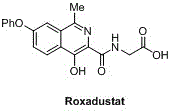
Commercializing Roxadustat Intermediate IV: A Safer Scalable Synthetic Route for Global API Production
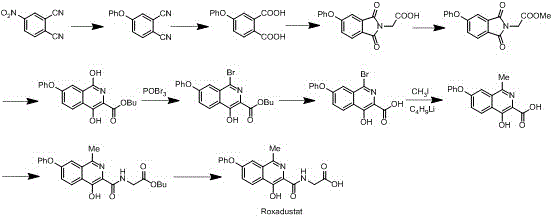
The pharmaceutical industry is constantly seeking robust synthetic pathways that balance molecular complexity with industrial safety, particularly for high-value Hypoxia Inducible Factor (HIF) prolyl hydroxylase inhibitors like Roxadustat. Patent CN112375057A introduces a transformative preparation method for Intermediate IV of Roxadustat, addressing critical bottlenecks found in legacy synthetic routes. This innovation focuses on a streamlined three-step sequence that begins with the precise addition of a methyl Grignard reagent to 4-bromophthalic anhydride, followed by a controlled reduction and cyclization, and concludes with a copper-catalyzed aryl alkylation. By shifting away from hazardous reagents such as n-butyllithium and phosphorus oxychloride, this methodology offers a safer, more economically viable pathway for the commercial scale-up of complex pharmaceutical intermediates. The strategic introduction of the methyl group at an early stage simplifies the overall molecular architecture, reducing the cumulative yield loss often associated with late-stage functionalization. For global procurement teams and R&D directors, this patent represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials without the regulatory baggage of toxic heavy metals or explosive precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Roxadustat and its key intermediates has been plagued by operational hazards and environmental inefficiencies that hinder large-scale adoption. Traditional routes, such as those disclosed in earlier patents like WO 2004108681A1, often rely on the use of n-butyllithium for methyl introduction, a reagent that is pyrophoric and requires stringent inert atmosphere conditions that drive up operational costs. Furthermore, many conventional processes utilize phosphorus oxybromide or phosphorus oxychloride for halogenation steps, generating substantial quantities of phosphorus-containing waste liquids that require expensive treatment protocols to meet environmental compliance standards. Another significant drawback in prior art involves the use of palladium-catalyzed Suzuki couplings with trimethyl borane, a highly flammable gas that poses severe safety risks during industrial amplification. These legacy methods also frequently necessitate special hydrogenation reactions or ultra-low temperature conditions below -70°C, demanding specialized equipment that limits production flexibility and increases capital expenditure. The cumulative effect of these factors is a supply chain vulnerable to disruptions, higher cost of goods sold (COGS), and increased regulatory scrutiny regarding residual heavy metals and solvent impurities.
The Novel Approach
In stark contrast, the novel approach detailed in CN112375057A重构 s the synthetic logic to prioritize safety and scalability without compromising chemical efficiency. This method cleverly introduces the methyl group at the very beginning of the sequence using a methyl Grignard reagent under mild thermal conditions, effectively bypassing the need for dangerous organolithium species. The subsequent reduction step utilizes sodium borohydride, a benign and easily handled reducing agent, which replaces more aggressive hydrides that often require quenching procedures generating hydrogen gas. The final cyclization and arylation steps are conducted in common organic solvents like dioxane or toluene using earth-abundant copper catalysts instead of precious palladium metals. This shift not only reduces the raw material cost significantly but also simplifies the purification process, as copper residues are generally easier to manage and remove to meet ICH Q3D guidelines compared to palladium. By eliminating high-energy oxidation steps and avoiding the use of toxic methyl iodide, this new route creates a manufacturing environment that is inherently safer for operators and more sustainable for the environment, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Cu-Catalyzed Arylation and Grignard Addition
The core of this synthetic breakthrough lies in the precise control of the Grignard addition and the subsequent copper-catalyzed coupling mechanism. In the first step, the reaction of 4-bromophthalic anhydride with methyl magnesium bromide is conducted at a tightly regulated temperature range of -10°C to 5°C. This thermal window is critical for controlling the regioselectivity of the nucleophilic attack on the anhydride ring, ensuring that the acetyl group is formed at the desired position while minimizing the formation of regio-isomeric impurities. The presence of a cuprous salt catalyst, such as cuprous iodide or cuprous bromide, further enhances the selectivity and yield of this transformation by facilitating the transmetallation process. Moving to the final step, the aryl alkylation of the bromo-lactone intermediate with phenol proceeds via a copper-mediated mechanism that activates the carbon-bromine bond for nucleophilic substitution. This catalytic cycle allows for the efficient formation of the carbon-oxygen bond necessary for the phenoxy substituent, which is a defining feature of the Roxadustat scaffold. The choice of base, typically potassium carbonate, and the solvent system plays a pivotal role in stabilizing the transition state and driving the reaction to completion without degrading the sensitive lactone ring.
Impurity control is another paramount aspect of this mechanistic design, particularly concerning the isomeric purity of the intermediate II. The patent data indicates that strict temperature control during the Grignard addition favors a specific isomer ratio, approximately 9:1, which is far superior to the mixtures obtained at higher temperatures. This high initial purity reduces the burden on downstream crystallization and chromatography steps, directly impacting the overall yield and cost efficiency. Furthermore, the use of sodium borohydride for the reduction of the keto-acid to the hydroxy-acid, followed by acid-catalyzed lactonization, avoids the over-reduction side reactions often seen with stronger reducing agents. The acidic cyclization condition is optimized to promote intramolecular esterification while preventing the hydrolysis of the newly formed lactone ring. By understanding these mechanistic nuances, R&D directors can appreciate how this route minimizes the generation of difficult-to-remove byproducts, thereby ensuring a consistent and high-quality supply of the intermediate suitable for subsequent API synthesis steps.
How to Synthesize 3-methyl-5-phenoxyisobenzofuran-1(3H)-one Efficiently
The practical implementation of this synthesis requires adherence to specific operational parameters to maximize yield and safety. The process begins with the preparation of the Grignard reagent and its slow addition to the anhydride solution under nitrogen protection, ensuring that moisture and oxygen are excluded to prevent reagent decomposition. Following the isolation of Intermediate II, the reduction step involves the batch-wise addition of sodium borohydride to an alkaline solution, followed by careful pH adjustment to induce cyclization. The final coupling reaction requires heating the mixture of the bromo-lactone and phenol with the copper catalyst in a high-boiling solvent to facilitate the arylation. Detailed standardized operating procedures (SOPs) are essential for managing the exothermic nature of the Grignard reaction and the gas evolution during the acid quench. For a comprehensive guide on the exact molar ratios, workup procedures, and purification techniques, please refer to the structured synthesis guide below.
- Perform Grignard addition of methyl magnesium bromide to 4-bromophthalic anhydride at -10 to 5°C with cuprous iodide catalyst.
- Reduce the resulting keto-acid intermediate using sodium borohydride followed by acidic cyclization to form the lactone ring.
- Execute copper-catalyzed aryl alkylation with phenol in dioxane solvent to yield the final phenoxy-substituted intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial advantages that directly address the pain points of procurement managers and supply chain heads in the pharmaceutical sector. The elimination of exotic and hazardous reagents translates into a more resilient supply chain, as the raw materials required—such as 4-bromophthalic anhydride, phenol, and sodium borohydride—are commodity chemicals available from multiple global suppliers. This diversification of the supply base reduces the risk of single-source bottlenecks and price volatility, ensuring a steady flow of materials for continuous manufacturing operations. Moreover, the avoidance of special unit operations like high-pressure hydrogenation or cryogenic cooling below -70°C means that the process can be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This compatibility with existing infrastructure drastically lowers the barrier to entry for contract manufacturing organizations (CMOs) and reduces the capital investment required for technology transfer.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the substitution of expensive and dangerous reagents with cost-effective alternatives. By removing the need for palladium catalysts and n-butyllithium, the direct material cost is significantly reduced, while the simplified workup procedures lower labor and utility expenses. The higher selectivity of the reaction minimizes waste generation, leading to improved atom economy and reduced costs associated with waste disposal and environmental compliance. Additionally, the use of recoverable solvents like dioxane and toluene further enhances the overall cost efficiency of the process. These factors combine to create a manufacturing profile that supports aggressive cost reduction in API manufacturing without sacrificing quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of stable, non-hazardous reagents that do not require special shipping permits or storage conditions. Unlike pyrophoric reagents that demand dedicated logistics and handling protocols, the chemicals used in this process can be transported and stored using standard industrial practices. This ease of handling reduces lead times for high-purity pharmaceutical intermediates by eliminating administrative delays associated with hazardous material transport. Furthermore, the robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production failures or out-of-specification results that could disrupt downstream API synthesis schedules.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, avoiding reaction steps that are difficult to control on a large scale, such as fast exotherms or gas-evolving reactions that pose pressure risks. The absence of phosphorus-containing reagents eliminates the generation of phosphorus waste streams, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. The use of copper catalysts, which are less toxic than heavy metals like palladium or platinum, eases the burden on purification systems to meet residual metal specifications. This environmental friendliness not only reduces operational costs but also enhances the corporate social responsibility profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These answers are derived directly from the experimental data and advantageous effects described in the patent literature, providing clarity on safety, scalability, and quality control. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this technology for their supply chains. The responses highlight the specific improvements over prior art and the practical implications for commercial production.
Q: How does this new route improve safety compared to conventional n-butyllithium methods?
A: This route eliminates the need for pyrophoric n-butyllithium and toxic methyl iodide, replacing them with stable Grignard reagents and mild sodium borohydride reduction, significantly lowering industrial safety risks.
Q: What are the impurity control advantages of the low-temperature Grignard step?
A: Maintaining the reaction temperature between -10°C and 5°C strictly controls regioselectivity, minimizing the formation of isomer impurities and ensuring a cleaner crude profile for downstream processing.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process avoids special hydrogenation units and ultra-low temperature requirements, utilizing common solvents like dioxane and readily available catalysts, making it highly scalable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-methyl-5-phenoxyisobenzofuran-1(3H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from pilot plant to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs and analytical instruments capable of verifying stringent purity specifications, guaranteeing that every batch of Roxadustat Intermediate IV meets the highest international standards. We understand the critical nature of API intermediates in the drug development timeline and are committed to providing a reliable pharmaceutical intermediate supplier partnership that prioritizes quality, safety, and timeliness.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain solution that combines cutting-edge chemistry with commercial reliability, positioning your organization for success in the competitive global pharmaceutical market.
