Advanced Pd-Catalyzed Synthesis of 9-Hydroxyfluorene-9-Carboxylates for Scalable Pharmaceutical Production
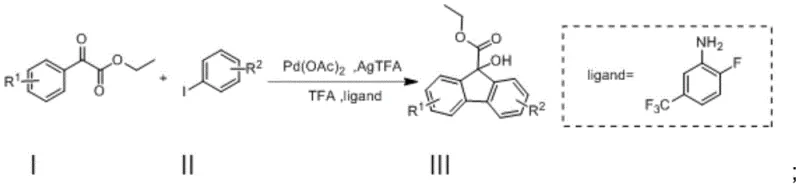
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for complex heterocyclic scaffolds, particularly fluorene derivatives which serve as critical precursors for bioactive molecules and optoelectronic materials. Patent CN111718262A introduces a groundbreaking methodology for the preparation of 9-hydroxyfluorene-9-carboxylate compounds, addressing long-standing challenges in substrate availability and operational complexity. This innovation leverages a palladium-catalyzed intramolecular cyclization strategy that transforms simple benzoylformate esters and iodobenzenes into high-value fluorene cores in a single synthetic operation. For R&D directors and procurement specialists, this represents a significant shift towards more efficient, cost-effective, and environmentally sustainable manufacturing processes. The protocol eliminates the need for exotic starting materials often required in traditional silver-catalyzed pathways, instead utilizing commercially accessible building blocks that streamline the supply chain. By integrating this technology, manufacturers can achieve substantial reductions in process mass intensity while maintaining rigorous purity standards essential for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 9-hydroxyfluorene derivatives has relied heavily on silver-catalyzed reactions involving diphenylacetylene and specialized aldehyde precursors, a route fraught with logistical and economic inefficiencies. These conventional methods often necessitate the use of expensive and difficult-to-source starting materials, such as 3-(2-formylphenyl)propynoic acid methyl esters, which significantly inflate raw material costs and extend lead times for procurement teams. Furthermore, the traditional AgNTf2 catalytic systems typically operate in chlorinated solvents like dichloroethane, raising serious environmental compliance concerns regarding waste disposal and solvent recovery. The structural limitations of these older methodologies also restrict the diversity of substituents that can be introduced onto the fluorene core, often yielding only specific regioisomers like 11-hydroxy-6-phenyl-11H-benzo[a]fluorene derivatives. This lack of versatility forces process chemists to develop custom synthetic routes for each new analog, resulting in prolonged development cycles and increased R&D expenditure. Additionally, the post-reaction workup in these legacy processes frequently involves harsh acidic or basic conditions to isolate the product, which can degrade sensitive functional groups and complicate purification efforts.
The Novel Approach
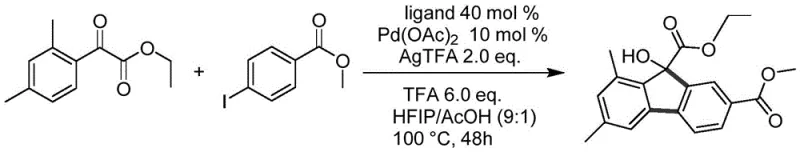
In stark contrast, the novel approach detailed in the patent utilizes a palladium-catalyzed oxidative cyclization that dramatically simplifies the synthetic landscape for 9-hydroxyfluorene-9-carboxylates. By employing benzoylformate compounds and iodobenzenes as the primary substrates, the method unlocks access to a vast library of commercially available starting materials, thereby enhancing supply chain resilience and reducing dependency on niche chemical suppliers. The reaction proceeds efficiently in a mixed solvent system of hexafluoroisopropanol (HFIP) and acetic acid, which not only facilitates the cyclization but also minimizes the environmental footprint compared to chlorinated alternatives. Crucially, this one-step transformation achieves impressive yields ranging from 60% to 90%, demonstrating high atom economy and reducing the burden on downstream purification units. The use of 2-fluoro-5-trifluoromethylaniline as a specialized ligand further optimizes the catalytic cycle, ensuring high selectivity and minimizing the formation of unwanted by-products. This streamlined process allows for direct purification via column chromatography without the need for aggressive acid-base extractions, preserving the integrity of sensitive functional groups and accelerating the time-to-market for new pharmaceutical candidates.
Mechanistic Insights into Pd-Catalyzed Oxidative Cyclization
The core of this technological breakthrough lies in the intricate palladium catalytic cycle that drives the formation of the fluorene skeleton through a sequence of oxidative addition, migratory insertion, and reductive elimination steps. The reaction initiates with the oxidative addition of the aryl iodide to the Pd(II) species, generating a reactive organopalladium intermediate that is primed for subsequent carbon-carbon bond formation. The presence of trifluoroacetic acid (TFA) and the specific aniline ligand plays a pivotal role in stabilizing the active catalytic species and facilitating the C-H activation step required for ring closure. Silver trifluoroacetate (AgTFA) serves as a stoichiometric oxidant, regenerating the active Pd(II) catalyst from the Pd(0) species formed after reductive elimination, thus sustaining the catalytic turnover. This mechanistic pathway is highly tolerant of various electronic environments on the aromatic rings, allowing for the successful incorporation of electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as esters and nitro functionalities. The robustness of this mechanism ensures consistent performance across a wide range of substrate combinations, providing process chemists with a reliable tool for diversifying their molecular libraries without needing to re-optimize reaction conditions for every new analog.
From an impurity control perspective, the mild reaction conditions and high selectivity of this Pd-catalyzed system significantly reduce the generation of complex by-product profiles that often plague fluorene syntheses. Traditional methods involving strong Lewis acids or harsh thermal conditions frequently lead to polymerization or decomposition side reactions, creating difficult-to-remove impurities that compromise the final API purity. In this novel protocol, the use of HFIP as a solvent helps to suppress non-specific radical pathways, while the controlled oxidation potential of AgTFA prevents over-oxidation of the sensitive hydroxyl and ester functionalities. The result is a crude reaction mixture that is remarkably clean, allowing for straightforward isolation of the target 9-hydroxyfluorene-9-carboxylate via standard silica gel chromatography. This high level of chemical fidelity is critical for pharmaceutical applications where strict limits on genotoxic impurities and heavy metal residues must be met. By minimizing the formation of side products at the source, manufacturers can reduce the number of purification cycles required, leading to higher overall throughput and lower production costs per kilogram of finished intermediate.
How to Synthesize 9-Hydroxyfluorene-9-Carboxylate Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction monitoring to maximize yield and purity. The process begins by combining the benzoylformate ester and the iodobenzene derivative in a pressure-resistant vessel with the catalytic system comprising Pd(OAc)2, AgTFA, TFA, and the fluorinated aniline ligand. The choice of solvent ratio, specifically the 9:1 mixture of HFIP to acetic acid, is critical for solubilizing the reactants and maintaining the optimal acidity for the cyclization event. Reaction progress should be meticulously tracked using thin-layer chromatography (TLC) on F-254 plates, looking for the complete consumption of the starting iodobenzene and the stabilization of the product spot intensity. Once the reaction reaches completion, typically within 24 to 48 hours at 100°C, the workup procedure involves a gentle quench with saturated aqueous sodium bicarbonate to neutralize residual acids without inducing hydrolysis of the ester groups. Detailed standardized synthesis steps follow below to ensure reproducibility and safety during scale-up operations.
- Mix benzoylformate compound I and iodobenzene compound II in a solvent mixture of HFIP and AcOH, adding Pd(OAc)2, AgTFA, TFA, and the specific aniline ligand.
- Heat the reaction mixture to 90-100°C and stir for 24-48 hours, monitoring progress via TLC until starting materials are consumed.
- Quench with saturated sodium bicarbonate, extract with dichloromethane, dry, and purify the crude product using column chromatography to obtain high-purity target compounds.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Pd-catalyzed methodology offers transformative benefits that directly impact the bottom line and operational agility. The primary advantage stems from the utilization of commodity chemicals as starting materials; benzoylformates and iodobenzenes are produced on a massive industrial scale for various applications, ensuring a stable and competitive pricing structure that shields manufacturers from the volatility associated with specialty reagents. This shift away from exotic substrates significantly reduces the risk of supply disruptions, a critical factor for companies managing long-term contracts for API production. Furthermore, the simplification of the synthetic route from multi-step sequences to a single pot operation drastically cuts down on labor hours, energy consumption, and equipment occupancy time. The elimination of harsh post-treatment steps not only enhances worker safety but also reduces the load on wastewater treatment facilities, aligning with increasingly stringent global environmental regulations. These cumulative efficiencies translate into a more resilient supply chain capable of responding rapidly to market demands while maintaining healthy profit margins.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the high atom economy and the avoidance of expensive, proprietary catalysts or ligands that characterize older technologies. By using palladium acetate in conjunction with a simple fluorinated aniline ligand, the catalyst loading remains low while maintaining high turnover numbers, effectively minimizing the cost contribution of precious metals to the final product. Additionally, the ability to purify the crude product directly via column chromatography without extensive aqueous workups or recrystallizations reduces solvent usage and waste disposal fees. The high yields reported, consistently reaching up to 90% in optimized examples, mean that less raw material is wasted, further driving down the cost of goods sold (COGS). This cost structure makes the production of complex fluorene intermediates commercially viable even for price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: Reliability in the chemical supply chain is paramount, and this method secures it by decoupling production from the availability of hard-to-find precursors. Since the key building blocks are standard catalog items available from multiple global suppliers, procurement teams can easily qualify alternative sources to mitigate single-point-of-failure risks. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, reducing the frequency of batch failures and reworks. This stability allows for more accurate forecasting and inventory planning, ensuring that downstream formulation teams receive their intermediates on schedule. Moreover, the scalability of the reaction from gram to kilogram scales has been demonstrated, providing confidence that supply can be ramped up quickly to meet surges in demand without requiring major process re-engineering.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but this one-pot cyclization is inherently designed for scalability due to its homogeneous nature and moderate temperature requirements. The use of HFIP, while a specialized solvent, is recoverable and reusable, fitting well into closed-loop solvent management systems that modern chemical plants employ. From an environmental standpoint, the reduction in waste generation is significant; the absence of heavy metal salts like silver triflimide and the avoidance of chlorinated solvents simplify the effluent profile. This makes obtaining environmental permits for production easier and reduces the long-term liability associated with hazardous waste storage. Companies adopting this green chemistry approach can also leverage it for sustainability reporting, appealing to eco-conscious investors and partners in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and practical application scenarios. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific capabilities and limitations observed during the development of this method, ensuring that expectations are aligned with experimental reality. Whether you are concerned about substrate compatibility or regulatory compliance, these insights offer a clear path forward for leveraging this innovation.
Q: What are the key advantages of this Pd-catalyzed method over traditional AgNTf2 catalysis?
A: The Pd-catalyzed method utilizes readily available benzoylformate and iodobenzene substrates, operates under milder conditions without strong acid/base post-treatment, and achieves high yields (60-90%) with simpler purification via column chromatography.
Q: Can this synthesis method tolerate diverse functional groups on the aromatic rings?
A: Yes, the method demonstrates excellent substrate scope, successfully accommodating substituents such as methyl, methoxy, ester, and nitro groups on both the benzoylformate and iodobenzene components without significant loss in efficiency.
Q: Is this process suitable for large-scale commercial manufacturing of API intermediates?
A: Absolutely. The one-step cyclization, use of common solvents like HFIP/AcOH, and straightforward workup procedure make it highly scalable and environmentally friendly, reducing waste discharge compared to multi-step conventional routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Hydroxyfluorene-9-Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the Pd-catalyzed cyclization described in CN111718262A for producing high-performance pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch of 9-hydroxyfluorene-9-carboxylate complies with the highest industry standards. Our state-of-the-art facilities are equipped to handle the specific solvent systems and catalytic requirements of this novel route, guaranteeing consistent quality and supply continuity for our global partners. By choosing us as your CDMO partner, you gain access to a wealth of technical expertise dedicated to optimizing yield and minimizing costs without compromising on safety or environmental responsibility.
We invite you to collaborate with us to explore how this cutting-edge synthesis technology can enhance your product pipeline and reduce your overall manufacturing expenses. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data for our fluorene derivatives and to discuss route feasibility assessments for your custom projects. Together, we can accelerate the development of next-generation therapeutics by leveraging the power of innovative organic synthesis and reliable supply chain management.
