Advanced Copper-Catalyzed Synthesis of Amino-Substituted Arylates for Commercial Scale-Up
The synthesis of amino-substituted arylate compounds represents a critical challenge in the development of advanced pharmaceutical intermediates and fine chemicals, particularly due to the structural complexity and sensitivity of the amino functional group. Patent CN103420860A introduces a groundbreaking methodology that leverages copper-catalyzed decarboxylation coupling to achieve high-yield and high-purity production of these valuable compounds. This technical breakthrough addresses the longstanding limitations of traditional synthetic routes by utilizing isatoic anhydride derivatives and organic cyclic borate compounds as key starting materials. The innovation lies in the specific selection of copper source compounds and ligands that facilitate the reaction under relatively mild conditions, thereby preserving the integrity of sensitive functional groups while ensuring robust conversion rates. For R&D directors and procurement managers in the global chemical industry, this patent offers a viable pathway to optimize manufacturing processes, reduce reliance on precious metals, and enhance the overall economic feasibility of producing complex aromatic esters for drug discovery and agrochemical applications.
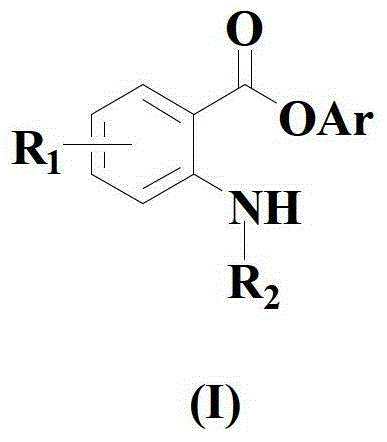
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of amino-substituted aromatic ester compounds has relied heavily on classical esterification techniques such as the Fischer esterification or transesterification reactions, which often necessitate harsh reaction environments involving strong acids or bases. These aggressive conditions can severely limit the scope of compatible functional groups, leading to decomposition of sensitive moieties or unwanted side reactions that complicate downstream purification. Furthermore, alternative approaches like the Baeyer-Villiger oxidation reaction suffer from issues related to regioselectivity, where the migration ability of substituents connected to the carbonyl group dictates the product distribution, often resulting in narrow-spectrum mixtures with low area selectivity. Previous attempts to utilize palladium catalysis, while effective in some contexts, introduce significant cost burdens due to the high price of precious metal catalysts and the requirement for specialized phosphine ligands that may not be readily available on a commercial scale. These inherent drawbacks in conventional methodologies have created a pressing need for a more efficient, cost-effective, and environmentally benign synthetic strategy that can meet the rigorous demands of modern industrial production.
The Novel Approach
The novel approach disclosed in the patent fundamentally shifts the paradigm by employing a copper-catalyzed decarboxylative coupling mechanism that bypasses the need for harsh acidic or alkaline conditions entirely. By reacting isatoic anhydride derivatives with organic cyclic borate compounds in the presence of a specific copper source compound and a 1,10-phenanthroline ligand, the method achieves a direct and efficient construction of the amino-substituted arylate skeleton. This strategy not only eliminates the use of expensive palladium catalysts but also demonstrates exceptional tolerance to various substituents, including halogens, nitro groups, and alkyl chains, without compromising yield or purity. The reaction proceeds smoothly in common organic solvents such as tetrahydrofuran, acetone, or toluene, and operates effectively under air or oxygen atmospheres, simplifying the operational requirements for large-scale manufacturing. This new compounding way provides a robust industrial value proposition by significantly reducing raw material costs and streamlining the purification process, making it an attractive option for reliable pharmaceutical intermediates supplier seeking to enhance their production capabilities.
Mechanistic Insights into Copper-Catalyzed Decarboxylative Coupling
The core of this synthetic innovation lies in the precise interaction between the copper source compound catalyst and the 1,10-phenanthroline ligand, which together create a highly active catalytic system capable of facilitating the decarboxylation of isatoic anhydride derivatives. The copper catalyst, specifically selected from structures such as those shown in formula (a) or (b) within the patent data, acts as the central metal center that coordinates with the organic cyclic borate compound to enable the transfer of the aryl group. This coordination is critical for activating the carbon-carbon bond cleavage required for decarboxylation, a step that is often energetically demanding in the absence of a suitable catalyst. The presence of the 1,10-phenanthroline ligand further stabilizes the copper species and enhances its electronic properties, ensuring that the catalytic cycle proceeds with high turnover numbers and minimal deactivation. This mechanistic understanding is vital for R&D teams aiming to replicate or optimize the process, as it highlights the importance of ligand selection and catalyst loading in achieving the reported high yields and purity levels.
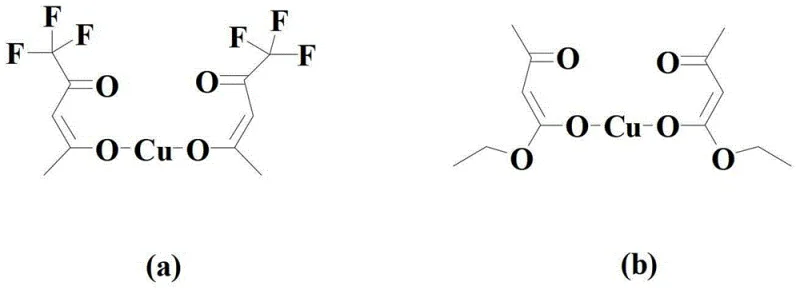
In terms of impurity control, the mechanism inherently favors the formation of the desired amino-substituted arylate product by minimizing competing side reactions that are common in traditional esterification methods. The decarboxylative coupling pathway avoids the formation of carboxylic acid by-products that typically require extensive washing or neutralization steps, thereby simplifying the workup procedure. Additionally, the mild reaction temperatures ranging from 40°C to 80°C prevent thermal degradation of the amino group, which is prone to oxidation or polymerization under more vigorous conditions. The patent data indicates that the reaction can be monitored via TLC or liquid chromatography to determine the optimal endpoint, ensuring that the conversion is complete before proceeding to purification. This level of control over the reaction trajectory allows for the production of high-purity intermediates that meet stringent quality specifications required for downstream pharmaceutical synthesis, reducing the risk of batch failures and ensuring consistent supply chain performance.
How to Synthesize Amino-Substituted Arylate Efficiently
To implement this synthesis effectively, one must adhere to the specific molar ratios and reaction conditions outlined in the patent to ensure optimal performance and reproducibility. The process begins with the preparation of a dried and clean reactor where the solvent, typically THF or acetone, is added followed by the sequential introduction of the isatoic anhydride derivative and the organic cyclic borate compound. The precise stoichiometry is crucial, with a preferred molar ratio of the isatoic anhydride to the borate compound ranging from 1:1 to 1:3, and the copper catalyst loading maintained between 0.05 to 0.2 equivalents relative to the substrate. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction system by mixing isatoic anhydride derivatives and organic cyclic borate compounds in a suitable organic solvent such as THF or acetone.
- Add the specific copper source compound catalyst and 1,10-phenanthroline ligand to the mixture under air or oxygen atmosphere.
- Heat the reaction mixture to 40-80°C for 15-30 hours, then purify the resulting product via column chromatography to obtain high-purity amino-substituted arylate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this copper-catalyzed methodology offers substantial advantages for procurement and supply chain teams by addressing key pain points related to cost, availability, and scalability. The elimination of precious metal palladium from the catalytic system translates directly into significant cost savings, as copper is abundant and considerably cheaper than noble metals, reducing the overall raw material expenditure for large-scale production runs. Furthermore, the use of readily available ligands like 1,10-phenanthroline ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialized phosphine ligands, enhancing the reliability of raw material sourcing. The mild reaction conditions also contribute to lower energy consumption and reduced safety risks, as the process does not require extreme temperatures or pressures, making it easier to implement in existing manufacturing facilities without major capital investment in new equipment.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with cost-effective copper source compounds drastically lowers the direct material costs associated with the synthesis of amino-substituted arylates. This shift allows manufacturers to achieve substantial cost savings without compromising on the quality or yield of the final product, making the process economically viable for high-volume production. Additionally, the simplified purification process reduces the consumption of solvents and chromatography materials, further contributing to the overall reduction in manufacturing expenses and improving the profit margin for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By utilizing common and commercially available reagents such as isatoic anhydride derivatives and organic cyclic borate compounds, the method minimizes the risk of supply disruptions caused by scarce or specialized raw materials. The robustness of the copper catalytic system ensures consistent performance across different batches, reducing the likelihood of production delays due to catalyst failure or variability. This reliability is crucial for maintaining continuous supply to downstream customers, particularly in the pharmaceutical sector where timely delivery of high-purity intermediates is essential for meeting regulatory deadlines and market demands.
- Scalability and Environmental Compliance: The reaction conditions, which operate at moderate temperatures and under air or oxygen atmosphere, are inherently safer and easier to scale up compared to processes requiring inert gases or high-pressure equipment. The reduced use of hazardous reagents and the generation of fewer by-products align with green chemistry principles, facilitating compliance with increasingly stringent environmental regulations. This environmental compatibility not only reduces waste disposal costs but also enhances the corporate sustainability profile, making the manufacturing process more attractive to eco-conscious partners and investors in the global chemical market.
Frequently Asked Questions (FAQ)
The following questions and answers are derived directly from the technical details and experimental data provided in the patent documentation to address common inquiries regarding the feasibility and application of this synthesis method. These insights are intended to clarify the operational parameters and potential benefits for stakeholders considering the adoption of this technology for their production needs. Understanding these specifics is key to evaluating the technical fit for your specific manufacturing requirements.
Q: What are the advantages of using copper catalysts over palladium for this synthesis?
A: Copper catalysts are significantly more cost-effective than precious metal palladium catalysts. The patent demonstrates that specific copper source compounds combined with 1,10-phenanthroline achieve high yields without the need for expensive noble metals, reducing overall manufacturing costs.
Q: What are the typical reaction conditions for this decarboxylation coupling?
A: The reaction typically proceeds in organic solvents like THF or acetone at moderate temperatures ranging from 40°C to 80°C. The process operates under air or oxygen atmosphere and requires reaction times between 15 to 30 hours depending on the specific substrates used.
Q: How does this method improve impurity control compared to traditional esterification?
A: Unlike traditional Fischer esterification which requires strong acidic or alkaline conditions leading to side reactions, this copper-catalyzed method offers better regioselectivity. The decarboxylative coupling mechanism minimizes the formation of by-products, resulting in higher purity products as confirmed by HPLC analysis in the patent examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amino-Substituted Arylate Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis route for the production of high-value pharmaceutical intermediates and fine chemicals. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this can be seamlessly translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards, guaranteeing that the amino-substituted arylate compounds we supply meet the exacting requirements of global drug manufacturers.
We invite you to collaborate with us to optimize your supply chain and leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this method can reduce your overall production costs. Please contact us to request specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver reliable high-purity amino-substituted arylate with the speed and precision your business demands.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
