Scalable Green Synthesis of Alkenyl Azaarenes for Advanced Pharmaceutical Manufacturing
Scalable Green Synthesis of Alkenyl Azaarenes for Advanced Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to synthesize complex heterocyclic scaffolds. Patent CN110128335B discloses a groundbreaking green synthesis method for alkenyl azaarene compounds, which are pivotal structural motifs in numerous bioactive macromolecules and natural products. This innovation addresses critical bottlenecks in traditional organic synthesis by utilizing a synergistic catalytic system comprising an acid and an active methylene compound. By enabling direct sp3 C-H functionalization of 2-methylquinoline derivatives with aldehydes, this technology offers a robust alternative to legacy methods that often suffer from harsh reaction conditions, excessive reagent consumption, and difficult purification processes. For R&D directors and procurement managers alike, this represents a significant opportunity to streamline supply chains for high-value pharmaceutical intermediates.
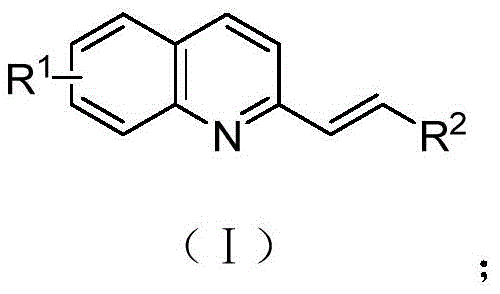
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkenyl azaarene derivatives has relied on several established but flawed methodologies, including the Wittig reaction of quinoline aldehydes, reductive olefination of quinoline N-oxides, and dehydration-dehydrogenation reactions involving alcohols. While the direct dehydration reaction between 2-methylquinolines and aldehydes is theoretically the most straightforward, existing implementations frequently demand excessively high temperatures exceeding 111°C and require a large excess of aldehyde reagents to drive equilibrium, leading to substantial raw material waste. Furthermore, many alternative routes necessitate the use of unstable raw materials, cumbersome multi-step procedures, strong oxidants, or expensive transition metal catalysts. These factors not only inflate production costs but also introduce significant challenges in downstream processing, such as the removal of toxic metal residues and the separation of complex by-product mixtures, which can severely impact the purity profile required for pharmaceutical applications.
The Novel Approach
In stark contrast to these conventional limitations, the method described in patent CN110128335B introduces a highly efficient one-step synthesis strategy that operates under remarkably mild conditions. By employing a dual-catalyst system of organic acid and active methylene compounds in a water and 1,4-dioxane mixed solvent, the reaction proceeds effectively at temperatures ranging from 25°C to 111°C, with an optimal performance observed at 61°C. This approach eliminates the need for strong oxidants and metal catalysts entirely, thereby simplifying the work-up procedure and enhancing the environmental profile of the manufacturing process. Crucially, this method drastically reduces the consumption of aldehyde raw materials, allowing for a near-stoichiometric molar ratio of 1:1 between the 2-methylquinoline derivative and the aldehyde, which translates directly into significant cost savings and improved atom economy for large-scale production.

Mechanistic Insights into Synergistic Organocatalysis
The core innovation of this technology lies in the discovery of a synergistic catalytic effect between specific organic acids and active methylene compounds, specifically 1,3-dimethylbarbituric acid (1,3-DMBBA). Extensive optimization studies revealed that neither the acid nor the active methylene compound alone could achieve high conversion rates; however, their combination facilitates the activation of the sp3 C-H bond at the methyl position of the quinoline ring. This activation enables a direct condensation with the carbonyl group of the aldehyde, followed by dehydration to form the desired alkene linkage. The presence of water as a major solvent component further enhances the green chemistry credentials of the process, while the small amount of 1,4-dioxane ensures adequate solubility of the organic substrates and catalysts, maximizing intermolecular contact and reaction efficiency without requiring hazardous organic solvents in large volumes.
From a quality control perspective, this mechanism offers superior impurity management compared to metal-catalyzed alternatives. The absence of transition metals removes the risk of heavy metal contamination, a critical parameter for API intermediates destined for human therapeutic use. Furthermore, the high selectivity of the organocatalytic system minimizes the formation of side products, resulting in crude reaction mixtures that are easier to purify. Experimental data indicates that yields can reach up to 95% under optimized conditions, with excellent tolerance for various functional groups including halogens, nitro groups, and alkoxy substituents on both the quinoline and aldehyde moieties. This robustness ensures consistent product quality across a wide range of substrate variations, providing R&D teams with the flexibility to explore diverse chemical spaces without compromising on yield or purity.
How to Synthesize Alkenyl Azaarene Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. The process involves charging a reactor with 2-methylquinoline derivatives and aldehyde compounds, followed by the addition of the synergistic catalyst system consisting of glacial acetic acid and 1,3-dimethylbarbituric acid. The reaction is conducted in a mixed solvent system of water and 1,4-dioxane at a controlled temperature of 61°C for approximately 24 hours. Upon completion, the work-up involves a straightforward extraction with ethyl acetate and purification via column chromatography using a petroleum ether and ethyl acetate gradient. For detailed standard operating procedures and specific stoichiometric ratios tailored to your specific substrate requirements, please refer to the technical guide below.
- Mix 2-methylquinoline derivatives and aldehyde compounds in a reactor with water and 1,4-dioxane as the mixed solvent system.
- Add glacial acetic acid and 1,3-dimethylbarbituric acid as synergistic catalysts to initiate the sp3 C-H functionalization.
- Heat the reaction mixture to 61°C for 24 hours, then perform extraction and column chromatography to isolate the high-purity target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits regarding cost structure and operational reliability. The elimination of expensive transition metal catalysts not only reduces direct material costs but also removes the need for specialized scavenging resins or complex filtration steps typically required to meet strict residual metal specifications. This simplification of the downstream processing workflow leads to shorter batch cycle times and reduced labor overheads. Additionally, the ability to use a stoichiometric amount of aldehyde rather than a large excess significantly lowers the raw material bill of materials, particularly when dealing with high-value or scarce aldehyde building blocks. These efficiencies combine to create a manufacturing process that is inherently more cost-competitive and resilient to fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic reduction in reagent consumption and the simplification of the purification train. By avoiding the use of precious metal catalysts and strong oxidants, the process eliminates costly waste disposal fees associated with hazardous chemical treatment. Furthermore, the high yield and selectivity minimize the loss of valuable starting materials, ensuring that a greater proportion of input costs are converted into saleable product. This lean manufacturing approach allows for substantial margin improvement, making the final alkenyl azaarene intermediates more price-competitive in the global market without sacrificing quality standards.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as 2-methylquinoline derivatives and common aromatic aldehydes, ensures a robust and secure supply chain. Unlike methods requiring sensitive or unstable reagents that may have long lead times or limited supplier bases, this protocol utilizes commodity chemicals that are readily accessible from multiple sources. This diversification of supply reduces the risk of production stoppages due to raw material shortages. Moreover, the mild reaction conditions reduce the stress on equipment and lower energy consumption, contributing to higher asset utilization rates and more predictable production schedules for long-term supply agreements.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of water as the primary solvent, which significantly reduces the volume of flammable organic solvents required. This aligns with increasingly stringent environmental regulations and corporate sustainability goals by lowering the carbon footprint and VOC emissions of the manufacturing facility. The simple work-up procedure, involving basic extraction and crystallization or chromatography, is easily adaptable to large-scale reactors, ensuring that the high yields observed in the lab can be maintained during commercial scale-up. This scalability ensures that supply can be rapidly ramped up to meet market demand for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is essential for evaluating the feasibility of integrating this method into your existing manufacturing portfolio.
Q: What are the primary advantages of this synthesis method over traditional Wittig reactions?
A: Unlike traditional Wittig reactions which often require harsh conditions and generate significant phosphorus waste, this novel method utilizes a metal-free organocatalytic system. It operates under mild temperatures (25-111°C) and significantly reduces the consumption of expensive aldehyde raw materials by enabling a near-stoichiometric 1:1 molar ratio, thereby lowering both operational costs and environmental impact.
Q: Does this process require expensive transition metal catalysts?
A: No, this process completely eliminates the need for transition metal catalysts or strong oxidants. By employing a synergistic combination of organic acids (such as glacial acetic acid) and active methylene compounds (like 1,3-dimethylbarbituric acid), the reaction achieves high yields without the risk of heavy metal contamination, which is critical for pharmaceutical intermediate production.
Q: What is the substrate scope for this alkenyl azaarene synthesis?
A: The method demonstrates excellent functional group tolerance and broad substrate applicability. It successfully accommodates various 2-methylquinoline derivatives substituted with halogens, alkyl, or alkoxy groups, and reacts efficiently with diverse aldehydes including aryl, heteroaryl, and aliphatic aldehydes, making it highly versatile for generating complex molecular libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkenyl Azaarene Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic technologies like the one described in patent CN110128335B to enhance our service offerings. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alkenyl azaarene intermediates delivered meets the highest international standards for pharmaceutical applications. We are committed to leveraging our technical expertise to optimize this green synthesis route for your specific project needs.
We invite you to engage with our technical team to explore how this cost-effective and environmentally friendly methodology can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic advantages specific to your target molecules. We encourage you to contact our technical procurement team today to discuss your requirements,索取 specific COA data, and receive comprehensive route feasibility assessments tailored to your development timeline. Let us partner with you to accelerate your drug development programs with reliable, high-quality chemical solutions.
