Advanced Triptycene-Based Linear Acene Trimers for High-Efficiency Organic Photoelectric Applications
Introduction to Next-Generation Organic Photoelectric Materials
The rapid evolution of the organic electronics sector demands materials that can transcend traditional efficiency limits while maintaining manufacturability. Patent CN112299982B introduces a groundbreaking class of triptycene-based linear acene compounds designed specifically to address these challenges. Unlike conventional planar acenes that suffer from severe aggregation-caused quenching, these novel structures utilize a rigid triptycene scaffold to create C3-symmetric trimers. This unique architecture not only enhances solubility and processability but also facilitates efficient intramolecular singlet fission, a critical mechanism for breaking the Shockley-Queisser limit in photovoltaic applications. The strategic placement of linear acene wings around a central three-dimensional core allows for precise tuning of electronic properties without sacrificing structural integrity.
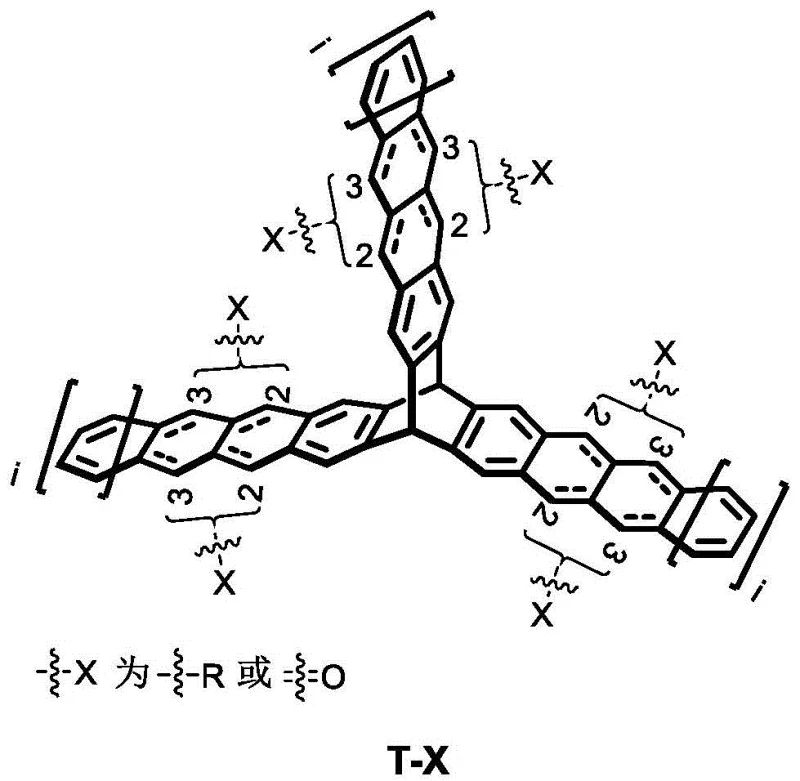
For R&D directors and procurement specialists, this technology represents a significant leap forward in high-purity OLED material and solar cell intermediate development. The patent details a versatile synthetic platform capable of generating a wide library of derivatives by varying the length of the acene units and the nature of the peripheral substituents. This modularity ensures that manufacturers can tailor materials for specific energy levels required in diverse device architectures, from bulk heterojunction solar cells to high-mobility transistor channels. The ability to control the substitution pattern at the 2- or 3-positions of the acene rings further expands the chemical space available for optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing linear acenes often rely on stepwise coupling reactions that are plagued by low yields and poor scalability. Conventional dimerization strategies frequently result in uncontrolled polymerization or the formation of insoluble aggregates that are difficult to purify. Furthermore, planar acene molecules tend to stack tightly in the solid state, leading to excimer formation that dissipates excited state energy as heat rather than useful charge carriers. This phenomenon severely limits the external quantum efficiency of devices fabricated from such materials. Additionally, many existing synthetic routes require expensive palladium catalysts or harsh conditions that introduce metallic impurities, necessitating costly purification steps that erode profit margins in electronic chemical manufacturing.
The Novel Approach
The methodology disclosed in CN112299982B overcomes these hurdles by employing a convergent synthesis strategy centered on the triptycene nucleus. By building three acene arms simultaneously from a single core, the process inherently ensures C3 symmetry and reduces the number of synthetic steps compared to linear assembly. The use of Friedel-Crafts acylation followed by acid-mediated cyclization provides a robust pathway to the core ketone structures with high atom economy. This approach minimizes the generation of hazardous waste and eliminates the need for precious metal catalysts. The resulting three-dimensional geometry prevents close packing, thereby preserving the intrinsic photophysical properties of the acene units. This structural innovation directly translates to improved film morphology and device stability, addressing key pain points for supply chain managers focused on commercial scale-up of complex polymer additives and small molecule semiconductors.
Mechanistic Insights into Friedel-Crafts Acylation and Cyclization
The core of this synthetic innovation lies in the sequential Friedel-Crafts reactions that construct the acene framework. In the first stage, triptycene reacts with an excess of acene-type phthalic anhydride in the presence of a Lewis acid, typically aluminum trichloride. The reaction is conducted in high-boiling solvents like tetrachloroethylene at temperatures ranging from 100°C to 180°C. This thermal energy is crucial for overcoming the steric hindrance associated with the bulky triptycene core, ensuring that acylation occurs selectively at the desired positions. The use of a significant molar excess of the anhydride drives the equilibrium towards the tri-substituted intermediate, maximizing the utilization of the valuable triptycene starting material.
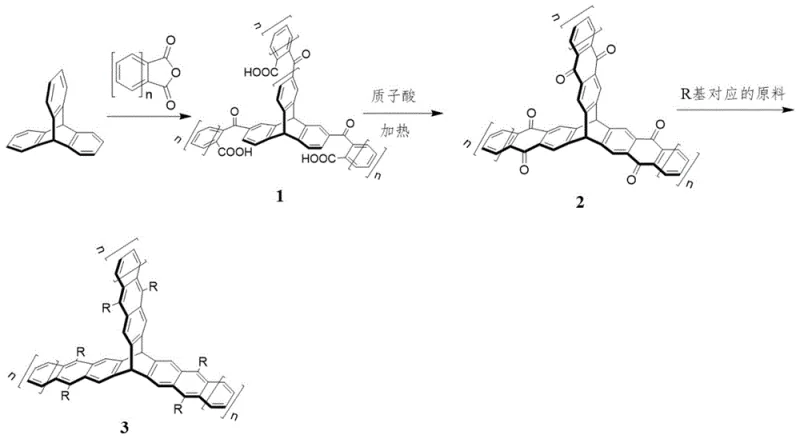
Following the initial acylation, the intermediate undergoes an intramolecular cyclization in concentrated sulfuric acid at approximately 120°C. This step is critical for forming the fused ring system that extends the conjugation length. The protonic acid environment facilitates the dehydration and aromatization processes required to lock the acene structure into place. For derivatives requiring functionalization at the 3-position, an alternative pathway involving bromination and Diels-Alder chemistry is employed. This route utilizes hexabromotriptycene as a precursor, which reacts with furan to form a masked diene system. Subsequent retro-Diels-Alder reactions with benzoquinones allow for the precise installation of acene units with different substitution patterns. This mechanistic flexibility allows chemists to fine-tune the HOMO-LUMO gap, a vital parameter for optimizing cost reduction in electronic chemical manufacturing by matching material properties to device requirements without extensive trial and error.
How to Synthesize Triptycene-Based Linear Acene Efficiently
The synthesis of these advanced materials follows a logical progression from core functionalization to peripheral modification. The process begins with the activation of the triptycene core, followed by the iterative growth of the acene wings. Careful control of reaction stoichiometry and temperature is essential to prevent over-reaction or decomposition of the sensitive acene intermediates. The final step often involves the installation of solubilizing groups, such as silyl-ethynyl moieties, which enhance processability without disrupting the electronic conjugation. Detailed standardized synthesis steps for specific derivatives are outlined in the guide below.
- Perform Friedel-Crafts acylation of triptycene with acene-type phthalic anhydride using AlCl3 in tetrachloroethylene at 115°C to obtain intermediate 1.
- Heat intermediate 1 in concentrated sulfuric acid at 120°C to induce intramolecular cyclization, yielding the triptycene acenone derivative 2.
- Conduct Michael addition with lithium acetylides followed by dehydration using SnCl2/HCl to install ethynyl substituents for final product 3.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthetic routes described in this patent offer substantial advantages in terms of cost efficiency and supply chain resilience. The reliance on commodity chemicals like aluminum trichloride, sulfuric acid, and bromine significantly lowers the raw material cost profile compared to routes dependent on rare earth metals or specialized organometallic reagents. This shift in reagent selection simplifies the procurement process and reduces exposure to volatile market prices associated with precious metal catalysts. Furthermore, the robustness of the reaction conditions suggests that the process can be easily transferred from laboratory scale to pilot and production scales with minimal re-optimization, ensuring a stable supply of high-purity OLED material precursors.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium or platinum removes the need for rigorous metal scavenging steps, which are often a bottleneck in pharmaceutical and electronic chemical production. By utilizing Lewis and Brønsted acids, the process simplifies the workup procedure, reducing solvent consumption and waste disposal costs. The high yields reported in the examples, such as the 84% yield in the initial acylation step, indicate a material-efficient process that maximizes output per batch. This efficiency directly contributes to lower unit costs, making these high-performance materials economically viable for mass-market applications.
- Enhanced Supply Chain Reliability: The starting materials, including triptycene and various phthalic anhydrides, are commercially available from multiple global suppliers, mitigating the risk of single-source dependency. The synthetic pathway does not require cryogenic conditions or ultra-high vacuum equipment, allowing production to be established in standard chemical manufacturing facilities. This accessibility ensures that lead times can be kept short and consistent, providing procurement managers with the predictability needed for long-term planning. The ability to synthesize a wide range of derivatives from a common intermediate further enhances supply flexibility, allowing manufacturers to respond quickly to changing customer specifications.
- Scalability and Environmental Compliance: The use of recyclable solvents like tetrachloroethylene and the potential for acid recovery in the cyclization step align with modern green chemistry principles. The process generates fewer hazardous byproducts compared to cross-coupling reactions, simplifying environmental compliance and reducing the burden on wastewater treatment systems. The solid-state properties of the final products, which resist aggregation, also imply better stability during storage and transport, reducing the risk of degradation before use. These factors collectively support a sustainable manufacturing model that meets the increasingly stringent regulatory requirements of the global electronic materials market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these triptycene-based compounds. The answers are derived directly from the experimental data and theoretical framework presented in the patent documentation. Understanding these details is crucial for evaluating the feasibility of integrating these materials into your existing product lines.
Q: What is the primary advantage of using a triptycene core for linear acenes?
A: The triptycene core provides a rigid C3-symmetric structure with large intramolecular free volume, which prevents detrimental pi-pi stacking aggregation and enhances solubility while maintaining efficient through-space electronic interactions for singlet fission.
Q: Can these compounds be synthesized on a commercial scale?
A: Yes, the synthesis relies on robust industrial reactions like Friedel-Crafts acylation and uses readily available reagents such as aluminum trichloride and sulfuric acid, avoiding expensive transition metal catalysts that complicate scale-up.
Q: What applications do these materials support beyond solar cells?
A: Due to their tunable bandgaps and high charge mobility, these linear acene trimers are also highly suitable for organic field-effect transistors (OFETs) and organic light-emitting diodes (OLEDs) as emissive or transport layers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triptycene-Based Linear Acene Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis for advanced electronic materials, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team of expert chemists is well-versed in the intricacies of Friedel-Crafts chemistry and acene functionalization, ensuring that we can deliver these complex triptycene derivatives with stringent purity specifications. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the structural integrity and electronic properties of every batch, guaranteeing performance consistency for your organic photoelectric devices.
We invite you to contact our technical procurement team to discuss your specific requirements. We are prepared to provide a Customized Cost-Saving Analysis tailored to your volume needs and timeline. Reach out today to request specific COA data and route feasibility assessments for your next-generation solar cell or OLED projects, and let us help you accelerate your path to market with reliable, high-quality materials.
