Advanced Rhodium-Catalyzed Synthesis of Aryl Eneyne Compounds for Commercial Scale-Up
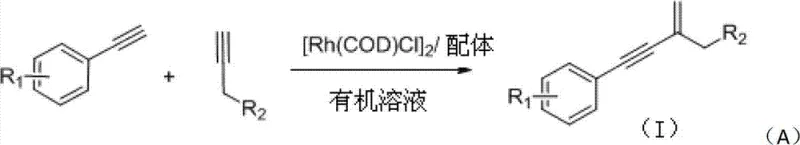
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient, atom-economical, and selective transformations. A significant breakthrough in this domain is detailed in Chinese Patent CN103102228A, which discloses a novel preparation method for aryl eneyne compounds. This technology leverages a rhodium-complex catalytic system to facilitate the direct cross-hydroalkynylation of terminal alkynes, specifically reacting aryl alkynes with propargyl alcohols or propargyl amines. Unlike traditional methods that often suffer from poor regioselectivity or require harsh conditions, this approach operates under mild temperatures ranging from 25°C to 40°C in common organic solvents. The innovation lies in the specific use of a catalyst generated in situ from [Rh(COD)Cl]2 and phosphine ligands, which enables the formation of valuable gem-2-alkynyl allyl alcohol and amine structures with high efficiency. For global procurement teams and R&D directors, this represents a pivotal shift towards more sustainable and cost-effective manufacturing routes for complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of conjugated eneyne compounds has relied heavily on methods such as metal-catalyzed homocoupling of alkynes or cross-coupling reactions involving vinyl halides. These conventional pathways often present significant challenges in terms of atom economy and selectivity. For instance, traditional coupling strategies may require pre-functionalized starting materials like vinyl halides, which add steps to the synthesis and generate stoichiometric amounts of salt waste. Furthermore, controlling the regioselectivity to ensure the desired head-to-tail arrangement of the alkyne units is notoriously difficult, often leading to complex mixtures of isomers that are costly and time-consuming to separate. The reliance on palladium or copper catalysts in some older methodologies can also introduce issues with heavy metal contamination, necessitating rigorous and expensive purification protocols to meet the stringent purity specifications required for active pharmaceutical ingredients (APIs).
The Novel Approach
The methodology described in the patent data offers a transformative solution by utilizing a direct cross-hydroalkynylation strategy. By employing a rhodium catalyst system composed of [Rh(COD)Cl]2 and specific ligands such as triphenylphosphine (PPh3), the reaction achieves exceptional regioselectivity, favoring the formation of the desired gem-2-alkynyl structure. This approach eliminates the need for pre-activated halide partners, thereby streamlining the synthetic route and improving the overall atom economy. The reaction conditions are remarkably mild, typically proceeding overnight at temperatures as low as 40°C in solvents like dichloromethane (DCM). This not only reduces energy consumption but also minimizes the thermal degradation of sensitive functional groups. The ability to tolerate a wide range of substituents on the aryl ring, including electron-withdrawing nitro groups and electron-donating methoxy groups, underscores the versatility of this novel catalytic system for generating diverse chemical libraries.
Mechanistic Insights into Rh-Catalyzed Cross-Hydroalkynylation
The core of this technological advancement lies in the unique behavior of the rhodium complex formed between [Rh(COD)Cl]2 and the phosphine ligand. Mechanistically, the reaction is believed to proceed through a catalytic cycle where the rhodium center activates the terminal alkyne C-H bond, forming a rhodium-acetylide species. This activation is crucial as it allows for the subsequent insertion of the second alkyne molecule in a highly controlled manner. The presence of the ligand, particularly PPh3, plays a critical role in stabilizing the active catalytic species and modulating the electronic environment around the metal center. This modulation is what drives the high regioselectivity observed, ensuring that the coupling occurs in a head-to-tail fashion rather than forming unwanted dimers or head-to-head isomers. The precise stoichiometry, typically involving a molar ratio of 1:1.5 for the aryl alkyne to propargyl derivative and 5 mol% catalyst loading, is optimized to maximize turnover frequency while minimizing catalyst decomposition.
From an impurity control perspective, this mechanism offers distinct advantages. The high selectivity of the rhodium catalyst significantly reduces the formation of regioisomeric byproducts, which are often the most difficult impurities to remove during downstream processing. In traditional free-radical or non-selective metal-catalyzed reactions, the impurity profile can be complex, requiring multiple recrystallizations or chromatographic steps. In contrast, the Rh-catalyzed pathway yields a cleaner crude reaction mixture. The patent data indicates that simple column chromatography using standard solvent systems like ethyl acetate and petroleum ether is sufficient to isolate the pure product. This simplification of the purification process is a key factor in reducing the overall cost of goods sold (COGS) and shortening the production cycle time, making it highly attractive for commercial scale-up.
How to Synthesize Aryl Eneyne Compounds Efficiently
The practical implementation of this synthesis route is straightforward and robust, designed for reproducibility in both laboratory and pilot plant settings. The process begins with the preparation of the active catalyst under an inert atmosphere, followed by the sequential addition of substrates. Detailed standardized synthesis steps are provided in the guide below to ensure consistent quality and yield.
- Prepare the catalyst system by mixing [Rh(COD)Cl]2 (5 mol%) and PPh3 ligand (20-30 mol%) in anhydrous dichloromethane under argon.
- Add the aryl alkyne substrate to the reaction mixture, followed by the slow addition of propargyl alcohol or propargyl amine via peristaltic pump.
- Stir the reaction overnight at 40°C, then filter, concentrate, and purify the crude product via column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rhodium-catalyzed technology translates into tangible strategic benefits beyond just chemical elegance. The primary driver is the significant reduction in manufacturing complexity and associated costs. By utilizing a direct coupling method that avoids pre-functionalized reagents, the supply chain becomes less dependent on specialized, high-cost starting materials. The raw materials, primarily aryl alkynes and propargyl alcohols or amines, are commodity chemicals that are readily available from multiple global suppliers, mitigating the risk of supply disruptions. Furthermore, the mild reaction conditions imply lower energy requirements for heating and cooling, contributing to a smaller carbon footprint and reduced utility costs. The high selectivity of the reaction means less waste generation and lower solvent consumption during purification, aligning with modern environmental compliance standards and reducing waste disposal expenses.
- Cost Reduction in Manufacturing: The elimination of expensive halide precursors and the use of low catalyst loadings (5 mol%) directly impact the bottom line. While rhodium is a precious metal, the high efficiency and turnover of the catalyst system ensure that the cost per kilogram of product remains competitive. Additionally, the simplified purification process reduces the consumption of silica gel and solvents, which are often hidden cost drivers in fine chemical manufacturing. The overall process efficiency leads to substantial cost savings compared to multi-step traditional routes.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions allows for flexible manufacturing scheduling. Since the reaction tolerates a broad range of functional groups, the same platform technology can be used to produce a variety of derivatives without needing to requalify entirely new processes. This flexibility ensures that production can be quickly adapted to meet changing market demands. The use of common solvents like DCM and standard workup procedures further ensures that the process can be easily transferred between different manufacturing sites without significant re-engineering.
- Scalability and Environmental Compliance: The atom-economical nature of the cross-hydroalkynylation reaction minimizes the generation of stoichiometric byproducts, adhering to green chemistry principles. This is increasingly important for meeting regulatory requirements in major markets like the EU and US. The process is inherently scalable, as demonstrated by the successful synthesis of various derivatives with consistent yields. The ability to produce high-purity intermediates with minimal heavy metal contamination simplifies the regulatory filing process for downstream API manufacturers, accelerating time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners.
Q: What is the primary advantage of this Rh-catalyzed method over traditional coupling reactions?
A: The primary advantage is the high regioselectivity for head-to-tail coupling without the need for pre-functionalized halides, offering superior atom economy compared to traditional cross-coupling methods.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes mild temperatures (25-40°C) and commercially available solvents like DCM, making it highly suitable for scale-up with manageable safety profiles and standard purification techniques.
Q: What types of functional groups are tolerated in this synthesis?
A: The method demonstrates excellent tolerance for various substituents including methoxy, nitro, bromo, amino, and acetylamino groups on the aryl ring, allowing for diverse derivative synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Eneyne Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics. Our technical team has extensively analyzed the Rh-catalyzed cross-hydroalkynylation pathway and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of aryl eneyne compound meets the highest industry standards. We are committed to delivering products that facilitate your R&D success while optimizing your supply chain efficiency.
We invite you to collaborate with us to leverage this advanced synthetic technology for your projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments for your target molecules. Let us partner with you to drive innovation and efficiency in your chemical supply chain.
