Revolutionizing Buprenorphine Production: A Scalable Total Synthesis Route for Global Pharma Supply Chains
The pharmaceutical industry is currently witnessing a paradigm shift in the production of critical opioid analgesics, driven by the urgent need for supply chain resilience and cost efficiency. Patent CN113956263B discloses a groundbreaking total synthesis method for Buprenorphine, a morphine derivative essential for pain management and addiction treatment. Unlike traditional semi-synthetic routes that depend heavily on the agricultural extraction of thebaine from poppies, this innovative approach utilizes a biomimetic strategy centered on an intramolecular oxidative dearomatization Heck reaction. This technological leap allows for the construction of the complex morphine skeleton from simple, commercially available starting materials, effectively bypassing the geopolitical and agricultural vulnerabilities associated with opium poppy cultivation. For global procurement leaders, this represents a significant opportunity to secure a more stable and predictable source of high-purity active pharmaceutical ingredients.
Furthermore, the strategic implementation of this synthesis pathway addresses the growing regulatory scrutiny surrounding narcotic raw materials. By shifting the production basis from plant extraction to chemical synthesis, manufacturers can mitigate the risks of crop failure, climate variability, and strict international control measures that often plague the traditional supply chain. The patent outlines a concise 15-step sequence from key intermediates, achieving a total yield of 12% with exceptional stereochemical control. This level of efficiency in total synthesis was previously considered challenging for such complex molecules, marking a substantial advancement in process chemistry. As a reliable buprenorphine supplier, understanding these underlying technological improvements is crucial for evaluating long-term partnership viability and ensuring continuous availability for downstream formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Buprenorphine and related morphine derivatives has been inextricably linked to the cultivation of Papaver somniferum, commonly known as the opium poppy. This agricultural dependency introduces a multitude of inefficiencies and risks that compromise the stability of the global pharmaceutical supply chain. The extraction of thebaine, the primary precursor for semi-synthesis, requires vast amounts of arable land and is highly susceptible to environmental factors such as drought, pests, and political instability in growing regions. Moreover, the legal framework governing poppy cultivation is extremely rigid, leading to complex licensing procedures and potential supply disruptions that can ripple through the entire manufacturing network. From a cost perspective, the multi-step semi-synthetic conversion from thebaine often involves cumbersome protection and deprotection strategies, particularly the difficult removal of the nitrogen methyl group, which adds significant operational complexity and waste generation.
In addition to supply volatility, the conventional extraction-based model faces increasing pressure from sustainability mandates and quality consistency requirements. Variations in the alkaloid content of poppy straw can lead to batch-to-batch inconsistencies in the starting material, necessitating rigorous and costly quality control measures to ensure the final API meets pharmacopeial standards. The reliance on natural products also limits the ability to rapidly scale production in response to sudden spikes in market demand, such as those seen during public health crises. Furthermore, the environmental footprint of large-scale agriculture, combined with the chemical processing required for extraction, creates a sustainability challenge that modern pharmaceutical companies are increasingly eager to resolve. These structural limitations highlight the critical need for a robust, fully synthetic alternative that can deliver cost reduction in opioid analgesic manufacturing without compromising on quality or compliance.
The Novel Approach
The novel synthetic route described in patent CN113956263B offers a transformative solution by constructing the morphine core structure through rational chemical design rather than natural extraction. At the heart of this methodology is the intramolecular oxidative dearomatization Heck reaction, which serves as the key step to establish the complex polycyclic framework with high precision. This reaction allows for the efficient conversion of simpler precursors into thebaine analogues, effectively mimicking the biogenic pathway but under controlled laboratory conditions. By eliminating the need for thebaine as a starting material, this approach decouples production from agricultural cycles, providing a consistent and scalable source of intermediates. The streamlined nature of the synthesis reduces the number of purification steps required, thereby minimizing solvent usage and waste disposal costs, which aligns perfectly with green chemistry principles and corporate sustainability goals.
Moreover, this new approach significantly enhances the feasibility of commercial scale-up of complex pharmaceutical intermediates by utilizing readily available reagents and mild reaction conditions. The process incorporates advanced asymmetric catalysis techniques, such as asymmetric transfer hydrogenation, to ensure high enantiomeric excess without the need for resolution steps that typically halve the yield. The ability to introduce functional groups like the tert-butyl and cyclopropylmethyl moieties at later stages allows for greater flexibility in process optimization and impurity control. For supply chain directors, this translates to a more resilient manufacturing pipeline capable of adapting to market fluctuations with shorter lead times. The overall result is a production method that not only matches but potentially exceeds the economic and operational metrics of traditional extraction, positioning it as the future standard for high-purity opioid APIs.
Mechanistic Insights into Intramolecular Oxidative Dearomatization Heck Reaction
The cornerstone of this synthetic breakthrough is the intramolecular oxidative dearomatization Heck reaction, which facilitates the construction of the quaternary carbon center and the fused ring system characteristic of the morphine skeleton. This transformation involves the palladium-catalyzed coupling of an aryl halide with an alkene moiety within the same molecule, driven by an oxidative process that restores aromaticity or establishes the desired oxidation state. The reaction conditions are meticulously optimized, utilizing specific phosphine ligands and palladium sources to achieve yields as high as 82% for this critical step. The choice of ligand, such as bulky alkylphosphines, plays a pivotal role in stabilizing the active catalytic species and promoting the reductive elimination step, which is often the rate-determining factor in such complex cyclizations. Understanding this mechanism is vital for R&D directors aiming to replicate or further optimize the process for GMP manufacturing environments.
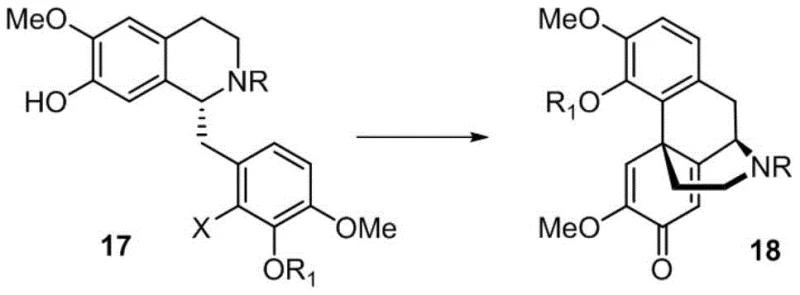
Complementing the Heck reaction is the implementation of asymmetric transfer hydrogenation, which ensures the precise installation of chiral centers essential for the biological activity of Buprenorphine. This step utilizes chiral diamine ligands coordinated with ruthenium or rhodium catalysts to transfer hydrogen from a donor like formic acid to the imine intermediate with high stereoselectivity. The patent reports achieving enantiomeric excess values of up to 99.9% after recrystallization, demonstrating the robustness of this chiral induction strategy. The mechanism involves a metal-ligand bifunctional catalysis where both the metal center and the ligand participate in the activation of the substrate and the hydrogen donor. This high level of stereocontrol eliminates the need for costly chiral separation processes, directly contributing to the overall efficiency and cost-effectiveness of the route. For technical teams, mastering these mechanistic nuances is key to maintaining batch consistency and meeting the stringent purity specifications required for regulatory approval.
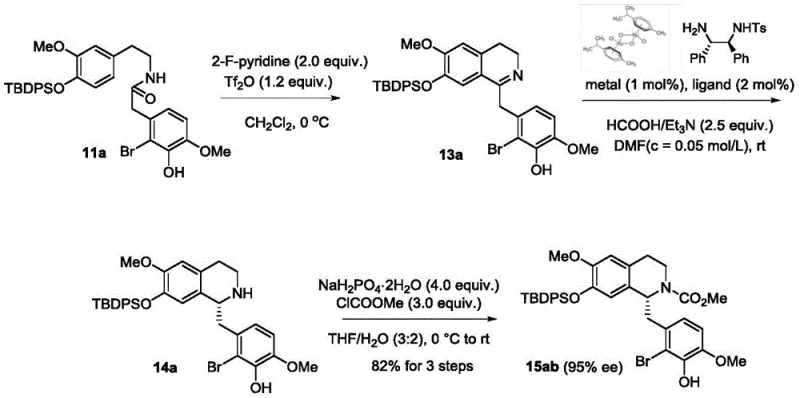
How to Synthesize Buprenorphine Efficiently
The practical execution of this synthesis involves a series of well-defined transformations that convert simple aromatic starting materials into the final active pharmaceutical ingredient. The process begins with the preparation of key intermediates through condensation and cyclization reactions, setting the stage for the critical Heck coupling. Following the establishment of the core skeleton, the route proceeds through a Diels-Alder reaction with butenone to introduce the necessary six-membered ring, followed by catalytic hydrogenation to saturate the double bonds. Subsequent steps involve the strategic addition of a tert-butyl group using a Grignard reagent and the introduction of the cyclopropylmethyl group via reductive amination or alkylation. The detailed standardized synthesis steps see the guide below for specific reaction conditions and stoichiometry.
- Prepare compound 17 and subject it to intramolecular oxidative dearomatization Heck reaction to generate compound 18.
- Perform Diels-Alder reaction with butenone to generate compound 22, followed by catalytic hydrogenation.
- Introduce tert-butyl group via Grignard reagent, remove protecting groups, and introduce cyclopropylmethyl to obtain Buprenorphine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this total synthesis route offers compelling advantages that extend beyond mere technical novelty. The primary benefit lies in the drastic simplification of the raw material sourcing strategy, moving away from volatile agricultural commodities to stable chemical feedstocks. This shift significantly mitigates the risk of supply interruptions caused by crop failures or regulatory changes in poppy-growing regions, ensuring a more reliable buprenorphine supplier relationship. Additionally, the reduction in the number of processing steps and the elimination of cumbersome deprotection sequences lead to substantial cost savings in manufacturing overheads. The ability to produce high-purity intermediates with minimal purification requirements further reduces waste disposal costs and environmental compliance burdens, aligning with modern ESG (Environmental, Social, and Governance) criteria.
- Cost Reduction in Manufacturing: The synthetic route eliminates the expensive and inefficient nitrogen methyl removal steps typical of semi-synthesis, leading to significant operational cost reductions. By utilizing common chemical reagents and avoiding specialized natural extracts, the overall cost of goods sold is optimized, allowing for more competitive pricing strategies in the global market. The high yields achieved in key steps, such as the 92% yield in the Diels-Alder reaction, contribute to better material utilization and lower waste generation. This efficiency translates directly into improved profit margins and the ability to invest in further process improvements or capacity expansion.
- Enhanced Supply Chain Reliability: Decoupling production from agricultural cycles ensures a consistent and predictable supply of Buprenorphine intermediates throughout the year. This stability is crucial for maintaining uninterrupted production schedules for downstream formulations, reducing the need for excessive safety stock and inventory holding costs. The use of commercially available starting materials means that sourcing can be diversified across multiple chemical suppliers, reducing dependency on single-source vendors. This resilience is particularly valuable in times of global disruption, ensuring that patient access to essential pain medication is maintained without compromise.
- Scalability and Environmental Compliance: The mild reaction conditions and straightforward workup procedures make this process highly amenable to scale-up from pilot plant to commercial tonnage production. The reduction in solvent usage and hazardous waste generation simplifies environmental permitting and compliance, reducing the regulatory burden on manufacturing sites. The process design inherently supports continuous manufacturing technologies, which can further enhance throughput and quality control. For supply chain planners, this scalability means the ability to rapidly respond to market demand surges without the long lead times associated with expanding agricultural capacity.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding the technical feasibility and commercial viability of this new synthetic method. They are derived from the specific technical advantages and process parameters detailed in the patent documentation. Understanding these aspects helps stakeholders make informed decisions about integrating this technology into their existing supply networks. The answers reflect the current state of the art as described in the intellectual property, providing a clear picture of the expected performance and benefits.
Q: How does this total synthesis method improve supply chain stability compared to traditional extraction?
A: Traditional methods rely on poppy cultivation, which is subject to agricultural risks and regulatory controls. This total synthesis route uses commercially available chemical raw materials, ensuring consistent supply independent of crop yields.
Q: What is the optical purity achieved in this synthetic route?
A: The process utilizes asymmetric transfer hydrogenation and recrystallization steps to achieve an optical purity of up to 99.9% ee, meeting stringent pharmaceutical standards.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild, and the reagents are commonly available. The process avoids cumbersome nitrogen methyl removal steps, facilitating easier scale-up and cost reduction in manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Buprenorphine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting advanced synthetic routes to secure the future of pharmaceutical supply chains. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN113956263B can be successfully transferred to industrial scale. We are committed to delivering high-purity buprenorphine intermediate and API solutions that meet stringent purity specifications and rigorous QC labs standards. Our state-of-the-art facilities are equipped to handle complex organic syntheses, including sensitive catalytic reactions and cryogenic processes, guaranteeing consistent quality and reliability for our global partners.
We invite you to collaborate with us to explore the full potential of this cost-effective and sustainable synthesis method. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your supply chain resilience. By partnering with us, you gain access to cutting-edge technology and a dedicated support team focused on driving value and efficiency in your opioid analgesic manufacturing operations.
