Advanced Zirconium-Catalyzed 1,1-Diboronation for High-Purity Pharmaceutical Intermediates Manufacturing
Introduction to Patent CN111217844A
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex molecular architectures, particularly those involving organoboron motifs which are indispensable in modern cross-coupling reactions. Patent CN111217844A introduces a groundbreaking method for the selective 1,1-diboronation of olefins to prepare geminal diboron compounds, addressing critical limitations in current synthetic methodologies. This technology leverages a cyclopentadienyl pre-transition metal catalyst system, specifically utilizing earth-abundant zirconium complexes, to facilitate the direct addition of two boron units across a carbon-carbon double bond. The significance of this innovation lies in its ability to transform readily available and inexpensive olefin feedstocks into high-value gem-diboron intermediates with exceptional atom economy. By operating under relatively mild conditions ranging from 60°C to 150°C and utilizing simple alkali bases, this process offers a robust platform for the scalable production of versatile synthetic building blocks essential for drug discovery and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for synthesizing gem-diboron compounds have historically been plagued by significant economic and operational inefficiencies that hinder their widespread industrial adoption. Conventional routes often rely on the dehalogenation of gem-dihalides or the alkylation of diboron methane with alkyl halides, processes that typically generate substantial amounts of stoichiometric salt waste and require harsh reaction conditions. Furthermore, many existing methods necessitate the use of expensive noble metal catalysts such as palladium or rhodium, coupled with sophisticated and air-sensitive phosphine ligands, which drastically inflate the cost of goods sold (COGS). Another major drawback is the reliance on bis(pinacolato)diboron (B2pin2) as the boron source, which is considerably more expensive than pinacolborane (HBpin) and often suffers from lower atom economy due to the loss of half the boron reagent during the transformation. These factors collectively create a bottleneck for procurement teams aiming to reduce costs in fine chemical manufacturing and limit the accessibility of these valuable intermediates for large-scale commercial projects.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN111217844A presents a streamlined and economically superior alternative that fundamentally reshapes the synthesis landscape for gem-diboron species. This novel approach utilizes pinacolborane (HBpin) as the sole boron source in conjunction with a cost-effective cyclopentadienyl zirconium catalyst, such as Cp2ZrCl2, to achieve direct 1,1-diboronation without the need for external hydrogen acceptors. The reaction mechanism involves a unique hydrogen discharge pathway that maximizes atom economy, ensuring that nearly all reactant atoms are incorporated into the final product or benign byproducts. As illustrated in the general reaction scheme below, this method allows for the direct conversion of diverse olefin substrates into gem-diboron products with high selectivity.
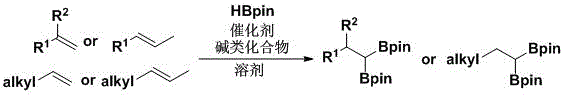
The operational simplicity is further enhanced by the use of common solvents like toluene and the absence of complex ligand systems, which simplifies the workup procedure and reduces the environmental footprint associated with solvent disposal and catalyst removal. This shift towards base-metal catalysis and simpler reagents represents a paradigm shift for supply chain heads looking to secure reliable sources of complex organic intermediates while minimizing dependency on volatile noble metal markets.
Mechanistic Insights into Zirconium-Catalyzed 1,1-Diboronation
The catalytic cycle underpinning this transformation is driven by the activation of the boron-hydrogen bond by the zirconium center, facilitated by the presence of an alkali alkoxide base such as lithium methoxide (MeOLi). The base plays a critical role in generating the active zirconium-boryl species in situ, which subsequently undergoes migratory insertion with the olefin substrate. Unlike traditional hydroboration which typically yields mono-boronated products with anti-Markovnikov selectivity, this specialized catalyst system promotes a second borylation event at the same carbon center, leading to the formation of the geminal diboron motif. The regioselectivity is meticulously controlled by the steric and electronic properties of the cyclopentadienyl ligands on the zirconium atom, ensuring that the reaction proceeds exclusively via the 1,1-addition pathway rather than the thermodynamically competitive 1,2-diboronation. This precise control is vital for R&D directors who require high-purity intermediates with defined structural integrity for downstream Suzuki-Miyaura couplings or other functionalization reactions.
Furthermore, the reaction system exhibits remarkable functional group tolerance, accommodating a wide array of substituents on the olefin backbone without compromising yield or selectivity. The patent data demonstrates successful conversion of styrenes bearing electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as fluoro and chloro, indicating a robust catalytic manifold that is insensitive to subtle electronic variations. The substrate scope extends beyond simple aryl olefins to include internal olefins, aliphatic chains, and even heterocyclic systems like thiophenes and carbazoles, as depicted in the structural diversity shown below.
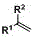
This broad compatibility minimizes the need for protecting group strategies, thereby shortening synthetic routes and reducing the overall number of unit operations required to reach the target molecule. From an impurity control perspective, the absence of noble metals eliminates the risk of heavy metal contamination, a critical quality attribute for pharmaceutical intermediates intended for clinical use. The clean reaction profile also facilitates easier purification, often requiring only standard silica gel chromatography or crystallization to achieve analytical purity, which is a significant advantage for process chemists optimizing for commercial viability.
How to Synthesize Gem-Diboron Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and reproducibility. The general protocol involves dissolving the olefin compound, the cyclopentadienyl pre-transition metal catalyst, and the alkali compound in a dry, degassed solvent under an inert atmosphere to prevent catalyst deactivation by oxygen or moisture. Following the dissolution, the pinacolborane reagent is added, and the mixture is heated to the optimal temperature range, typically between 100°C and 130°C for styrene derivatives, although the patent specifies a broader window of 60°C to 150°C depending on the substrate reactivity. The detailed standardized synthesis steps, including specific molar ratios and workup procedures validated by the patent examples, are outlined in the guide below.
- Dissolve the olefin substrate, cyclopentadienyl pre-transition metal catalyst (e.g., Cp2ZrCl2), and an alkali compound (e.g., MeOLi) in a suitable solvent like toluene.
- Add the borane compound (pinacolborane) to the reaction mixture under a protective nitrogen or argon atmosphere.
- Heat the reaction mixture to between 60°C and 150°C for 1 to 16 hours, then separate and purify the resulting gem-diboron compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this zirconium-catalyzed diboronation technology offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic reduction of raw material costs achieved by substituting expensive noble metal catalysts and diboron reagents with earth-abundant zirconium salts and economical pinacolborane. This substitution not only lowers the direct material cost but also mitigates supply risks associated with the geopolitical volatility of precious metal markets, ensuring a more stable and predictable cost structure for long-term production contracts. Additionally, the simplified reaction design eliminates the need for specialized ligands and complex additive packages, further streamlining the procurement of ancillary chemicals and reducing inventory complexity.
- Cost Reduction in Manufacturing: The elimination of noble metals such as palladium or rhodium from the catalytic cycle results in significant savings on catalyst procurement, which is often a major cost driver in fine chemical synthesis. Moreover, the use of pinacolborane instead of bis(pinacolato)diboron improves the atom economy of the reaction, meaning less reagent is wasted per kilogram of product formed, directly translating to lower variable costs. The simplified downstream processing, devoid of heavy metal scavenging steps, reduces the consumption of purification media and solvents, contributing to an overall leaner and more cost-efficient manufacturing process that enhances profit margins for high-volume commodities.
- Enhanced Supply Chain Reliability: By relying on readily available and commodity-grade starting materials like styrenes and simple olefins, this method decouples production from the supply constraints of exotic or custom-synthesized precursors. The robustness of the catalyst system against air and moisture, relative to sensitive noble metal complexes, allows for more flexible handling and storage conditions, reducing the risk of batch failures due to reagent degradation. This reliability ensures consistent delivery schedules and reduces the lead time for high-purity synthetic building blocks, enabling pharmaceutical partners to accelerate their own development timelines without being bottlenecked by intermediate availability.
- Scalability and Environmental Compliance: The reaction conditions described in the patent, utilizing common solvents like toluene and moderate temperatures, are inherently scalable from gram to multi-ton scales without requiring specialized high-pressure or cryogenic equipment. The high atom economy and lack of stoichiometric salt byproducts align with green chemistry principles, minimizing the generation of hazardous waste and simplifying effluent treatment protocols. This environmental compatibility facilitates regulatory approval and supports corporate sustainability goals, making the process attractive for companies aiming to reduce their carbon footprint while maintaining high production throughput and operational safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diboronation technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational boundaries and potential applications of the method for technical decision-makers evaluating its fit for their specific project requirements. Understanding these nuances is crucial for assessing the feasibility of integrating this chemistry into existing manufacturing workflows or new product development pipelines.
Q: What are the advantages of using Cp2ZrCl2 over noble metal catalysts for diboronation?
A: Using Cp2ZrCl2 eliminates the need for expensive noble metals like palladium or rhodium, significantly reducing raw material costs. Additionally, this earth-abundant catalyst system avoids the requirement for complex phosphine ligands, simplifying the reaction setup and downstream purification processes.
Q: Does this method require additional hydrogen acceptors?
A: No, unlike some conventional methods that require external hydrogen acceptors or expensive diboron reagents like B2pin2, this novel approach utilizes pinacolborane (HBpin) directly. The reaction proceeds via direct hydrogen discharge, which improves atom economy and reduces waste generation.
Q: What is the substrate scope for this 1,1-diboronation reaction?
A: The method demonstrates excellent compatibility with a wide range of olefins, including aryl olefins (styrenes), internal olefins, and long-chain aliphatic olefins. It tolerates various functional groups such as halogens, ethers, and amines, making it highly versatile for synthesizing complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gem-Diboron Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this zirconium-catalyzed 1,1-diboronation technology in advancing the synthesis of next-generation pharmaceutical intermediates and functional materials. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of gem-diboron compounds delivered meets the highest standards of quality and consistency required by global regulatory bodies.
We invite you to collaborate with our technical team to explore how this cost-effective and scalable synthesis route can optimize your supply chain and reduce your overall manufacturing expenses. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise in organoboron chemistry can serve as a strategic asset for your organization's growth and innovation objectives.
