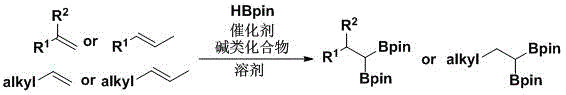
The strategic value of this patent lies in its ability to utilize readily available olefin feedstocks to generate high-value gem-diboron products. Unlike traditional methods that often suffer from poor regioselectivity or require harsh conditions, this catalytic system operates under a protective atmosphere at moderate temperatures ranging from 60°C to 150°C. The versatility of the method is evidenced by its compatibility with a broad spectrum of substrates, including aryl olefins, internal olefins, and long-chain aliphatic olefins. For procurement managers and supply chain heads, this translates to a robust manufacturing process that minimizes dependency on volatile precious metal markets while ensuring a consistent supply of critical intermediates for downstream drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of gem-diboron compounds has been plagued by significant inefficiencies and economic drawbacks that hinder large-scale adoption. Traditional routes often involve the dehalogenation of gem-dihalides or the alkylation of diboron methane with alkyl halides, processes that generate substantial amounts of salt waste and require multiple synthetic steps. Furthermore, existing catalytic methods frequently rely on noble metals such as palladium or rhodium, which not only inflate the cost of goods sold but also introduce stringent requirements for metal residue removal in pharmaceutical applications. Another major limitation is the reliance on bis(pinacolato)diboron (B2pin2) as the boron source, which is considerably more expensive than pinacolborane (HBpin) and often results in lower atom economy due to the formation of boron-containing byproducts. These factors collectively create a high barrier to entry for cost-sensitive manufacturing sectors.
The Novel Approach
The methodology disclosed in CN111217844A offers a compelling solution by directly utilizing pinacolborane (HBpin) as the boron source in conjunction with an earth-abundant zirconium catalyst. This novel approach eliminates the need for external hydrogen acceptors, thereby streamlining the reaction stoichiometry and improving the overall atom economy. The use of simple alkali compounds like lithium methoxide as activators further simplifies the reaction setup, removing the need for complex ligand systems that are often sensitive to air and moisture. By shifting from noble metals to zirconium-based catalysts, the process drastically reduces the raw material costs associated with the catalytic cycle. Additionally, the reaction demonstrates exceptional selectivity for the 1,1-diboronation product, minimizing the formation of saturated alkyl borate byproducts that complicate purification. This streamlined workflow is ideally suited for cost reduction in fine chemical manufacturing, offering a clear path to more competitive pricing for high-purity organoboron intermediates.
Mechanistic Insights into Zirconocene-Catalyzed 1,1-Diboronation
The core of this technological advancement lies in the unique reactivity of the cyclopentadienyl zirconium species generated in situ. The reaction initiates with the activation of the zirconocene precatalyst, such as Cp2ZrCl2, by the alkali base, likely forming a reactive zirconium-hydride or zirconium-boryl species. This active catalyst then interacts with the olefin substrate to facilitate the insertion of the carbon-carbon double bond into the metal-boron or metal-hydride bond. The specific electronic and steric environment provided by the cyclopentadienyl ligands directs the regioselectivity towards the geminal position, ensuring that both boron units are installed on the same carbon atom. This level of control is crucial for R&D directors who require precise structural outcomes for subsequent functionalization steps. The mechanism avoids the radical pathways often seen in other hydroboration reactions, which can lead to scrambling of the boron positions and reduced product purity.
Impurity control is another critical aspect where this mechanism excels. The catalytic cycle is designed to suppress the formation of mono-boronated species or over-reduced alkyl chains, which are common side reactions in hydroboration chemistry. The use of a protective nitrogen or argon atmosphere prevents oxidative degradation of the sensitive zirconium intermediates, ensuring high reproducibility across different batches. For quality assurance teams, this means a cleaner crude reaction profile that simplifies downstream purification processes such as silica gel chromatography or crystallization. The ability to tolerate various functional groups, including halogens, ethers, and amines, without interfering with the catalytic cycle further underscores the robustness of this mechanistic pathway. This makes it an ideal candidate for the commercial scale-up of complex polymer additives and pharmaceutical precursors where impurity profiles are strictly regulated.
How to Synthesize Geminal Diboron Compounds Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, making it accessible for both laboratory research and industrial production facilities. The general procedure involves dissolving the olefin substrate, the zirconium catalyst, and the alkali activator in a solvent like toluene, followed by the addition of pinacolborane. The reaction is then heated to the optimal temperature, typically around 100°C to 130°C, and stirred for a period ranging from 1 to 16 hours depending on the specific substrate reactivity. Detailed standardized synthesis steps see the guide below.
- Dissolve the olefin compound, cyclopentadienyl pre-transition metal catalyst (e.g., Cp2ZrCl2), and an alkali compound (e.g., MeOLi) in a suitable solvent such as toluene.
- Add the borane compound (pinacolborane) to the reaction mixture under a protective atmosphere like nitrogen or argon.
- Heat the reaction mixture to a temperature between 60°C and 150°C and stir for 1 to 16 hours to complete the selective 1,1-diboronation.
- Upon completion, separate the product through standard workup procedures including solvent removal and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this zirconium-catalyzed diboronation technology offers tangible strategic benefits that extend beyond mere chemical novelty. The shift away from noble metal catalysts mitigates the financial risks associated with the fluctuating prices of palladium and rhodium, providing a more stable cost structure for long-term production contracts. Moreover, the use of pinacolborane instead of diboron reagents significantly lowers the input material costs, as HBpin is generally more affordable and easier to handle on a large scale. The simplified reaction conditions, which do not require high-pressure reactors or cryogenic temperatures, reduce the capital expenditure needed for plant modifications, allowing for faster integration into existing manufacturing lines. These factors combine to create a supply chain that is both resilient and cost-efficient.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the use of cheaper boron sources result in substantial cost savings per kilogram of product. By avoiding the need for additional hydrogen acceptors, the process reduces the total mass of reagents required, which in turn lowers waste disposal costs and improves the overall green chemistry profile of the manufacturing site. The high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts, ensuring that the yield of the desired gem-diboron compound is maximized. This efficiency translates directly into a lower cost of goods sold, enabling more competitive pricing strategies in the global market for fine chemicals.
- Enhanced Supply Chain Reliability: The reliance on earth-abundant zirconium ensures a secure supply of the catalyst, free from the geopolitical constraints that often affect noble metal availability. The robustness of the reaction conditions allows for flexible scheduling and batch processing, reducing the risk of production delays caused by sensitive operational parameters. Furthermore, the compatibility with common solvents like toluene and hexane simplifies logistics and inventory management, as these materials are readily available from multiple suppliers. This reliability is crucial for maintaining continuous production flows and meeting the tight delivery deadlines demanded by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively across a wide range of substrate concentrations and reaction scales. The absence of toxic heavy metals in the catalyst system simplifies the regulatory compliance process, particularly for products intended for human consumption. Waste streams are easier to treat due to the lower toxicity of zirconium residues compared to palladium or platinum, aligning with increasingly stringent environmental regulations. This environmental advantage not only reduces the burden on waste treatment facilities but also enhances the corporate sustainability profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this olefin diboronation technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for potential partners and licensees. Understanding these details is essential for evaluating the feasibility of integrating this method into your current production portfolio.
Q: What are the primary advantages of this zirconium-catalyzed method over traditional noble metal catalysis?
A: This method utilizes earth-abundant zirconium catalysts instead of expensive noble metals like palladium or rhodium, significantly reducing raw material costs. Furthermore, it avoids the need for additional hydrogen acceptors or expensive diboron reagents like B2pin2, leading to superior atom economy and simplified waste management.
Q: Does this process support a wide range of functional groups for pharmaceutical applications?
A: Yes, the protocol demonstrates excellent functional group tolerance. It successfully processes aryl olefins with electron-donating and electron-withdrawing substituents, as well as heterocyclic systems like thiophene, furan, and carbazole derivatives, making it highly suitable for synthesizing complex pharmaceutical intermediates.
Q: How does this method impact the scalability of organoboron production?
A: The reaction operates under relatively mild conditions (60-150°C) in common solvents like toluene, without requiring specialized high-pressure equipment. The simplicity of the operation and the robustness of the catalyst system facilitate straightforward scale-up from laboratory grams to industrial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Geminal Diboron Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this zirconium-catalyzed synthesis route for the next generation of organoboron intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of gem-diboron compounds delivered meets the highest industry standards. We are committed to leveraging this innovative technology to provide our clients with a competitive edge in the development of novel therapeutics and advanced materials.
We invite you to engage with our technical procurement team to discuss how this methodology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this catalytic system for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments for your target molecules. Let us collaborate to optimize your synthesis strategy and secure a reliable supply of high-quality pharmaceutical intermediates for your future success.
