Advancing Pharmaceutical Intermediate Manufacturing with Mild Photocatalytic N-Substitution Technology
The landscape of fine chemical synthesis is undergoing a significant transformation driven by the demand for greener, more efficient methodologies, particularly in the realm of nitrogen-containing heterocycles and sulfur-based intermediates. A pivotal advancement in this sector is detailed in Chinese Patent CN111925308B, which discloses a novel method for synthesizing N-substituted sulfone diimines via photocatalysis. This technology represents a substantial leap forward from classical oxidative imidization techniques, utilizing a specialized iridium catalyst under visible light irradiation to achieve N-C bond formation under remarkably mild conditions. For R&D directors and process chemists, this patent offers a robust alternative to harsh chemical oxidants, enabling the functionalization of sulfone diimines at room temperature without the need for extreme thermal energy or hazardous reagents. The implications for the supply chain of high-purity pharmaceutical intermediates are profound, as this method simplifies post-treatment procedures and expands the accessible chemical space for drug discovery programs targeting sulfur-rich bioactive scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfone diimines has been plagued by significant operational challenges and substrate limitations that hindered their widespread adoption in medicinal chemistry. Early methodologies, such as those reported by Cogliano and Braude, relied on the reaction of thioethers with tert-butyl hypochlorite and liquid ammonia, a process that was not only operationally difficult but also failed to accommodate S-aryl or S,S-diaryl substituted variants effectively. Subsequent improvements by Oae utilizing Chloramine-T allowed for the preparation of S,S-diaryl substituted sulfone diimines but remained incompatible with S-alkyl substrates, thereby restricting the structural diversity available to chemists. Furthermore, traditional N-C coupling reactions often necessitated very harsh reaction conditions and highly reactive substituent groups, leading to poor functional group tolerance and complicating the purification of sensitive intermediates. These legacy methods frequently resulted in low yields, significant waste generation, and safety concerns associated with strong oxidants, creating bottlenecks in the reliable production of complex sulfone diimine derivatives required for advanced agrochemical and pharmaceutical applications.
The Novel Approach
In stark contrast to these restrictive historical precedents, the photocatalytic method described in the patent data introduces a versatile and mild platform for N-substitution that overcomes previous substrate barriers. By employing an iridium-based photocatalyst activated by white light, the reaction proceeds efficiently at room temperature in common aprotic solvents like tetrahydrofuran, eliminating the need for cryogenic conditions or high-temperature heating. This approach successfully enables the synthesis of S-alkyl S-aryl substituted sulfone diimines, S-benzyl S-aryl substituted variants, and even certain S,S-diaryl substituted compounds that were previously difficult to access. The use of simple inorganic bases such as potassium carbonate further enhances the practicality of the process, allowing for a straightforward workup that minimizes environmental impact. This technological shift not only broadens the applicable substrate range to include electron-donating and electron-withdrawing groups on aromatic rings but also facilitates the incorporation of heterocycles like pyridine and thiophene, which are critical motifs in modern drug design.

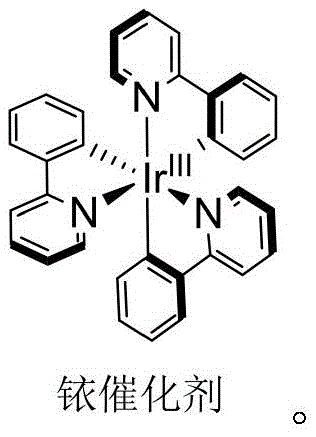
Mechanistic Insights into Iridium-Catalyzed Photocatalytic N-Substitution
The core of this innovative synthesis lies in the unique photophysical properties of the iridium catalyst, which acts as a potent mediator for single-electron transfer processes under visible light irradiation. Upon absorption of photons from a 5W white light source, the iridium complex enters an excited state capable of interacting with the bromide substrate, likely generating a carbon-centered radical species through homolytic cleavage of the carbon-bromine bond. This radical intermediate then engages with the nitrogen center of the sulfone diimine scaffold, facilitating the formation of the new N-C bond without the requirement for aggressive electrophiles or nucleophiles. The catalytic cycle is sustained by the regeneration of the ground-state iridium species, ensuring that the reaction proceeds with high atom economy and minimal catalyst loading, typically around 5 mol%. This mechanism avoids the formation of harsh byproducts associated with stoichiometric oxidants, thereby preserving the integrity of sensitive functional groups present on the R1 and R2 substituents of the sulfone diimine core.
From an impurity control perspective, the mildness of the photocatalytic conditions significantly reduces the risk of side reactions such as over-oxidation or decomposition of the sulfone diimine backbone. Traditional oxidative methods often lead to complex impurity profiles due to the non-selective nature of strong oxidants like chloramine or hypochlorite, necessitating rigorous and costly purification steps. In this photocatalytic system, the selectivity is governed by the redox potentials of the excited catalyst and the specific substrates, leading to a cleaner reaction profile that simplifies downstream processing. The ability to tolerate diverse substituents, including halogens and alkoxy groups, without dehalogenation or ether cleavage further underscores the precision of this radical-mediated pathway. Consequently, manufacturers can achieve higher purity specifications with fewer recrystallization or chromatography steps, directly translating to improved process efficiency and reduced production costs for high-value intermediates.
How to Synthesize N-Substituted Sulfone Diimine Efficiently
To implement this cutting-edge methodology in a laboratory or pilot plant setting, operators must adhere to specific protocols regarding reagent mixing and light exposure to ensure optimal conversion rates. The process begins with the precise weighing of the sulfone diimine compound, the chosen bromide coupling partner, an inorganic base, and the iridium photocatalyst, which are then sealed in a reaction tube under an inert gas atmosphere to prevent oxygen quenching of the excited catalyst state.
- Combine sulfone diimine compound, bromide substrate, inorganic base (e.g., K2CO3), and iridium photocatalyst in a closed reaction tube under inert atmosphere.
- Add aprotic solvent such as THF and irradiate the mixture with a 5W white light lamp at room temperature for 6 to 24 hours.
- Upon completion, remove the solvent and purify the crude product via silica gel column chromatography to isolate the target N-substituted sulfone diimine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers tangible strategic benefits centered around cost stability and operational flexibility. The elimination of hazardous and expensive stoichiometric oxidants drastically simplifies the raw material sourcing strategy, reducing reliance on volatile chemical markets for reagents like chloramine-T or tert-butyl hypochlorite. Moreover, the ability to run reactions at room temperature using low-energy LED light sources significantly lowers the utility costs associated with heating or cooling large-scale reactors, contributing to substantial overall cost reduction in fine chemical manufacturing. The robustness of the method across a wide range of substrates means that a single standardized protocol can be adapted for multiple product lines, enhancing manufacturing agility and reducing the need for specialized equipment or extensive operator training.
- Cost Reduction in Manufacturing: The transition from stoichiometric oxidants to a catalytic system fundamentally alters the cost structure of producing N-substituted sulfone diimines by minimizing reagent consumption and waste disposal fees. Since the iridium catalyst is used in small catalytic quantities and the reaction does not require energy-intensive thermal inputs, the variable costs per kilogram of product are significantly optimized. Additionally, the simplified post-treatment process, which often involves basic filtration and chromatography rather than complex neutralization and extraction sequences, reduces labor hours and solvent usage, further driving down the total cost of ownership for these critical intermediates.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials such as simple bromides and inorganic bases, this method mitigates the risk of supply disruptions often associated with specialized or regulated oxidizing agents. The broad substrate scope ensures that if one specific building block becomes unavailable, alternative analogs can often be synthesized using the same established protocol without requiring extensive re-validation of the process. This flexibility allows supply chain planners to maintain continuous production schedules and meet tight delivery deadlines for pharmaceutical clients who require consistent quality and uninterrupted flow of key intermediates for their own drug development pipelines.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metal waste or corrosive byproducts make this process inherently safer and easier to scale from gram to multi-kilogram quantities. Regulatory compliance is streamlined as the process generates less hazardous waste, aligning with increasingly stringent environmental protection standards and corporate sustainability goals. The use of visible light as the energy source is a green chemistry principle that enhances the company's environmental profile, potentially qualifying the manufacturing site for green incentives and improving relationships with eco-conscious stakeholders and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this photocatalytic synthesis method, providing clarity for technical teams evaluating its adoption.
Q: What are the primary advantages of this photocatalytic method over traditional oxidation routes?
A: Unlike traditional methods requiring harsh oxidants like tert-butyl hypochlorite or chloramine-T which limit substrate scope, this photocatalytic approach operates at room temperature with visible light, enabling the synthesis of sensitive S-alkyl and S-aryl substituted derivatives with higher functional group tolerance.
Q: Which substrates are compatible with this iridium-catalyzed N-substitution reaction?
A: The method demonstrates broad applicability, successfully accommodating aromatic groups, five-membered or six-membered heterocycles (such as pyridine and thiophene), and various alkyl chains including benzyl and allyl groups, although steric hindrance from tert-butyl groups may inhibit reaction efficiency.
Q: Is the iridium photocatalyst reusable or required in stoichiometric amounts?
A: The process utilizes the iridium catalyst in catalytic amounts, typically around 0.05 mmol relative to 1 mmol of substrate (5 mol%), making it economically viable for scale-up compared to stoichiometric reagent consumption in older methodologies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted Sulfone Diimine Supplier
As the global demand for sulfur-containing pharmaceutical intermediates continues to rise, partnering with a manufacturer that possesses advanced synthetic capabilities is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM leverages deep expertise in photocatalytic technologies to deliver high-purity N-substituted sulfone diimines that meet the rigorous standards of the international pharmaceutical industry. Our facility is equipped with state-of-the-art photo-reactors and rigorous QC labs, ensuring that every batch produced adheres to stringent purity specifications and is free from the impurities typical of older oxidative methods. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that we can support your needs from early-stage clinical trials through to full-scale commercial manufacturing.
We invite you to collaborate with our technical team to explore how this innovative photocatalytic route can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into how switching to our advanced manufacturing process can reduce your overall procurement costs while improving material quality. Please contact our technical procurement team today to索取 specific COA data and route feasibility assessments tailored to your project's unique molecular targets, ensuring a seamless integration of our high-performance intermediates into your drug development workflow.
