Advanced Iridium-Carbene Complexes for Efficient Pharmaceutical Intermediate Manufacturing
Introduction to Patent CN111548372A and Technological Breakthroughs
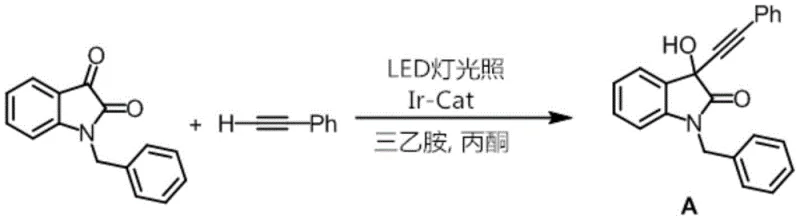
The landscape of fine chemical synthesis is undergoing a paradigm shift driven by the urgent need for sustainable and energy-efficient methodologies, a transition vividly exemplified by the innovations disclosed in Chinese Patent CN111548372A. This pivotal intellectual property introduces a novel metal iridium-carbene complex, specifically characterized by the molecular formula C34H36Cl2IrN6PF6, which serves as a highly efficient photocatalyst for the addition of terminal alkynes to isatin derivatives. Traditionally, such transformations have been plagued by severe operational constraints, necessitating the use of hazardous strong bases and cryogenic conditions that escalate both safety risks and production costs. However, the technology described in this patent leverages the unique electronic properties of N-heterocyclic carbene (NHC) ligands coordinated to an iridium(III) center to facilitate these reactions under mild, visible-light irradiation at ambient temperature. For R&D directors and procurement strategists alike, this represents a critical advancement towards greener manufacturing processes that do not compromise on yield or purity, positioning this catalyst as a cornerstone for next-generation pharmaceutical intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the nucleophilic addition of terminal alkynes to carbonyl compounds, such as isatin derivatives, has relied heavily on the generation of highly reactive acetylide anions using stoichiometric amounts of strong bases like butyllithium or Grignard reagents. This conventional approach imposes a formidable burden on industrial infrastructure, as it mandates strictly anhydrous and anaerobic environments to prevent the rapid decomposition of the organometallic intermediates by moisture or oxygen. Furthermore, the requirement for ultra-low temperature conditions, often reaching cryogenic levels below -70°C, necessitates specialized refrigeration equipment and substantial energy consumption, creating a significant bottleneck for commercial scale-up of complex organic transformations. The inherent instability of these reagents also leads to challenging impurity profiles and safety hazards associated with the handling of pyrophoric materials, making the supply chain vulnerable to disruptions and increasing the overall cost of goods sold for high-value intermediates.
The Novel Approach
In stark contrast to these archaic methodologies, the novel approach detailed in the patent utilizes a robust iridium-carbene photocatalyst that activates terminal alkynes through a completely different mechanistic pathway involving visible light energy. By employing simple household LED lamps as the energy source and weak organic bases like triethylamine, the reaction proceeds smoothly at room temperature, effectively eliminating the need for expensive cryogenic setups and rigorous inert atmosphere techniques. This methodological shift not only drastically simplifies the operational workflow but also enhances the safety profile of the manufacturing process by removing pyrophoric reagents from the equation. The versatility of this system is demonstrated in the efficient synthesis of propargylic alcohol derivatives, where the catalyst enables high conversion rates with excellent functional group tolerance, thereby offering a reliable pharmaceutical intermediates supplier with a distinct competitive advantage in process efficiency.
Mechanistic Insights into Iridium-NHC Photocatalysis
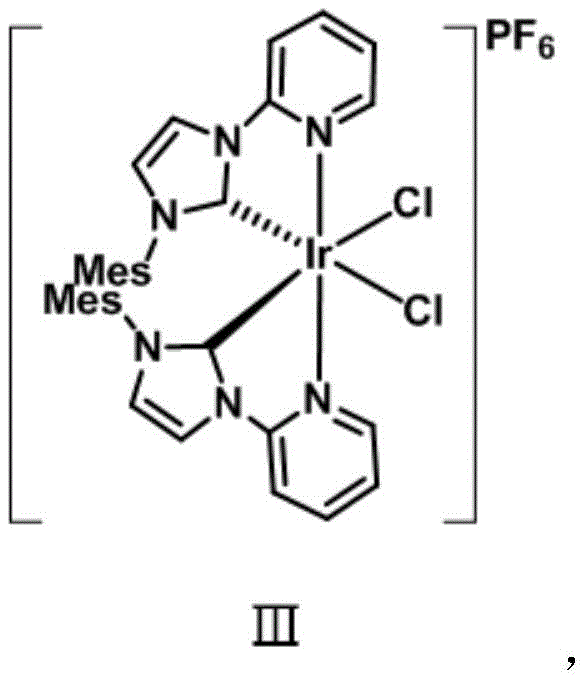
The exceptional performance of this catalyst stems from the sophisticated interplay between the central iridium atom and the bulky, electron-donating N-heterocyclic carbene ligands, which stabilize the metal center while facilitating crucial electron transfer processes. Upon absorption of visible light photons, the iridium(III) complex undergoes excitation to a higher energy state, potentially involving metal-to-ligand charge transfer (MLCT) or ligand-centered transitions that render the metal center more oxidizing or reducing depending on the specific cycle. This photo-excited species is capable of interacting with the terminal alkyne substrate, likely through a mechanism where the iridium center coordinates with the alkyne triple bond, thereby lowering the pKa of the terminal proton and allowing for deprotonation by the mild amine base. The resulting iridium-acetylide intermediate is sufficiently stable to persist under reaction conditions yet reactive enough to attack the electrophilic carbonyl carbon of the isatin derivative, driving the formation of the new carbon-carbon bond with high stereochemical control.
From a quality control perspective, the structural integrity of the catalyst is paramount, as evidenced by the single-crystal X-ray diffraction data which confirms a monoclinic crystal system with a P21/c space group. The precise arrangement of the ligands creates a chiral environment around the metal center, which can be exploited to achieve enantioselective transformations in future iterations, although the current patent focuses on racemic synthesis. The robustness of the Ir-C and Ir-N bonds within the coordination sphere ensures that the catalyst does not leach significant amounts of heavy metal into the final product, a critical factor for meeting stringent purity specifications in pharmaceutical applications. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters such as light intensity and solvent polarity to maximize turnover numbers and minimize the formation of side products, ensuring the delivery of high-purity pharmaceutical intermediates.
How to Synthesize Iridium-Carbene Complex Efficiently
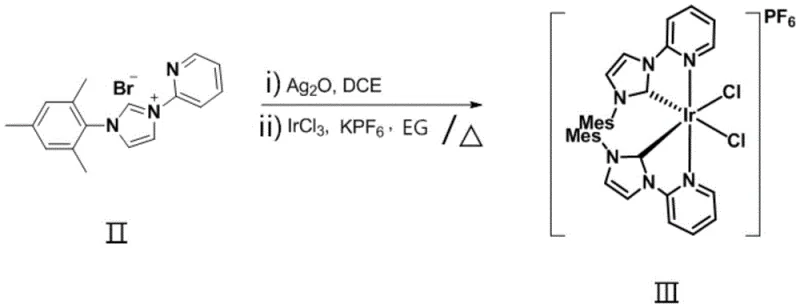
The preparation of this high-performance photocatalyst follows a logical, multi-step synthetic route that begins with the construction of the pyridine-imidazolium salt precursor, followed by transmetallation to silver and finally to iridium. This sequence is designed to maximize yield and purity at each stage, utilizing solvent-free conditions for the initial salt formation to drive the equilibrium towards the product without the need for extensive purification steps. The subsequent conversion to the silver-carbene intermediate serves as a crucial activation step, generating a highly reactive species that readily transfers the carbene ligand to the iridium center under thermal conditions in ethylene glycol. Detailed standard operating procedures for the synthesis, including specific molar ratios, temperature gradients, and workup protocols, are essential for reproducibility and are outlined in the comprehensive guide below.
- Synthesize the pyridine-imidazolium bromide precursor by reacting N-(2,4,6-trimethylphenyl)imidazole with 2-bromopyridine under solvent-free thermal conditions.
- Generate the silver-carbene intermediate by reacting the precursor with silver oxide in a halogenated hydrocarbon solvent at room temperature in the dark.
- Complete the metallation by reacting the silver intermediate with iridium trichloride and potassium hexafluorophosphate in ethylene glycol under heating.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible strategic benefits that extend far beyond the laboratory bench, fundamentally altering the cost structure of producing valuable chemical building blocks. The elimination of cryogenic cooling systems and the replacement of hazardous strong bases with benign amines result in a drastic simplification of the required reactor infrastructure, allowing for the utilization of standard glass-lined or stainless steel vessels without specialized low-temperature jackets. This reduction in capital expenditure (CAPEX) is complemented by a significant decrease in operational expenditure (OPEX), as the energy demand for maintaining sub-zero temperatures is completely removed, and the cost of purchasing and disposing of dangerous reagents is minimized. Furthermore, the use of visible light as a traceless reagent aligns perfectly with global sustainability goals, reducing the carbon footprint of the manufacturing process and enhancing the corporate social responsibility profile of the supply chain.
- Cost Reduction in Manufacturing: The economic implications of switching to this photocatalytic method are profound, primarily driven by the removal of expensive and dangerous reagents like butyllithium which require special handling and storage protocols. By operating at ambient temperature, the process avoids the high energy costs associated with industrial refrigeration, leading to substantial cost savings in utility bills over the lifecycle of the production campaign. Additionally, the simplified workup procedures, which do not require quenching of reactive organometallic species, reduce the consumption of auxiliary chemicals and shorten the batch cycle time, thereby increasing the overall throughput of the manufacturing facility and improving the return on investment for production assets.
- Enhanced Supply Chain Reliability: Reliance on specialized cryogenic reagents often introduces fragility into the supply chain, as these materials have short shelf lives and require cold-chain logistics that are susceptible to disruption. In contrast, the reagents required for this photocatalytic process, such as the iridium complex, terminal alkynes, and common amines, are stable solids or liquids that can be stored under standard warehouse conditions for extended periods. This stability ensures a consistent availability of raw materials, reducing the risk of production stoppages due to supply shortages and enabling more accurate forecasting and inventory management, which is critical for maintaining reducing lead time for high-purity pharmaceutical intermediates in a just-in-time manufacturing environment.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to the Beer-Lambert law limiting light penetration in large vessels, but the advent of flow chemistry and high-efficiency LED arrays has largely overcome this barrier, making this technology highly scalable for industrial production. The process generates minimal hazardous waste compared to traditional methods, as there are no heavy metal salts from quenched organolithiums to dispose of, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. This ease of scale-up and regulatory compliance makes the technology an attractive option for contract development and manufacturing organizations (CDMOs) looking to offer cost reduction in fine chemical manufacturing services to their global clientele.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the implementation and capabilities of this technology, we have compiled a set of answers based on the technical specifications and experimental data provided in the patent documentation. These questions cover critical aspects ranging from reaction conditions and catalyst loading to product isolation and purity standards, providing a quick reference for technical teams evaluating the feasibility of integrating this method into their existing workflows. The responses are grounded in the empirical results observed during the development of the iridium-carbene complex, ensuring that the information provided is both accurate and actionable for decision-makers.
Q: What are the primary advantages of this iridium-carbene photocatalyst over traditional methods?
A: Unlike traditional methods requiring cryogenic temperatures and strong bases like butyllithium, this photocatalyst operates at room temperature using visible light and weak organic bases, significantly improving safety and reducing energy costs.
Q: Is the catalyst sensitive to moisture or oxygen during the reaction?
A: The reaction system demonstrates robust tolerance to moisture, eliminating the need for rigorous anhydrous conditions typically required for organolithium reagents, thereby simplifying operational protocols.
Q: What is the typical yield for the addition reaction using this catalyst?
A: Experimental data indicates that the addition product yield exceeds 82wt%, with specific examples achieving up to 95% yield under optimized LED irradiation conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iridium-Carbene Complex Supplier
As the demand for advanced catalytic solutions continues to grow, NINGBO INNO PHARMCHEM stands ready to support your organization with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with the necessary infrastructure to handle photochemical processes safely and efficiently, ensuring that the transition from laboratory discovery to industrial reality is seamless and robust. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of catalyst or intermediate meets the highest international standards, providing you with the confidence needed to integrate these materials into your critical drug synthesis pipelines without compromising on quality or regulatory compliance.
We invite you to engage with our technical procurement team to discuss how this innovative photocatalytic technology can be tailored to your specific project needs, offering a Customized Cost-Saving Analysis that quantifies the potential economic benefits for your operation. By requesting specific COA data and route feasibility assessments, you can gain deeper insights into the practical application of this iridium-carbene complex and explore opportunities for collaboration that drive efficiency and innovation in your chemical manufacturing processes. Let us partner with you to unlock the full potential of green chemistry and secure a sustainable future for your supply chain.
