Advanced Synthesis of Acylpyridinehydrazine Difluoroboron Dyes for High-Performance OLEDs
Introduction to Next-Generation Fluorescent Materials
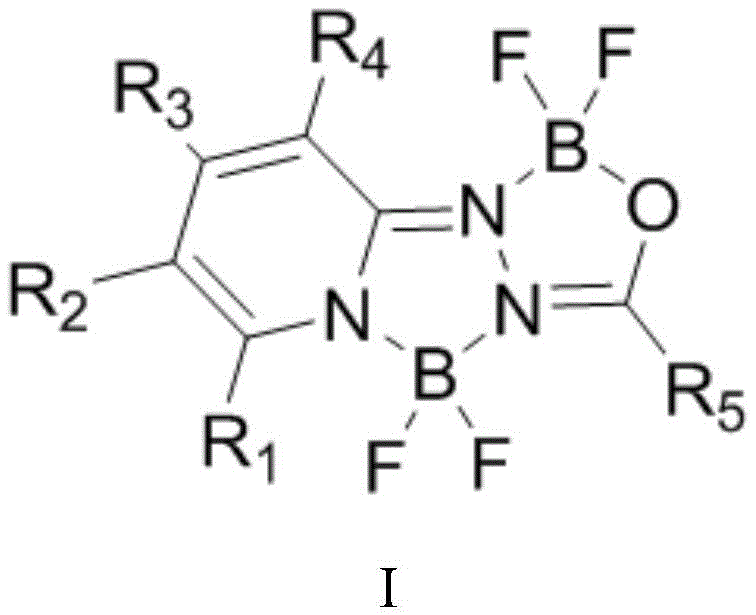
The landscape of organic optoelectronics is continuously evolving, driven by the demand for materials with superior photophysical properties. A significant breakthrough in this domain is documented in patent CN109438487B, which discloses a novel class of acylpyridinehydrazine difluoroboron strong fluorescent dyes. These compounds represent a strategic advancement over traditional fluorophores, offering a unique molecular architecture that combines high fluorescence quantum yields with exceptional photostability. The core innovation lies in the integration of a boron-nitrogen-oxygen coordination system within a rigid pyridine-hydrazine framework, creating a planar structure that facilitates efficient electron transport and light emission. This technology is particularly relevant for manufacturers seeking reliable electronic chemical suppliers capable of delivering high-purity OLED materials with tunable emission spectra.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-performance organoboron dyes, such as the widely known BODIPY family, has often involved multi-step sequences requiring stringent anhydrous conditions and expensive starting materials. Traditional routes frequently rely on the condensation of pyrrole aldehydes, which can suffer from limited commercial availability and high cost variability. Furthermore, the introduction of the boron center typically requires harsh Lewis acids and subsequent purification steps to remove metal impurities that can quench fluorescence. These conventional methodologies often result in lower overall yields due to cumulative losses at each isolation stage, and the structural rigidity required for high quantum efficiency is difficult to achieve without complex cyclization protocols. For procurement managers, these factors translate into higher raw material costs and extended lead times for critical intermediates.
The Novel Approach
In contrast, the methodology outlined in the patent introduces a streamlined 'one-pot' strategy that fundamentally simplifies the manufacturing workflow. By utilizing readily commercialized 2-hydrazinopyridine derivatives and reacting them directly with accessible acid chlorides or acid anhydrides, the process bypasses the need for specialized aldehyde precursors. The subsequent in-situ coordination with boron trifluoride etherate eliminates the isolation of unstable intermediates, thereby reducing solvent consumption and waste generation. This approach not only accelerates the reaction timeline but also enhances the reproducibility of the final product's optical characteristics. For supply chain heads, this translates to a more robust production model where raw material sourcing is decoupled from niche chemical suppliers, ensuring greater continuity and cost reduction in electronic chemical manufacturing.
Mechanistic Insights into One-Pot Fluoroboron Coordination
The chemical elegance of this synthesis lies in its two-stage mechanistic pathway occurring within a single reaction vessel. Initially, the nucleophilic nitrogen of the 2-hydrazinopyridine derivative attacks the electrophilic carbonyl carbon of the acid chloride or anhydride. This acylation step forms an amide-like intermediate (M1), establishing the foundational carbon-nitrogen backbone of the dye. The reaction is typically conducted at mild temperatures ranging from -20°C to 30°C to control exothermicity and prevent side reactions. Following this, the addition of a base, such as triethylamine or diisopropylethylamine, serves to scavenge the hydrochloric acid byproduct, driving the equilibrium forward and protecting the sensitive hydrazine moiety from degradation. This careful pH management is crucial for maintaining the integrity of the intermediate prior to cyclization.
The second critical phase involves the introduction of boron trifluoride etherate under elevated thermal conditions, typically between 80°C and 120°C. At this stage, the Lewis acidic boron atom coordinates with the nitrogen and oxygen atoms of the intermediate, inducing a cyclization that locks the molecular conformation into a rigid, planar geometry. This rigidification is the key to the dye's high fluorescence quantum yield, as it restricts non-radiative decay pathways caused by molecular vibration. The resulting difluoroboron complex exhibits a large Stokes shift and high molar absorption coefficients, making it ideal for applications requiring distinct signal separation. Understanding this mechanism allows R&D directors to predict how varying substituents (R1-R5) will influence the electronic density and, consequently, the emission color of the final dye.
How to Synthesize Acylpyridinehydrazine Difluoroboron Dye Efficiently
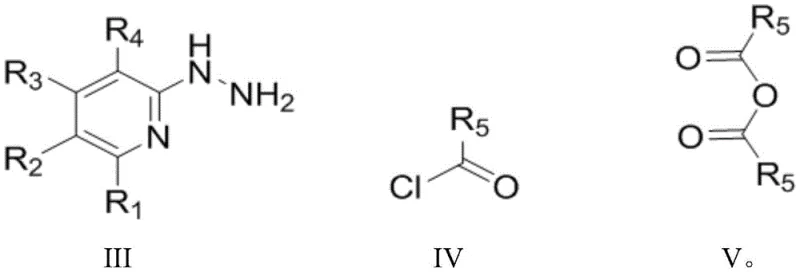
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the dissolution of the hydrazine precursor in a chlorinated solvent like 1,2-dichloroethane, followed by the controlled addition of the acylating agent. After the initial condensation and base treatment, the reaction mixture is concentrated before the addition of the boron source. The final heating step promotes the coordination chemistry necessary to form the fluorescent core. Detailed standardized synthetic steps see the guide below.
- Condense 2-hydrazinopyridine derivatives with substituted acid chlorides or anhydrides in a solvent like 1,2-dichloroethane at temperatures between -20°C and 30°C.
- Treat the intermediate mixture with an organic base such as triethylamine or diisopropylethylamine to neutralize acid byproducts.
- Add boron trifluoride etherate and heat the mixture to 80-120°C to facilitate cyclization and fluoroboron coordination, yielding the final fluorescent dye.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patent offers substantial value by addressing key pain points in the supply of specialty fluorescent dyes. The reliance on commodity chemicals rather than bespoke intermediates significantly de-risks the supply chain, ensuring that production is not bottlenecked by the availability of a single obscure reagent. The simplified workflow reduces the operational complexity associated with multi-step syntheses, leading to lower labor and utility costs per kilogram of product. Furthermore, the high purity achievable through standard recrystallization techniques minimizes the need for expensive chromatographic purification on a large scale, directly impacting the bottom line.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of inexpensive acid chlorides drastically reduce the bill of materials. By avoiding precious metal catalysts, the process removes the need for costly metal scavenging steps, which are often required to meet the stringent purity specifications of the electronics industry. This qualitative shift in reagent selection ensures that the cost of goods sold remains competitive even as production volumes scale up.
- Enhanced Supply Chain Reliability: Since the starting materials, such as 2-hydrazinopyridine and various benzoyl chlorides, are widely produced for other pharmaceutical and agrochemical applications, the risk of supply disruption is minimized. This ubiquity allows for flexible sourcing strategies, enabling procurement teams to negotiate better terms and secure long-term contracts with multiple vendors. The robustness of the 'one-pot' method also means that production schedules are less susceptible to delays caused by intermediate isolation failures.
- Scalability and Environmental Compliance: The process utilizes common organic solvents that are easily recovered and recycled, aligning with modern green chemistry principles. The absence of heavy metal waste streams simplifies effluent treatment and regulatory compliance, reducing the environmental overhead associated with manufacturing. This scalability ensures that the technology can be seamlessly transferred from laboratory benchtop to multi-ton commercial reactors without significant re-engineering of the process infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced fluorophores. The answers are derived from the specific embodiments and data presented in the patent documentation, providing clarity on performance metrics and process feasibility.
Q: What are the key advantages of this difluoroboron dye synthesis?
A: The method utilizes readily available commercial reagents and a simplified one-pot procedure, eliminating complex multi-step purification and reducing overall production costs while maintaining high fluorescence quantum yields.
Q: Can the optical properties of these dyes be tuned?
A: Yes, the structure allows for extensive modification via substituents R1-R5, enabling precise tuning of absorption and emission wavelengths for specific OLED or sensor applications.
Q: Is this process scalable for industrial production?
A: The use of common solvents like dichloromethane or toluene and standard reagents like acid chlorides suggests high scalability, avoiding exotic catalysts that hinder large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acylpyridinehydrazine Difluoroboron Dye Supplier
As the demand for high-efficiency OLED materials continues to surge, partnering with an experienced CDMO is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our rigorous QC labs and adherence to stringent purity specifications guarantee that every batch of fluorescent dye meets the exacting standards required for next-generation display technologies. We understand the critical nature of material consistency in optoelectronics and are committed to delivering products that perform reliably in your devices.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall material costs. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the suitability of these novel dyes for your application pipeline.
