Advanced Copper-Catalyzed Synthesis of 2-Azabicyclo[3.2.0] Scaffolds for Commercial Manufacturing
Introduction to Next-Generation 2-Azabicyclo[3.2.0] Synthesis
The 2-azabicyclo[3.2.0] structural motif represents a privileged scaffold in modern medicinal chemistry, frequently appearing in complex natural products and biologically active molecules such as phantasmidine and various IDO inhibitors. As depicted in the structural overview below, these fused ring systems offer unique three-dimensional architectures that are crucial for optimizing drug-target interactions. However, accessing these cores has historically been a significant bottleneck in process chemistry due to the lack of efficient, general synthetic methodologies. The recent disclosure in patent CN111518014A introduces a transformative approach that addresses these long-standing challenges by utilizing silyl enol ether-bonded alkynamines as versatile precursors.
![Structural diversity of bioactive 2-azabicyclo[3.2.0] compounds including phantasmidine and IDO inhibitors](/insights/img/2-azabicyclo-compound-synthesis-pharma-supplier-20260305211832-01.webp)
This novel methodology leverages a transition metal-catalyzed cyclization strategy that operates under remarkably mild and economical conditions. Unlike traditional multi-step sequences that often suffer from poor atom economy and low overall yields, this one-pot transformation delivers the target bicyclic frameworks with exceptional efficiency. For R&D directors and process chemists, this represents a critical advancement, as it simplifies the synthetic route to high-value pharmaceutical intermediates. The ability to generate these complex cores directly from readily available starting materials significantly accelerates the timeline for lead optimization and candidate selection in drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the construction of bridged N-heterocyclic compounds relied heavily on noble metal catalysis, which imposed severe economic and logistical constraints on manufacturing. As illustrated in the prior art reaction scheme, earlier methods reported by researchers such as Miesch utilized silver catalysts to effect similar cyclizations. While scientifically valid, the reliance on silver (Ag) or gold (Au) complexes introduces substantial cost burdens, particularly when scaling to kilogram or ton quantities required for clinical supply. Furthermore, these noble metal-catalyzed reactions often necessitate rigorous exclusion of moisture and oxygen, demanding specialized equipment and increasing the complexity of the operational protocol.
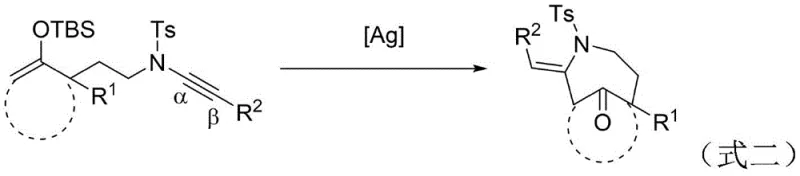
Beyond the cost of the catalyst itself, the removal of residual heavy metals from the final active pharmaceutical ingredient (API) is a stringent regulatory requirement. Processes utilizing silver or gold often require additional purification steps, such as scavenger treatments or extensive recrystallization, to meet ppm-level specifications. These downstream processing requirements not only extend the production cycle time but also contribute to increased waste generation and reduced overall process mass intensity (PMI). Consequently, there has been an urgent industry demand for a more sustainable and cost-effective alternative that maintains high selectivity without the baggage of precious metal dependency.
The Novel Approach
The methodology described in CN111518014A fundamentally shifts the paradigm by employing earth-abundant copper catalysts to drive the cyclization of silyl enol ether-bonded alkynamines. This strategic substitution replaces expensive noble metals with inexpensive copper salts, such as Cu(MeCN)4BF4, which are commercially available at a fraction of the cost. The reaction proceeds smoothly at room temperature, eliminating the need for energy-intensive heating or cooling cycles that are often required to activate less reactive catalytic systems. This thermal neutrality not only reduces utility costs but also enhances the safety profile of the process by minimizing the risk of thermal runaway events.
Moreover, the new approach demonstrates superior functional group tolerance, allowing for the direct incorporation of diverse substituents without the need for extensive protecting group manipulations. The reaction exhibits high regioselectivity and stereoselectivity, consistently delivering the desired 2-azabicyclo[3.2.0] core with minimal formation of byproducts. This high level of control simplifies the purification workflow, often allowing for direct isolation of the product via standard silica gel chromatography. For procurement and supply chain teams, this translates to a more robust and predictable manufacturing process that is less susceptible to variability in raw material quality or minor fluctuations in reaction parameters.
Mechanistic Insights into Copper-Catalyzed Cyclization
The core of this technological breakthrough lies in the unique reactivity of the silyl enol ether-bonded alkynamine substrate under copper catalysis. The proposed mechanism involves the activation of the alkyne moiety by the copper center, which facilitates an intramolecular nucleophilic attack by the enol ether oxygen or the adjacent pi-system. This initiates a cascade cyclization event that constructs the strained four-membered ring fused to the five-membered nitrogen heterocycle. The use of Cu(MeCN)4BF4 is particularly effective, as the acetonitrile ligands provide a labile coordination environment that allows for rapid substrate binding and turnover. This mechanistic pathway differs significantly from the silver-catalyzed variants, offering a distinct electronic environment that favors the formation of the 2-azabicyclo[3.2.0] skeleton over other potential isomers.
![General reaction scheme for copper-catalyzed synthesis of 2-azabicyclo[3.2.0] compounds from Formula I to Formula II](/insights/img/2-azabicyclo-compound-synthesis-pharma-supplier-20260305211832-03.webp)
Impurity control is a critical aspect of this mechanism, as the mild conditions prevent the degradation of sensitive functional groups that might otherwise decompose under harsher thermal or acidic conditions. The reaction tolerates a broad spectrum of electronic environments on the aromatic ring, as evidenced by the successful synthesis of derivatives bearing electron-withdrawing halogens and electron-donating methoxy groups. The substrate scope study reveals that variations in the R group, ranging from simple phenyl rings to complex heterocycles and aliphatic chains, do not significantly impede the cyclization efficiency. This robustness suggests that the catalytic cycle is resilient to steric hindrance and electronic perturbations, making it a highly reliable tool for synthesizing diverse libraries of analogues for structure-activity relationship (SAR) studies.
![Substrate scope showing diverse 2-azabicyclo[3.2.0] derivatives II-2 to II-16 with various substituents](/insights/img/2-azabicyclo-compound-synthesis-pharma-supplier-20260305211832-07.webp)
How to Synthesize 2-Azabicyclo[3.2.0] Compounds Efficiently
The practical implementation of this synthesis is straightforward and aligns well with standard laboratory and pilot plant operations. The protocol begins with the dissolution of the silyl enol ether-bonded alkynamine precursor in a chlorinated solvent such as 1,2-dichloroethane (DCE), followed by the addition of the copper catalyst under an inert atmosphere. The mixture is then stirred at ambient temperature, with reaction progress monitored by thin-layer chromatography (TLC). Upon completion, typically achieved within 30 minutes to a few hours, the solvent is removed under reduced pressure, and the crude product is purified. The detailed standardized synthesis steps for replicating this high-yielding transformation are provided in the guide below.
- Charge a reactor with the silyl enol ether-bonded alkynamine substrate (Formula I), a copper catalyst such as Cu(MeCN)4BF4, and an organic solvent like 1,2-dichloroethane (DCE).
- Maintain the reaction mixture under an inert atmosphere (argon or nitrogen) at room temperature and stir for approximately 30 minutes to 14 hours depending on substrate reactivity.
- Upon completion monitored by TLC, concentrate the mixture and purify the crude residue via silica gel column chromatography using a petroleum ether and ethyl acetate gradient to isolate the target 2-azabicyclo[3.2.0] product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this copper-catalyzed route offers compelling advantages that directly impact the bottom line and supply chain resilience. The most immediate benefit is the drastic reduction in raw material costs associated with the catalyst. By shifting from precious metals like silver or gold to base metal copper, manufacturers can achieve substantial cost savings on a per-kilogram basis. This is particularly significant for high-volume production where catalyst loading, even at low mole percentages, can represent a major expense. Furthermore, the elimination of expensive metal scavengers required for noble metal removal further reduces the bill of materials and simplifies the supply chain for auxiliary reagents.
- Cost Reduction in Manufacturing: The economic benefits extend beyond just the catalyst price. The ability to run the reaction at room temperature significantly lowers energy consumption compared to processes requiring reflux or cryogenic conditions. This reduction in thermal load decreases the demand on HVAC and utility systems within the manufacturing facility. Additionally, the short reaction times observed in the optimization studies, often completing in as little as 30 minutes, increase the throughput capacity of existing reactor vessels. This intensification of the process allows for more batches to be produced in the same timeframe, effectively lowering the fixed cost allocation per unit of product and enhancing overall manufacturing efficiency.
- Enhanced Supply Chain Reliability: Relying on copper catalysts mitigates the supply risk associated with precious metals, which are subject to geopolitical volatility and market price fluctuations. Copper is an abundant, globally sourced commodity with a stable supply chain, ensuring consistent availability for long-term production campaigns. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality or environmental factors, leading to higher batch-to-batch consistency. This reliability is crucial for maintaining uninterrupted supply to downstream customers and meeting strict delivery schedules without the risk of campaign failures due to catalyst deactivation or sensitivity.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving standard concentration and chromatography, facilitates easy scale-up from gram to kilogram scales without the need for specialized equipment. The use of common organic solvents like DCE, which are widely handled in the fine chemical industry, ensures compatibility with existing infrastructure. From an environmental standpoint, the high atom economy and reduced waste generation associated with the high-yielding nature of the reaction contribute to a greener manufacturing profile. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, making the process attractive for companies aiming to reduce their carbon footprint and improve their E-factor metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the operational parameters and expected outcomes. Understanding these details is essential for project managers and technical leads evaluating the feasibility of integrating this route into their current manufacturing portfolios.
Q: What are the primary advantages of this copper-catalyzed method over previous silver-catalyzed routes?
A: The primary advantage is the substitution of expensive noble metals like silver or gold with inexpensive and abundant copper catalysts. Additionally, this method operates efficiently at room temperature with significantly shorter reaction times, reducing energy consumption and operational costs while maintaining high yields up to 98%.
Q: What is the substrate scope for this synthesis method?
A: The method demonstrates excellent tolerance for a wide variety of functional groups. It successfully accommodates substrates with electron-withdrawing groups like halogens and cyano groups, as well as electron-donating groups such as alkyl and alkoxy substituents on the aromatic ring, allowing for the diverse synthesis of pharmaceutical intermediates.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable due to its mild reaction conditions (room temperature), use of common solvents like DCE, and the avoidance of sensitive or hazardous reagents. The short reaction time and simple workup procedure involving standard column chromatography facilitate efficient scale-up for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Azabicyclo[3.2.0] Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient synthetic routes like the one described in CN111518014A for accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are seamlessly translated into robust industrial processes. We are committed to delivering high-purity 2-azabicyclo[3.2.0] intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee quality and consistency.
We invite potential partners to engage with our technical procurement team to discuss how this advanced copper-catalyzed technology can be tailored to your specific project needs. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this methodology for your specific target molecule. Please contact us to request specific COA data for our available catalog compounds or to initiate a discussion on route feasibility assessments for your custom synthesis requirements.
