Advanced Copper-Catalyzed Synthesis of Polysubstituted Pyridines for Commercial Scale-Up
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for efficient pathways to access nitrogen-containing scaffolds that serve as the backbone of modern therapeutics. Patent CN108912044B introduces a transformative methodology for the construction of polysubstituted pyridines, leveraging a copper-catalyzed [3+2+1] cyclization strategy that fundamentally shifts the paradigm from traditional, labor-intensive syntheses to streamlined, one-pot operations. This innovation addresses critical bottlenecks in the supply chain of high-purity pharmaceutical intermediates by utilizing inexpensive cuprous iodide instead of scarce noble metals, thereby aligning synthetic efficiency with economic viability. The technical breakthrough lies in the ability to couple alkenyl azides with difluoromethylene compounds under remarkably mild thermal conditions, achieving high regioselectivity without the need for rigorous anhydrous environments or cryogenic temperatures. For R&D directors seeking robust routes to complex pyridine derivatives, this patent offers a validated protocol that minimizes side reactions and maximizes atom economy, ensuring that the resulting chemical entities meet the stringent purity specifications required for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyridine rings has relied heavily on classical condensation reactions such as the Hantzsch or Chichibabin methodologies, which, while foundational, suffer from significant operational drawbacks in a modern industrial context. These traditional pathways often necessitate multi-step sequences involving the pre-functionalization of substrates, leading to accumulated waste and reduced overall throughput due to the isolation of unstable intermediates. Furthermore, the reliance on harsh acidic or basic conditions can compromise sensitive functional groups, limiting the scope of accessible chemical space and necessitating extensive protection-deprotection strategies that inflate production costs. Transition metal-catalyzed alternatives using rhodium or palladium have emerged to mitigate some of these issues, yet they introduce new challenges related to the high cost of the catalysts and the difficulty of removing trace metal residues to meet regulatory limits for active pharmaceutical ingredients. The inherent complexity of controlling regioselectivity in these older methods often results in isomeric mixtures that are difficult to separate, creating a bottleneck in the purification stage that delays time-to-market for new drug candidates.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in CN108912044B employs a sophisticated yet operationally simple copper-catalyzed cascade that constructs the pyridine core in a single synthetic step from readily available building blocks. By utilizing alkenyl azides as three-atom synthons and difluoromethylene compounds as two-atom components, the reaction achieves a precise [3+2+1] assembly that inherently dictates the substitution pattern of the final product, effectively eliminating the formation of unwanted regioisomers. The use of a copper-based catalytic system, specifically cuprous iodide augmented by ligands like triphenylphosphine and PMDETA, allows the transformation to proceed at a moderate temperature of 40°C, drastically reducing energy consumption compared to high-temperature reflux protocols. This approach not only simplifies the workflow by avoiding intermediate isolation but also expands the substrate tolerance to include a wide array of electron-rich and electron-deficient aryl and heteroaryl groups, providing medicinal chemists with a versatile tool for rapid library generation.
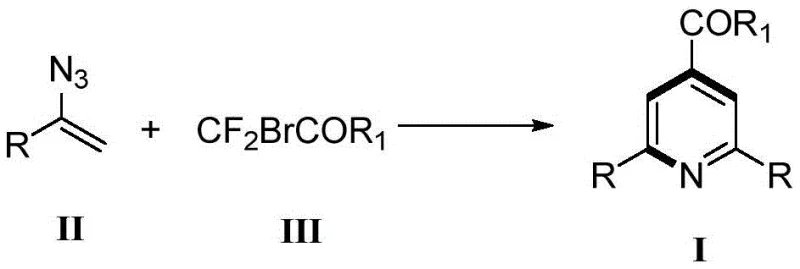
Mechanistic Insights into Copper-Catalyzed [3+2+1] Cyclization
The mechanistic underpinning of this transformation involves a complex interplay between the copper catalyst and the reactive azide functionality, initiating a cascade that ultimately furnishes the aromatic pyridine system with high fidelity. The reaction likely proceeds through the generation of a copper-nitrenoid or copper-carbene species upon interaction with the alkenyl azide, which then undergoes insertion or cycloaddition with the difluoromethylene partner to form the initial cyclic intermediate. The presence of sodium iodide and phosphine ligands plays a crucial role in stabilizing the active copper species and facilitating the reductive elimination steps necessary to restore aromaticity and release the final product. This catalytic cycle is highly efficient, as evidenced by the ability to use catalyst loadings as low as 10-50 mol% relative to the substrate while maintaining excellent conversion rates, suggesting a robust turnover number that is essential for cost-effective manufacturing. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as ligand ratios and solvent polarity, to further optimize the kinetics and suppress potential side reactions like azide decomposition or polymerization.
From an impurity control perspective, the high regioselectivity of this copper-catalyzed process is a significant advantage, as it minimizes the formation of structural isomers that often plague non-catalytic or less selective metal-mediated reactions. The specific coordination environment created by the PMDETA and triphenylphosphine ligands directs the approach of the reactants, ensuring that the cyclization occurs exclusively at the desired positions to yield the 2,4,6-substituted pyridine architecture. This precision reduces the burden on downstream purification units, as the crude reaction mixture contains fewer closely related impurities that would otherwise require resource-intensive chromatographic separation. Furthermore, the mild reaction conditions prevent the degradation of sensitive functional groups on the aryl rings, preserving the integrity of the molecular scaffold and ensuring that the impurity profile remains clean and predictable, which is a critical factor for regulatory approval in pharmaceutical applications.
How to Synthesize Polysubstituted Pyridines Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators should adhere to the optimized protocol detailed in the patent examples, which balances reaction speed with maximum yield. The procedure begins by dissolving the alkenyl azide and the difluoromethylene compound in dimethyl sulfoxide (DMSO), followed by the sequential addition of the ligand system comprising PMDETA and triphenylphosphine along with sodium iodide to activate the catalyst precursor. Once the mixture is homogenized and heated to the optimal temperature of 40°C, the cuprous iodide catalyst is introduced to initiate the cyclization, with the reaction typically reaching completion within two hours as monitored by thin-layer chromatography. The detailed standardized synthesis steps for scaling this process are outlined below.
- Dissolve alkenyl azide and difluoromethylene compound in DMSO solvent with PMDETA, triphenylphosphine, and sodium iodide additives.
- Add cuprous iodide (CuI) catalyst to the mixture and heat the reaction system to 40°C while stirring continuously.
- Monitor reaction progress via TLC, then perform aqueous workup, extraction with dichloromethane, and purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed methodology represents a strategic opportunity to reduce the total cost of ownership for pyridine-based intermediates while enhancing supply security. The shift from precious metal catalysts to abundant copper salts eliminates the volatility associated with rhodium and palladium pricing, providing a stable cost structure that facilitates long-term budget planning and contract negotiations. Additionally, the simplified one-pot nature of the reaction reduces the requirement for specialized reactor configurations and extensive solvent swaps, leading to significant operational efficiencies in multipurpose manufacturing facilities. This streamlined process flow translates directly into shorter production cycles and increased asset utilization, allowing suppliers to respond more agilely to fluctuating market demands without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with inexpensive cuprous iodide results in substantial raw material cost savings, particularly when scaled to metric ton quantities where catalyst expense becomes a major line item. The elimination of complex multi-step sequences and intermediate isolations further drives down labor and utility costs, as the process requires fewer unit operations and less solvent consumption per kilogram of product. Moreover, the high yields achieved under mild conditions minimize the loss of valuable starting materials, improving the overall mass balance and reducing the cost of goods sold for the final active pharmaceutical ingredient. These cumulative efficiencies enable a more competitive pricing strategy for high-purity pharmaceutical intermediates in the global market.
- Enhanced Supply Chain Reliability: Utilizing broadly available and stable reagents such as alkenyl azides and difluoromethylene compounds mitigates the risk of supply disruptions often associated with exotic or highly specialized starting materials. The robustness of the copper catalytic system ensures consistent batch-to-batch reproducibility, which is essential for maintaining continuous supply lines to downstream drug manufacturers who operate on tight just-in-time schedules. By simplifying the synthetic route, the potential for process deviations is lowered, leading to higher first-pass success rates and reducing the likelihood of batch failures that could jeopardize delivery commitments. This reliability strengthens the partnership between chemical suppliers and their clients, fostering trust and long-term collaboration.
- Scalability and Environmental Compliance: The mild reaction temperature of 40°C significantly lowers the energy footprint of the manufacturing process compared to high-temperature alternatives, aligning with corporate sustainability goals and reducing utility expenditures. The use of DMSO as a solvent, which can be recovered and recycled, combined with the absence of toxic heavy metals, simplifies waste treatment protocols and ensures compliance with increasingly stringent environmental regulations. The straightforward workup procedure involving aqueous quenching and extraction facilitates easy scale-up from gram to kilogram scales without the need for complex engineering controls, making this technology ideal for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed pyridine synthesis, drawing directly from the experimental data and scope defined in the patent documentation. These insights are intended to clarify the operational feasibility and strategic benefits for stakeholders evaluating this technology for integration into their existing manufacturing portfolios. By understanding the specific advantages and limitations outlined here, decision-makers can better assess the fit of this methodology for their specific project requirements.
Q: What are the primary advantages of this copper-catalyzed method over traditional pyridine synthesis?
A: Unlike traditional Hantzsch or Chichibabin methods which often require harsh conditions and complex multi-step sequences, this copper-catalyzed [3+2+1] cyclization operates under mild conditions (40°C) with inexpensive catalysts, offering superior regioselectivity and simplified purification processes.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes readily available raw materials like alkenyl azides and difluoromethylene compounds, avoids expensive noble metals like rhodium or palladium, and demonstrates high yields up to 90%, making it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the substrate scope for the R groups in this pyridine synthesis?
A: The protocol exhibits excellent universality, accommodating electron-rich and electron-deficient aryl groups, substituted phenyls with halogens or alkyl chains, and heteroaromatic systems like thiophene, allowing for diverse functionalization of the pyridine core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyridines Supplier
As the pharmaceutical industry continues to demand more efficient and sustainable routes to complex heterocycles, NINGBO INNO PHARMCHEM stands ready to leverage this advanced copper-catalyzed technology to support your development pipeline. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and compliant. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of polysubstituted pyridines meets the exacting standards required for clinical and commercial applications, providing you with a reliable partner for your critical supply chain needs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific molecule requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this copper-catalyzed route for your projects. Please contact us today to obtain specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical intermediates can accelerate your time to market while optimizing your production costs.
