Scalable Synthesis of Chiral Gamma-Butyrolactone Intermediates for Brivaracetam Production
The pharmaceutical industry continuously seeks robust and scalable pathways for synthesizing complex chiral intermediates, particularly for antiepileptic drugs like Brivaracetam. Patent CN109553595B, published on May 28, 2021, introduces a groundbreaking preparation method for (R)-4-propyl-4,5-dihydrofuran-2(3H)-one, a critical chiral gamma-butyrolactone intermediate. This technology addresses the longstanding challenges of high costs, severe reaction conditions, and safety hazards associated with prior art synthesis routes. By leveraging cheap raw materials such as chiral 1,2-pentanediol and employing mild weak-base conditions for key coupling reactions, this invention offers a superior alternative for industrial manufacturing. The process eliminates the need for expensive chiral auxiliaries or dangerous diazotization steps, positioning it as a highly attractive option for reliable pharmaceutical intermediate supplier networks aiming to optimize their supply chains for next-generation neurological therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral gamma-butyrolactones has been plagued by inefficiencies that hinder large-scale production. Early approaches, such as the one reported by Benoit M., involved reacting 2(5H)-furanone with propylmagnesium bromide. As illustrated in the reaction scheme below, this method inevitably generates a racemate, necessitating a costly and yield-limiting chiral resolution step to isolate the desired enantiomer. Furthermore, other existing patents disclose routes relying on ethyl-metal reagents, which introduce severe potential safety hazards due to their pyrophoric nature and sensitivity to moisture. Additionally, methods utilizing Evans chiral auxiliaries suffer from high reagent costs and complicated multi-step operations, while routes starting from unnatural amino acids require dangerous diazotization reactions and harsh strong-base conditions like sodium hydride, making them unsuitable for safe, green industrial application.
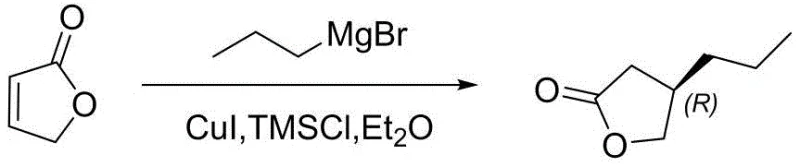
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in CN109553595B utilizes a strategic disconnection that begins with readily available chiral 1,2-pentanediol derivatives. The core innovation lies in the construction of the carbon skeleton via a butt-joint reaction with Meldrum's acid, which can be executed under remarkably mild conditions using weak bases such as potassium carbonate or triethylamine. This shift from strong, hazardous bases to mild inorganic or organic bases significantly lowers the barrier for commercial scale-up of complex pharmaceutical intermediates. The subsequent transformation involves a controlled decarboxylation and lactonization sequence that preserves the chiral integrity established at the beginning of the synthesis. This streamlined workflow not only simplifies the operational procedure but also drastically reduces the environmental footprint and safety risks associated with traditional strong-base chemistry, offering a clear path toward cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Copper-Catalyzed Decarboxylation and Lactonization
The heart of this synthetic strategy involves the conversion of a protected Meldrum's acid derivative (Formula III) into the final lactone (Formula I) through a carefully orchestrated two-step sequence. The first critical stage is the decarboxylation reaction, which transforms the six-membered Meldrum's acid ring into a linear ester intermediate (Formula IV). This step is typically catalyzed by cuprous oxide or copper powder in high-boiling solvents like pyridine or DMF under reflux conditions. The copper catalyst facilitates the loss of carbon dioxide and acetone, effectively unmasking the reactive methylene group while maintaining the stereochemical configuration at the adjacent chiral center. Following this, the second stage involves the removal of the hydroxyl protecting group (R1), which triggers an intramolecular nucleophilic attack by the liberated hydroxyl group onto the ester carbonyl. Depending on the nature of the protecting group—whether it is a benzyl ether, silyl ether, or benzoate—specific deprotection reagents such as hydrogen with palladium on carbon, tetrabutylammonium fluoride, or aqueous alkali are employed to drive the reaction to completion, yielding the target chiral gamma-butyrolactone with high purity.
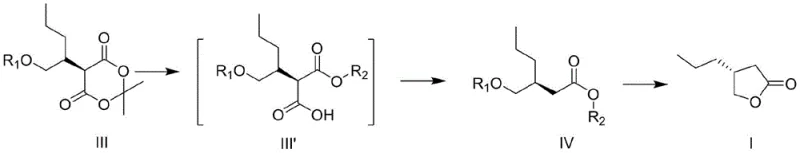
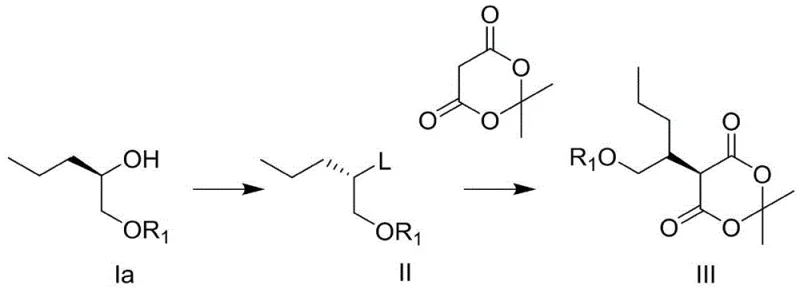
Beyond the final cyclization, the preparation of the key intermediate (Formula III) demonstrates remarkable flexibility in stereochemical control. The patent outlines multiple pathways to access this precursor, allowing manufacturers to choose starting materials based on availability and cost. For instance, starting from a chiral alcohol with inverted configuration (Formula Ia), a halogenation or Mitsunobu reaction converts the hydroxyl into a leaving group (Formula II), which then undergoes nucleophilic substitution with Meldrum's acid. Alternatively, starting from a retained configuration alcohol (Formula Ib), a direct Mitsunobu coupling or a two-step activation-substitution sequence can be employed. This mechanistic versatility ensures that the process can be adapted to various supply chain constraints without compromising the optical purity of the final product. The use of Meldrum's acid as a nucleophile is particularly advantageous because its high acidity allows for alkylation under weak basic conditions, avoiding the side reactions and decomposition often seen with more aggressive enolates.
How to Synthesize (R)-4-propyl-4,5-dihydrofuran-2(3H)-one Efficiently
The synthesis of this high-value chiral building block is designed for operational simplicity and scalability, making it ideal for contract development and manufacturing organizations. The process begins with the preparation of the Meldrum's acid derivative, followed by the thermal decarboxylation and final cyclization. Detailed standard operating procedures regarding specific molar ratios, solvent volumes, and work-up protocols are essential for reproducibility.
- Perform decarboxylation on the Meldrum's acid intermediate (Formula III) using copper catalyst in refluxing solvent to obtain the ester intermediate (Formula IV).
- Execute deprotection of the hydroxyl group in Formula IV using specific reagents like Pd/C hydrogenation or TBAF depending on the protecting group.
- Facilitate spontaneous or acid-catalyzed lactonization to cyclize the linear chain into the final chiral gamma-butyrolactone (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial cost savings achieved by replacing expensive chiral auxiliaries and unnatural amino acids with commodity chemicals like chiral pentanediol. This shift in raw material sourcing significantly stabilizes the supply chain against price volatility often associated with specialized fine chemicals. Moreover, the elimination of hazardous reagents such as ethyl-metal compounds and strong bases like sodium hydride reduces the need for specialized containment infrastructure and expensive waste treatment protocols. This translates directly into lower overhead costs and a safer working environment, which is a critical metric for modern ESG-compliant manufacturing facilities. The simplified operational steps also mean shorter batch cycles, enhancing the overall throughput and reliability of the production line.
- Cost Reduction in Manufacturing: The replacement of costly Evans auxiliaries and resolution steps with a direct chiral pool synthesis significantly lowers the bill of materials. By utilizing weak bases like potassium carbonate instead of expensive and hazardous strong bases, the process reduces reagent costs and minimizes the need for rigorous anhydrous conditions, leading to substantial cost savings in utility and equipment maintenance.
- Enhanced Supply Chain Reliability: Starting from commercially available chiral 1,2-pentanediol ensures a stable and continuous supply of raw materials, unlike routes dependent on custom-synthesized unnatural amino acids. The robustness of the weak-base coupling reaction reduces the risk of batch failures due to moisture sensitivity, thereby improving delivery consistency and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The avoidance of pyrophoric reagents and dangerous diazotization steps makes this process inherently safer for large-scale production. The use of common solvents and mild conditions simplifies waste stream management, facilitating easier compliance with environmental regulations and enabling seamless commercial scale-up of complex intermediates without major capital investment in safety infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, highlighting the practical benefits for industrial partners.
Q: Why is the new method safer than previous ethyl-metal reagent routes?
A: Previous routes utilized ethyl-metal reagents which pose significant safety hazards due to their pyrophoric nature. The new method replaces these with stable Meldrum's acid derivatives and weak bases like potassium carbonate, drastically reducing operational risk.
Q: How does this process avoid the high costs associated with chiral resolution?
A: Unlike early methods that generated racemates requiring expensive resolution, this patent starts from commercially available chiral 1,2-pentanediol. The stereochemistry is preserved or inverted predictably during the substitution step, ensuring high optical purity without resolution steps.
Q: What are the advantages of using weak bases in the coupling reaction?
A: Traditional methods often required strong bases like sodium hydride or sodium alkoxides, which demand strict anhydrous conditions and pose safety risks. This invention enables the coupling reaction using mild weak bases such as potassium carbonate or triethylamine, simplifying equipment requirements and improving safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-4-propyl-4,5-dihydrofuran-2(3H)-one Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising chemistry described in CN109553595B can be seamlessly integrated into your supply chain. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (R)-4-propyl-4,5-dihydrofuran-2(3H)-one meets the exacting standards required for API synthesis. Our commitment to quality and safety aligns perfectly with the advantages of this new mild-base methodology, offering you a secure source for this critical Brivaracetam intermediate.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your production costs and timelines. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic impact of switching to this safer, more efficient process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements, ensuring a smooth and successful partnership for your neurological drug development projects.
