Scalable Cobalt-Catalyzed Synthesis of 2-Alkoxyindoles for Commercial API Production
Introduction to Advanced 2-Alkoxyindole Synthesis
The synthesis of 2-alkoxyindole compounds represents a critical challenge and opportunity in modern medicinal chemistry, particularly given their prevalence in high-value bioactive molecules. As detailed in the recent patent literature, specifically CN115772157A, new methodologies are emerging that promise to revolutionize how these scaffolds are accessed. The 2-alkoxyindole motif is not merely an academic curiosity; it serves as the structural backbone for potent selective 5-HT4 receptor antagonists, including notable candidates like GR-125487 and SB-207266. These molecules are pivotal in the treatment of gastrointestinal disorders and other central nervous system conditions, driving a consistent demand for reliable supply chains capable of delivering high-purity intermediates.
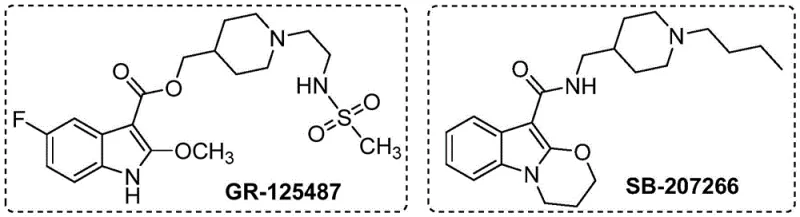
Traditionally, accessing these structures has required convoluted multi-step sequences or the use of prohibitively expensive noble metal catalysts. However, the technological breakthrough described in the referenced patent introduces a transition metal cobalt-catalyzed C-H activated alkoxylation reaction. This approach utilizes indole compounds as starting materials, directly functionalizing the C2 position with high efficiency. For R&D directors and procurement managers alike, this shift represents a move towards more sustainable and economically viable manufacturing processes. By leveraging earth-abundant cobalt instead of scarce precious metals, the industry can achieve significant cost reduction in pharmaceutical intermediate manufacturing while maintaining rigorous quality standards required for drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 2-alkoxyindole frameworks has been plagued by synthetic inefficiencies that hinder large-scale production. Conventional routes often rely on pre-functionalized starting materials, necessitating multiple protection and deprotection steps that drastically reduce overall atom economy. Furthermore, many established methods depend heavily on palladium or rhodium catalysis. While effective on a milligram scale in academic settings, these noble metals introduce severe bottlenecks for commercial operations. The high cost of the catalysts themselves, coupled with the stringent regulatory requirements for removing trace heavy metals from final API products, creates a substantial financial burden. Additionally, traditional cross-coupling reactions often require harsh conditions, specialized ligands that are difficult to source in bulk, and generate significant amounts of toxic waste, complicating environmental compliance and increasing the total cost of ownership for the synthesis.
The Novel Approach
In stark contrast, the novel cobalt-catalyzed strategy outlined in patent CN115772157A offers a streamlined alternative that directly addresses these pain points. This method employs a direct C-H activation mechanism, effectively bypassing the need for pre-halogenated substrates and reducing the step count significantly. The use of cobalt acetylacetonate [Co(acac)2] as the catalyst is a game-changer; it is inexpensive, commercially available in metric ton quantities, and robust under the reaction conditions. The process operates in the alcohol solvent itself, which acts as both the reaction medium and the alkoxylating reagent, simplifying the formulation and reducing solvent waste. This "one-pot" style efficiency translates directly into operational simplicity, allowing for faster turnaround times in process development and a more agile response to market demands for diverse indole derivatives.
Mechanistic Insights into Cobalt-Catalyzed C-H Alkoxylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction initiates with the oxidation of the cobalt(II) catalyst by silver carbonate (Ag2CO3), generating a reactive cobalt(III) species. This high-valent metal center then coordinates with the indole substrate, likely facilitated by the 8-quinolinyl directing group present in the starting material. This coordination is essential for achieving regioselectivity at the C2 position, ensuring that the alkoxylation occurs precisely where needed without affecting other sensitive functional groups on the indole ring. The subsequent steps involve a single-electron transfer (SET) process that generates a radical cobalt(II) complex, followed by further oxidation and C-H bond activation to form a stable cobalt(III)-indole intermediate.
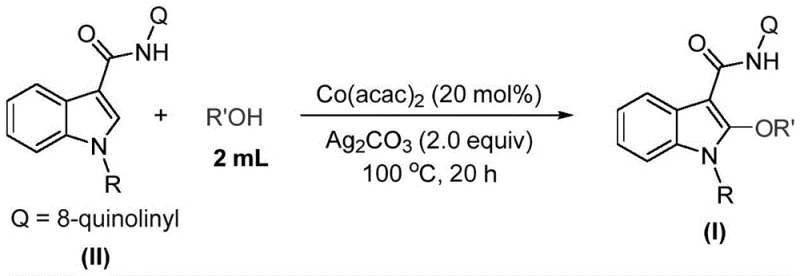
The final stage of the catalytic cycle involves the coordination of the alcohol molecule to the cobalt center, followed by migratory insertion and reductive elimination. This sequence releases the desired 2-alkoxyindole product and regenerates the active cobalt species, allowing the cycle to continue. From a purity perspective, this mechanism is advantageous because it avoids the formation of homocoupling byproducts often seen in radical reactions, provided the oxidant loading is controlled. The patent specifies a stoichiometry of 1:0.2:2 for the indole, catalyst, and oxidant respectively, which balances reaction rate with impurity profile. The use of silver carbonate as a terminal oxidant ensures that the cobalt remains in the active oxidation state throughout the 16 to 24-hour reaction window at 100°C, driving the conversion to completion with yields reaching up to 90% for optimized substrates.
How to Synthesize 2-Alkoxyindole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to ensure reproducibility and safety. The protocol is remarkably robust, tolerating a wide range of substituents on both the indole nitrogen and the alcohol component. Whether utilizing simple methanol for methoxy derivatives or bulkier secondary alcohols like isopropanol, the system maintains high efficiency. The reaction is typically conducted in a Schlenk tube or sealed vessel to maintain the integrity of the solvent at elevated temperatures. Post-reaction processing is equally straightforward, involving simple filtration to remove silver salts followed by standard silica gel chromatography. This ease of purification is a critical factor for supply chain reliability, as it minimizes the time and resources spent on downstream processing.
- Combine cobalt acetylacetonate (20 mol%), the indole substrate (1.0 equiv), and silver carbonate (2.0 equiv) in a reaction vessel.
- Add the corresponding alcohol (acting as both solvent and reagent, approx. 2 mL per 0.2 mmol substrate) to the mixture.
- Heat the reaction mixture to 100°C for 20 hours, then filter and purify via column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed technology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for expensive noble metals and complex ligands, companies can insulate themselves from the volatile pricing of precious metal markets. Furthermore, the reliance on commodity chemicals like cobalt acetylacetonate and silver carbonate ensures a stable and continuous supply of critical reagents, reducing the risk of production stoppages due to material shortages. The operational simplicity of the process also means that it can be transferred between manufacturing sites with minimal re-validation, enhancing overall supply chain resilience.
- Cost Reduction in Manufacturing: The economic impact of switching from noble metal catalysis to base metal catalysis cannot be overstated. Cobalt is orders of magnitude cheaper than palladium or rhodium, leading to immediate savings on the bill of materials. Additionally, the simplified workup procedure reduces labor costs and solvent consumption. The elimination of specialized scavengers required for removing trace palladium from APIs further lowers the cost of goods sold (COGS), making the final drug product more competitive in the marketplace.
- Enhanced Supply Chain Reliability: Sourcing high-purity precursors is often a bottleneck in API production. This method utilizes widely available indole derivatives and common alcohols, which are produced globally at massive scales. This abundance ensures that lead times for raw materials remain short and predictable. Moreover, the robustness of the reaction conditions means that minor variations in reagent quality do not compromise the outcome, providing a buffer against supply chain fluctuations and ensuring consistent delivery schedules to downstream customers.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry, this process aligns perfectly with sustainability goals. The use of earth-abundant metals reduces the environmental footprint associated with mining and refining precious metals. The reaction generates less hazardous waste compared to traditional halogenation/coupling sequences, simplifying waste disposal and lowering compliance costs. The scalability of the reaction, demonstrated by its successful application across a broad substrate scope, indicates that it can be readily adapted from gram-scale R&D to multi-kilogram commercial production without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of using cobalt catalysts over noble metals for indole functionalization?
A: Cobalt catalysts, such as Co(acac)2, offer a substantial cost advantage over traditional palladium or rhodium systems. They are earth-abundant, significantly cheaper, and often exhibit unique reactivity profiles that allow for milder conditions and broader substrate tolerance, reducing overall production costs.
Q: Can this C-H activation method be scaled for industrial manufacturing?
A: Yes, the protocol described in patent CN115772157A is designed for scalability. It utilizes simple reagents, avoids exotic ligands, and employs a straightforward workup procedure involving filtration and chromatography, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the substrate scope regarding the alcohol component in this alkoxylation reaction?
A: The method demonstrates excellent compatibility with various primary and secondary alcohols, including methanol, ethanol, isopropanol, and even trifluoroethanol. This versatility allows for the rapid generation of diverse 2-alkoxyindole libraries for SAR studies without needing to change the core catalytic system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkoxyindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating drug discovery and development. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory methods like the cobalt-catalyzed alkoxylation described here can be seamlessly transitioned to industrial reality. We pride ourselves on our stringent purity specifications and rigorous QC labs, which guarantee that every batch of 2-alkoxyindole intermediate meets the exacting standards required by global regulatory bodies. Our commitment to quality ensures that your supply chain remains uninterrupted and compliant.
We invite you to collaborate with us to leverage these cutting-edge synthetic strategies for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in cobalt catalysis can drive efficiency and value for your organization.
