Advanced Quinoline Intermediate Synthesis for Oncology Drug Manufacturing
Advanced Quinoline Intermediate Synthesis for Oncology Drug Manufacturing
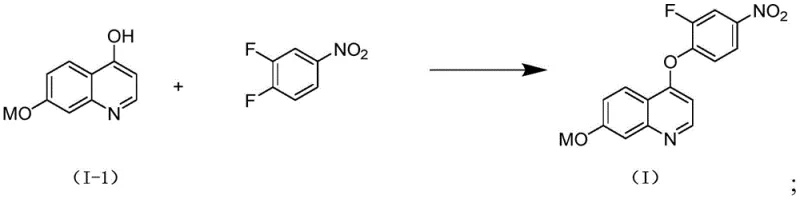
The pharmaceutical industry is constantly seeking more efficient and safer pathways for synthesizing complex heterocyclic scaffolds, particularly those serving as key intermediates for oncology therapeutics. Patent CN110845406A introduces a groundbreaking preparation method for quinoline compounds, specifically targeting the synthesis of 4-(2-fluoro-4-nitrophenyl)-7-hydroxyquinoline derivatives. These structures are pivotal precursors for high-value tyrosine kinase inhibitors such as Nigatinib tosylate, Cabozantinib malate, and Anlotinib hydrochloride, which are essential in modern targeted cancer therapies. The disclosed technology addresses critical bottlenecks in traditional manufacturing by eliminating the use of hazardous phosphorus oxychloride, thereby streamlining the production workflow and enhancing overall process safety. This innovation represents a significant leap forward for reliable pharmaceutical intermediate suppliers aiming to optimize their supply chains for anti-tumor drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted quinoline derivatives required the activation of hydroxyl groups using phosphorus oxychloride (POCl3) to facilitate subsequent nucleophilic substitution reactions. This conventional approach presents severe drawbacks, including the handling of highly corrosive reagents that demand specialized equipment and rigorous safety protocols to prevent accidents. Furthermore, the use of POCl3 generates substantial amounts of acidic wastewater and phosphorus-containing waste, creating significant environmental compliance burdens and escalating waste treatment costs for manufacturers. The post-reaction workup often involves complex neutralization and extraction steps, which can lead to product loss and lower overall yields. Additionally, the harsh reaction conditions associated with chlorinating agents can promote side reactions, resulting in difficult-to-remove impurities that compromise the purity profile required for pharmaceutical-grade intermediates.
The Novel Approach
In stark contrast, the novel methodology described in the patent leverages the intrinsic electronic properties of the reactants to drive the reaction without aggressive activating agents. By utilizing 3,4-difluoronitrobenzene, the process capitalizes on the strong electron-withdrawing nature of both the nitro and fluorine substituents to activate the aromatic ring for direct nucleophilic aromatic substitution. This allows the hydroxyl group of the quinoline precursor to attack the fluorine-bearing carbon directly under mild basic conditions, completely bypassing the need for phosphorus oxychloride. This strategic shift not only simplifies the reaction setup but also drastically reduces the generation of hazardous byproducts, aligning with green chemistry principles. The result is a cleaner reaction profile that facilitates easier purification through simple crystallization or filtration, significantly improving the operational efficiency for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Nucleophilic Aromatic Substitution and Cyclization
The success of this synthetic route relies heavily on the precise manipulation of electronic effects within the molecular framework. In the final coupling step, the presence of the nitro group at the para-position relative to the leaving fluorine atom creates a powerful electron-deficient center on the benzene ring. This effect is further amplified by the additional fluorine atom at the meta-position, which enhances the electrophilicity of the system through inductive withdrawal. When the quinoline phenoxide anion, generated in situ by the action of a base like DIPEA, approaches this activated ring, the energy barrier for the formation of the Meisenheimer complex is significantly lowered. This mechanistic advantage ensures that the substitution occurs selectively at the para-position, minimizing the formation of regioisomers and other structural impurities. Consequently, the reaction proceeds with high fidelity, yielding the desired ether-linked product with exceptional purity levels suitable for downstream drug synthesis.
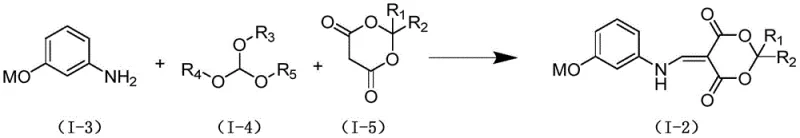
Prior to this final coupling, the construction of the quinoline core itself involves a sophisticated cascade of condensation and cyclization events. The initial step involves the reaction of 3-benzyloxyaniline with triethyl orthoformate and isopropylidene malonate. This multicomponent condensation forms an enamine intermediate that is primed for ring closure. The subsequent thermal cyclization step, conducted at elevated temperatures in a high-boiling solvent like o-dichlorobenzene, drives the intramolecular condensation to form the pyridine ring of the quinoline system. This thermal process is robust and avoids the use of sensitive catalysts, relying instead on thermodynamic control to achieve the stable aromatic heterocycle. The combination of these steps demonstrates a deep understanding of physical organic chemistry, ensuring that each transformation is optimized for yield and scalability.
How to Synthesize 4-(2-fluoro-4-nitrophenyl)-7-hydroxyquinoline Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing this valuable intermediate with high efficiency and reproducibility. The process begins with the condensation of readily available starting materials in ethanol, followed by a thermal cyclization to build the core scaffold, and concludes with the key etherification step. Each stage is designed to minimize operational complexity, utilizing standard laboratory and plant equipment without the need for exotic catalysts or extreme pressure conditions. The detailed standardized synthesis steps below provide a comprehensive guide for technical teams looking to implement this route, ensuring consistent quality and performance across different production batches.
- Condense 3-benzyloxyaniline with triethyl orthoformate and isopropylidene malonate in ethanol under reflux to form the enamine intermediate.
- Perform thermal cyclization of the intermediate in o-dichlorobenzene at 180°C to construct the quinoline core structure.
- React the resulting hydroxyquinoline with 3,4-difluoronitrobenzene using DIPEA base in DMF at 85°C to achieve the final substitution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible benefits that extend beyond mere chemical elegance. The elimination of phosphorus oxychloride removes a major bottleneck related to hazardous material logistics, storage, and disposal, which are often costly and regulatory-intensive. By switching to safer reagents like 3,4-difluoronitrobenzene and organic bases, facilities can reduce their environmental footprint and lower the total cost of ownership associated with safety compliance and waste management. Furthermore, the simplified workup procedures, which rely on filtration and washing rather than column chromatography, significantly reduce solvent consumption and processing time. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of phosphorus oxychloride from the process eliminates the need for expensive corrosion-resistant reactors and complex neutralization systems, leading to substantial capital and operational expenditure savings. Additionally, the high selectivity of the reaction reduces the formation of byproducts, which minimizes the loss of valuable starting materials and lowers the cost per kilogram of the final active intermediate. The ability to use common solvents like ethanol and DMF, which are easily recoverable and recyclable, further enhances the economic viability of the process on an industrial scale.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including 3-benzyloxyaniline and difluoronitrobenzene, are commodity chemicals with stable global supply chains, reducing the risk of production delays due to material shortages. The robustness of the reaction conditions, which tolerate minor variations in temperature and stoichiometry without compromising yield, ensures consistent output even in large-scale manufacturing environments. This reliability is crucial for maintaining continuous supply to downstream drug manufacturers who depend on timely delivery of critical oncology intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of reactions in multi-liter setups without the need for specialized high-pressure equipment. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, making it easier for manufacturers to obtain necessary permits and maintain good standing with regulatory bodies. The straightforward isolation of the product through crystallization or filtration simplifies the scale-up process, allowing for rapid transition from pilot plant to full commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinoline synthesis route. These answers are derived directly from the experimental data and technical disclosures within the patent, providing clarity on reaction mechanisms, safety profiles, and scalability potential. Understanding these details is essential for technical teams evaluating the feasibility of adopting this new methodology for their specific manufacturing needs.
Q: Why is avoiding phosphorus oxychloride critical in quinoline synthesis?
A: Phosphorus oxychloride (POCl3) is highly corrosive and generates significant hazardous waste, complicating safety protocols and increasing disposal costs. The new method eliminates this reagent entirely.
Q: What drives the high yield in the final substitution step?
A: The presence of both nitro and fluorine atoms on the benzene ring creates strong electron-withdrawing effects, significantly activating the ring for nucleophilic attack by the quinoline hydroxyl group.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes common solvents like ethanol and DMF, avoids chromatography through simple crystallization, and operates at manageable temperatures, ensuring excellent scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(2-fluoro-4-nitrophenyl)-7-hydroxyquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthesis routes in the development of life-saving oncology medications. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex molecules like quinoline intermediates are delivered with the highest standards of quality. We utilize stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting requirements of global pharmaceutical partners. Our commitment to process innovation allows us to offer competitive solutions that balance cost, speed, and safety effectively.
We invite you to collaborate with us to leverage this advanced synthesis technology for your drug development programs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your supply chain goals and accelerate your time to market.
