Advanced Alkaline Dehydration Strategy for Commercial Scale-up of Complex Olefin Intermediates
Advanced Alkaline Dehydration Strategy for Commercial Scale-up of Complex Olefin Intermediates
The landscape of organic synthesis is constantly evolving, driven by the need for more sustainable, efficient, and selective methodologies for constructing carbon-carbon double bonds. A pivotal advancement in this domain is detailed in patent CN112851456A, which discloses a novel method for preparing olefin compounds under alkaline conditions. This technology represents a significant departure from conventional acid-catalyzed dehydration routes, offering a robust pathway for transforming readily available long-chain alcohols into valuable olefin intermediates. By utilizing sulfuryl fluoride (SO2F2) as an activating agent in the presence of a base catalyst, this process achieves high conversion rates under remarkably mild thermal conditions. For R&D directors and process chemists, this innovation addresses critical challenges regarding functional group tolerance and reaction severity, positioning it as a key technology for the reliable olefin intermediate supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-carbon double bonds from alcohol precursors has relied heavily on acid-catalyzed dehydration or coupling reactions involving stoichiometric metal reagents. Traditional acid-catalyzed methods often necessitate extremely harsh reaction environments, typically requiring temperatures higher than 150 °C to drive the elimination of water. Such severe thermal conditions are not only energy-intensive but also pose significant risks to the structural integrity of complex molecules. Furthermore, the acidic nature of these protocols is incompatible with a wide array of acid-sensitive functional groups, such as acetals, ketals, and epoxies, which are frequently encountered in advanced pharmaceutical intermediates. Consequently, the narrow substrate scope and the potential for side reactions or decomposition limit the utility of these classical approaches in the synthesis of sophisticated molecular architectures required by modern drug discovery programs.
The Novel Approach
In stark contrast to these legacy methods, the alkaline condition-based synthesis described in the patent offers a transformative solution by shifting the mechanistic paradigm from acid catalysis to base-promoted elimination. This innovative route leverages the unique reactivity of sulfuryl fluoride to activate the hydroxyl group of the alcohol, facilitating a smooth elimination process at significantly lower temperatures, optimally around 60 °C. The mildness of this protocol ensures that sensitive moieties within the molecular framework remain intact, thereby vastly expanding the range of applicable substrates. Moreover, the use of inexpensive long-chain alcohols as starting materials, combined with the operational simplicity of the one-pot procedure, underscores the economic and practical viability of this method for industrial applications. This approach effectively bypasses the limitations of high-temperature thermal dehydration, providing a green and efficient synthetic path.
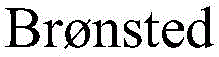
The general transformation is elegantly depicted in the reaction scheme, where diverse alcohol substrates bearing phenyl, heterocyclic, or alkyl chains are seamlessly converted into their corresponding terminal olefins. This versatility is crucial for manufacturers aiming to produce high-purity olefin intermediates for diverse applications, ranging from active pharmaceutical ingredients to agrochemical precursors. The ability to tolerate various substituents without compromising yield or purity makes this technology a cornerstone for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Base-Promoted Fluorosulfonation and Elimination
Understanding the underlying mechanism is essential for optimizing this process for commercial scale-up of complex olefin intermediates. The reaction proceeds through a distinct two-stage sequence initiated by the nucleophilic attack of the alcohol's hydroxyl oxygen on the sulfur center of the sulfuryl fluoride molecule. This interaction generates a reactive fluorosulfonate intermediate in situ, effectively converting the poor leaving group (hydroxyl) into an excellent leaving group (fluorosulfonate). Subsequently, the presence of a strong base catalyst, such as potassium tert-butoxide, facilitates the abstraction of a beta-proton. This deprotonation event triggers an E2-type elimination mechanism, resulting in the expulsion of fluorosulfonic acid (HSO3F) and the formation of the desired carbon-carbon double bond. This mechanistic pathway avoids the carbocation intermediates typical of acid-catalyzed reactions, thereby minimizing rearrangement side products and ensuring high regioselectivity.
The control of impurities is inherently superior in this alkaline system due to the absence of strong acids that could promote polymerization or skeletal rearrangements. The reaction conditions are sufficiently mild to prevent the degradation of the olefin product, which can be susceptible to acid-catalyzed hydration or oligomerization under traditional conditions. Furthermore, the byproduct, fluorosulfonic acid, is neutralized by the excess base present in the reaction mixture, simplifying the downstream workup and purification processes. This clean reaction profile is particularly advantageous for producing reducing lead time for high-purity olefin intermediates, as it reduces the burden on purification teams and minimizes the loss of valuable material during chromatographic separation.
How to Synthesize Olefin Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and gas handling, although the overall procedure remains straightforward. The standard protocol involves dissolving the long-chain alcohol substrate in a polar aprotic solvent, with dimethyl sulfoxide (DMSO) being the preferred medium due to its ability to solubilize both the organic substrate and the inorganic base. A molar excess of the base catalyst, typically potassium tert-butoxide, is added to ensure complete deprotonation and drive the equilibrium towards product formation. The critical step involves the introduction of sulfuryl fluoride gas, which can be conveniently supplied via a balloon or gas line, allowing for precise control over the activation step without the need for specialized high-pressure equipment.
- Dissolve the long-chain alcohol substrate in a polar aprotic solvent such as DMSO and add a strong base catalyst like potassium tert-butoxide.
- Introduce sulfuryl fluoride (SO2F2) gas into the reaction mixture at a controlled temperature, typically around 60 °C, to activate the hydroxyl group.
- Stir the reaction for 12-15 hours to ensure complete conversion, followed by aqueous workup and silica gel chromatography to isolate the high-purity olefin product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers compelling advantages that directly impact the bottom line and operational resilience. The shift towards this alkaline methodology addresses several pain points associated with traditional olefin synthesis, including high energy consumption, expensive reagents, and complex waste management. By adopting this route, organizations can achieve substantial cost savings and enhance their supply chain reliability through the use of commoditized raw materials and simplified processing requirements.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the utilization of long-chain alcohols, which are inexpensive and widely available commodity chemicals, as opposed to specialized organometallic reagents or activated halides. The elimination of stoichiometric metal waste and the reduction in energy costs due to lower operating temperatures (60 °C vs >150 °C) contribute to a significantly leaner cost structure. Additionally, the high yields reported in the patent examples, often exceeding 85-90%, minimize raw material waste and maximize the output per batch, further driving down the unit cost of production.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain security by reducing dependency on scarce or volatile reagents. The reagents used, such as sulfuryl fluoride and common bases like potassium tert-butoxide, are stable and easily sourced from multiple global suppliers, mitigating the risk of supply disruptions. The mild reaction conditions also reduce the wear and tear on reactor vessels and ancillary equipment, leading to lower maintenance costs and higher equipment availability, which is critical for maintaining consistent delivery schedules to downstream customers.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the simplicity of the operation, which does not require exotic catalysts or extreme pressures. The avoidance of heavy metals and strong mineral acids aligns with increasingly stringent environmental regulations, simplifying waste treatment and disposal protocols. The generation of fewer hazardous byproducts and the potential for solvent recovery make this method an environmentally responsible choice, supporting corporate sustainability goals while ensuring regulatory compliance in major manufacturing hubs.
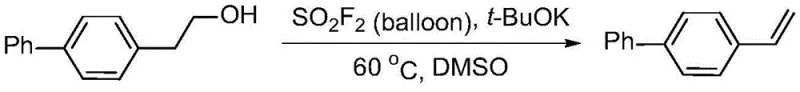
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkaline olefin synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for stakeholders evaluating this process for adoption.
Q: What are the primary advantages of this alkaline method over traditional acid-catalyzed dehydration?
A: Unlike traditional methods requiring harsh acidic conditions and temperatures exceeding 150 °C, this alkaline approach operates at mild temperatures (e.g., 60 °C) and effectively tolerates acid-sensitive functional groups such as acetals, ketals, and epoxies, preventing substrate degradation.
Q: Which substrates are compatible with this sulfuryl fluoride-mediated elimination?
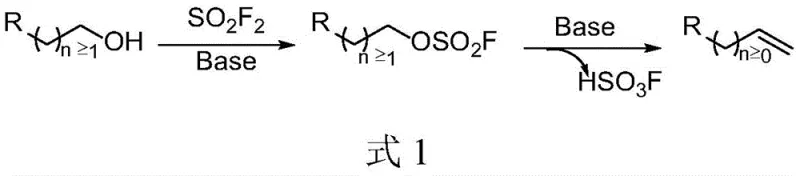
A: The method demonstrates broad substrate scope, successfully converting various long-chain alcohols containing phenyl, substituted phenyl, heterocyclic aryl, fused-ring aryl, and alkyl groups into their corresponding olefins with high efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes inexpensive and widely available long-chain alcohols as raw materials, employs simple operational procedures with low equipment requirements, and achieves high yields, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olefin Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this alkaline dehydration technology for the production of high-value olefin intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of olefin compound delivered meets the exacting standards required for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to leverage this efficient synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this method can optimize your manufacturing budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with reliable, high-quality chemical solutions.
