Advanced Synthesis of Polysubstituted Enamine Aldehydes for Commercial Pharmaceutical Applications
Introduction to Novel Enamine Aldehyde Technology
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for more efficient and cost-effective pathways to complex molecular scaffolds. Patent CN109232284B introduces a groundbreaking methodology for the preparation of polysubstituted enamine aldehyde derivatives, a class of compounds pivotal in the development of antimalarial, antitumor, and hypoglycemic agents. This technology addresses critical bottlenecks in traditional synthesis by utilizing a robust acid-catalyzed protocol that operates under mild低温 conditions. By leveraging stable acrolein derivatives and azide precursors, this invention circumvents the instability issues associated with malondialdehyde and the high costs of noble metal catalysts. For R&D teams seeking reliable access to these versatile building blocks, understanding the nuances of this patented route is essential for optimizing supply chains and reducing time-to-market for new drug candidates.
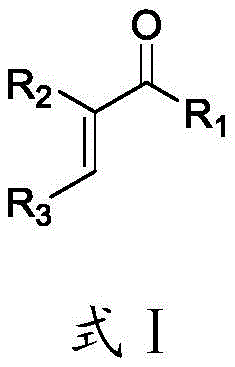
The structural versatility offered by this method is exemplified by the broad range of substituents tolerated at the R1, R2, and R3 positions, allowing for the rapid generation of diverse chemical libraries. This capability is crucial for medicinal chemists engaged in structure-activity relationship (SAR) studies where subtle changes in molecular architecture can dramatically impact biological efficacy. Furthermore, the presence of a hydrogen atom on the amine group in the final product facilitates further downstream functionalization, a feature often lacking in competing synthetic strategies. As a leading entity in the fine chemical sector, recognizing the potential of such innovations allows us to better serve our partners in the global pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of enamine aldehydes has been plagued by significant technical hurdles that limit their widespread application in large-scale manufacturing. Traditional approaches often rely on the condensation of malondialdehyde with amines; however, malondialdehyde is notoriously unstable and prone to polymerization, making it difficult to handle and store safely in an industrial setting. Another common pathway involves the reaction of alkynyl aldehydes with amines, but the preparation of these alkynyl precursors is itself challenging and often yields unstable intermediates that degrade before the final coupling can occur. Perhaps most critically, existing gold-catalyzed methods, while effective for certain substrates, fail to produce alpha-substituted enaminoaldehydes and often result in products lacking the necessary hydrogen on the amine nitrogen, thereby restricting their utility in subsequent chemical transformations. These limitations collectively create a bottleneck for procurement managers seeking consistent, high-quality supplies of these key intermediates.
The Novel Approach
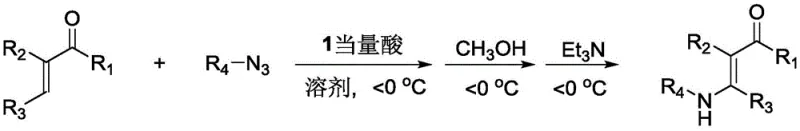
The methodology disclosed in the patent represents a paradigm shift by employing a direct reaction between acrolein derivatives and azide compounds mediated by strong acids. This approach eliminates the need for unstable malondialdehyde or difficult-to-prepare alkynals, relying instead on commercially available and stable starting materials. The use of strong acids such as trifluoromethanesulfonic acid or boron trifluoride etherate provides a highly active catalytic environment that drives the reaction to completion rapidly, often within 10 to 60 minutes. Moreover, the inclusion of a methanol quenching step followed by triethylamine neutralization ensures a clean reaction profile with minimal byproduct formation. This streamlined process not only enhances the overall yield but also simplifies the post-reaction workup, making it an attractive option for cost reduction in pharmaceutical intermediate manufacturing where efficiency is paramount.
Mechanistic Insights into Acid-Catalyzed Azide-Aldehyde Coupling
The core of this innovation lies in the unique mechanistic pathway facilitated by the strong acid catalyst, which activates the acrolein derivative towards nucleophilic attack by the azide species. Under low-temperature conditions (typically below 0°C), the acid protonates the carbonyl oxygen or coordinates with the double bond, increasing the electrophilicity of the beta-carbon. This activation allows the azide ion to attack selectively, forming a transient intermediate that subsequently undergoes rearrangement and elimination of nitrogen gas to yield the enamine structure. The precise control of temperature is critical here, as it prevents side reactions such as polymerization of the acrolein or decomposition of the azide, ensuring high selectivity for the desired E-isomer. Understanding this mechanism allows process chemists to fine-tune reaction parameters for optimal performance across different substrate classes.
Impurity control is another vital aspect of this mechanism, particularly regarding the suppression of oligomerization and over-reaction. The rapid consumption of the activated acrolein by the azide minimizes the residence time of reactive intermediates, thereby reducing the formation of polymeric tars that often complicate purification. Additionally, the choice of solvent, such as dichloromethane or acetonitrile, plays a significant role in stabilizing the transition states and solubilizing the ionic intermediates generated during the acid catalysis. The final neutralization step with triethylamine effectively quenches any remaining acid activity, preventing acid-catalyzed degradation of the sensitive enamine aldehyde product during isolation. This robust mechanistic framework ensures that the final product meets stringent purity specifications required for GMP manufacturing environments.
How to Synthesize Polysubstituted Enamine Aldehyde Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the sequential addition of reagents and temperature management to ensure safety and reproducibility. The process begins with the dissolution of the acrolein derivative and azide compound in a suitable anhydrous solvent, followed by the slow addition of the strong acid catalyst while maintaining the reaction mixture at sub-zero temperatures. Detailed standard operating procedures for this transformation, including specific molar ratios and quenching protocols, are outlined in the comprehensive guide below to assist technical teams in replicating these high-yielding results.
- React acrolein derivatives with azide compounds and strong acid in solvent at below 0°C for 10-60 minutes.
- Add methanol to the reaction mixture and continue stirring for 10-120 minutes before quenching with triethylamine.
- Dilute with dichloromethane, wash with water, dry the organic phase, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial benefits that directly address the pain points of procurement managers and supply chain directors in the pharmaceutical sector. By replacing expensive noble metal catalysts with inexpensive strong acids, the direct material costs associated with catalysis are drastically reduced, leading to significant overall cost savings in the production of these high-value intermediates. Furthermore, the stability of the raw materials—acrolein derivatives and organic azides—ensures a reliable supply chain that is less susceptible to the volatility often seen with specialized reagents like malondialdehyde. This reliability translates into shorter lead times and greater predictability in delivery schedules, which is critical for maintaining continuous manufacturing operations.
- Cost Reduction in Manufacturing: The elimination of gold or other transition metal catalysts removes the need for costly metal scavenging steps and reduces the burden of heavy metal residue testing, which is a major regulatory hurdle in API production. The use of commodity chemicals like trifluoromethanesulfonic acid and triethylamine further drives down the bill of materials, allowing for more competitive pricing structures without compromising on quality. Additionally, the high yields reported in the patent examples mean less raw material waste and higher throughput per batch, maximizing the return on investment for manufacturing facilities.
- Enhanced Supply Chain Reliability: Sourcing stable acrolein derivatives and azides is significantly easier than procuring unstable alkynals or malondialdehyde, reducing the risk of production delays due to raw material shortages. The robustness of the reaction conditions allows for flexibility in sourcing, as the process tolerates a wide range of substituents, meaning alternative suppliers for starting materials can be qualified more easily. This diversification of the supply base strengthens the overall resilience of the supply chain against market fluctuations and geopolitical disruptions.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving standard extraction and column chromatography, facilitates easy scale-up from gram to kilogram scales without requiring specialized equipment. The absence of heavy metals simplifies waste treatment protocols, aligning with increasingly stringent environmental regulations and reducing the cost of waste disposal. The short reaction times also contribute to energy efficiency, as less heating or prolonged cooling is required compared to traditional multi-step syntheses, supporting sustainability goals in chemical manufacturing.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled a list of frequently asked questions based on the patent data and industry standards. These insights are designed to help decision-makers evaluate the feasibility of integrating this technology into their existing workflows and to understand the specific advantages it holds over legacy methods. Addressing these common queries ensures transparency and builds confidence in the technical viability of the process.
Q: What are the advantages of this acid-catalyzed method over traditional gold catalysis?
A: Unlike gold-catalyzed methods which often require expensive metals and complex ligands, this process utilizes low-cost strong acids like trifluoromethanesulfonic acid, significantly reducing catalyst costs and simplifying metal removal steps.
Q: Can this method produce alpha-substituted enaminoaldehydes effectively?
A: Yes, the patent explicitly demonstrates the successful synthesis of alpha-substituted derivatives which are difficult to obtain via malondialdehyde routes due to stability issues, offering a broader substrate scope.
Q: What are the typical reaction conditions required for high yield?
A: The reaction requires strict temperature control below 0°C (typically -40°C) during the initial addition, followed by methanol treatment and neutralization, ensuring high conversion rates up to 98%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Enamine Aldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN109232284B for accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can optimize your supply chain and reduce your overall production costs.
