Advanced Copper-Catalyzed Synthesis of 4-Aryl-1H-1,2,3-Triazoles for Commercial Scale-Up
Advanced Copper-Catalyzed Synthesis of 4-Aryl-1H-1,2,3-Triazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing heterocyclic scaffolds that serve as the backbone of modern therapeutics. Patent CN102127024A introduces a transformative approach to synthesizing 4-aryl-1H-1,2,3-triazoles, a class of compounds renowned for their profound biological activity and structural versatility. This technology leverages the reaction between 1,1-dibromo-1-alkenes and azide compounds under copper catalysis, offering a distinct alternative to traditional alkyne-based routes. The significance of this innovation cannot be overstated, as 1,2,3-triazole derivatives are pivotal in the development of beta-lactamase inhibitors like Tazobactam, as well as various antitumor and antifungal agents. By shifting the synthetic paradigm from terminal alkynes to readily accessible dibromo-olefins, this method addresses critical supply chain vulnerabilities associated with precursor availability and stability.
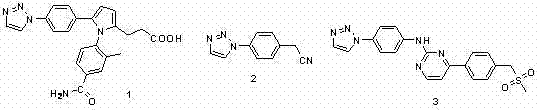
For R&D directors and procurement specialists, the implications of this patent extend far beyond mere academic interest. The ability to access high-purity 4-aryl-1,2,3-triazoles through a streamlined, copper-catalyzed process represents a significant opportunity for cost reduction in API manufacturing. The patent details a protocol that operates efficiently in polar aprotic solvents like dimethyl sulfoxide (DMSO), utilizing inexpensive inorganic bases and abundant copper salts. This eliminates the reliance on precious metal catalysts often required in cross-coupling reactions, thereby drastically lowering the raw material costs and simplifying the downstream purification processes required to meet stringent pharmaceutical purity specifications. Furthermore, the broad substrate tolerance described in the invention suggests that a wide array of functionalized aryl groups can be incorporated, facilitating rapid library synthesis for drug screening programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
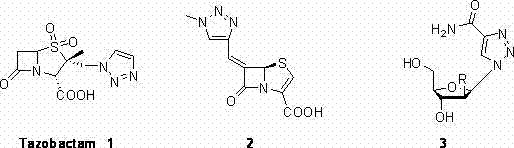
Historically, the construction of the 1,2,3-triazole ring has been dominated by the Huisgen 1,3-dipolar cycloaddition between alkynes and azides. While the copper-catalyzed variant (CuAAC), often termed "Click Chemistry," revolutionized the field with its high selectivity and yield, it is not without significant drawbacks in an industrial setting. The primary limitation lies in the requirement for terminal alkynes, which can be unstable, prone to polymerization, and sometimes difficult to source in bulk quantities with consistent quality. Additionally, achieving specific regioselectivity (1,4-disubstituted vs. 1,5-disubstituted) often demands precise control over reaction conditions and catalyst systems, which can complicate scale-up efforts. In many cases, the use of ruthenium catalysts to access 1,5-isomers introduces further cost burdens due to the expense of the metal and the rigorous removal protocols necessary to ensure residual metal levels comply with ICH guidelines for pharmaceutical products.
The Novel Approach
The methodology disclosed in CN102127024A circumvents these challenges by utilizing 1,1-dibromo-1-alkenes as the starting olefinic component. This strategic shift offers several compelling advantages for process chemists. Firstly, 1,1-dibromo-1-alkenes are generally more stable and easier to handle than their terminal alkyne counterparts, reducing safety risks during storage and transportation. Secondly, the reaction proceeds smoothly under relatively mild thermal conditions in the presence of a base and a copper catalyst, yielding the desired 4-aryl-1H-1,2,3-triazole with high efficiency. The use of sodium azide or organic azides allows for flexible substitution patterns at the N1 position, while the aryl group on the alkene dictates the C4 substitution. This modularity is crucial for medicinal chemists aiming to explore structure-activity relationships (SAR) without being constrained by the availability of specific alkyne building blocks.
Mechanistic Insights into Copper-Catalyzed Cyclization
The core of this synthetic innovation lies in the copper-catalyzed activation of the 1,1-dibromo-1-alkene towards nucleophilic attack by the azide species. The proposed mechanism involves the coordination of the copper catalyst to the electron-deficient double bond of the dibromo-olefin, thereby enhancing its electrophilicity. Subsequent nucleophilic addition of the azide anion initiates the cyclization cascade. The presence of the two bromine atoms on the same carbon is critical; one bromine atom acts as a leaving group during the aromatization process, driving the formation of the stable triazole ring. This elimination step is facilitated by the base present in the reaction mixture, which neutralizes the generated acid and pushes the equilibrium towards product formation. The choice of ligand, such as sodium ascorbate or diamines, plays a pivotal role in stabilizing the active copper(I) species and preventing oxidation to inactive copper(II) states, ensuring sustained catalytic turnover throughout the reaction duration.
From an impurity control perspective, this mechanism offers a cleaner profile compared to traditional methods. The reaction pathway is highly specific, minimizing the formation of regio-isomeric byproducts that often plague alkyne-azide cycloadditions. The primary side reactions typically involve incomplete conversion or hydrolysis of the starting material, both of which are easily managed through optimized reaction times and stoichiometry. The use of DMSO as a solvent not only solubilizes the inorganic azide salts effectively but also stabilizes the transition states involved in the cyclization. For quality assurance teams, this translates to a more predictable impurity spectrum, simplifying the validation of analytical methods and the establishment of control strategies for commercial batches. The robustness of the catalytic cycle ensures that even with slight variations in raw material quality, the process remains resilient, consistently delivering the target triazole scaffold.
How to Synthesize 4-Aryl-1H-1,2,3-Triazole Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and reaction parameters to maximize yield and purity. The patent provides detailed embodiments that serve as a blueprint for process optimization. Typically, the reaction is conducted by charging a reactor with the 1,1-dibromo-1-alkene, a copper salt catalyst (such as CuI or CuBr), a suitable ligand, sodium azide, and an inorganic base like potassium carbonate or cesium carbonate. The mixture is then heated, often in a sealed vessel to prevent solvent loss, and stirred for a period ranging from 2 to 20 hours depending on the specific substrate reactivity. Workup involves standard extraction techniques followed by chromatographic purification, although for large-scale operations, crystallization methods would be developed to replace chromatography for cost efficiency.
- Combine 1,1-dibromo-1-alkene, copper catalyst (e.g., CuI), ligand (e.g., sodium ascorbate), sodium azide, and base (e.g., K2CO3) in DMSO solvent.
- Stir the reaction mixture magnetically at temperatures ranging from room temperature to 180°C for a duration of 2 to 20 hours.
- Extract the completed reaction with ethyl acetate, wash the organic layer with saturated brine, dry over anhydrous sodium sulfate, and purify via column chromatography.
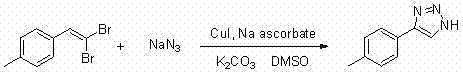
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that directly impact the bottom line and operational resilience. The shift away from precious metal catalysts and unstable alkynes towards base metals and stable dibromo-olefins fundamentally alters the cost structure of producing these valuable intermediates. This transition not only reduces the direct cost of goods sold (COGS) but also mitigates the risks associated with the volatility of noble metal markets. Furthermore, the simplified reaction setup and workup procedures reduce the burden on manufacturing facilities, allowing for faster batch turnover and more efficient utilization of production assets.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the substitution of expensive reagents with commodity chemicals. Copper salts and sodium azide are significantly cheaper than the palladium or ruthenium catalysts often used in alternative heterocycle syntheses. Additionally, the elimination of complex ligand systems and the use of common solvents like DMSO reduce the overall material spend. From a processing standpoint, the mild reaction conditions mean lower energy consumption for heating and cooling, contributing to substantial cost savings in utility expenses over the lifecycle of the product. The high yields reported in the patent examples suggest that raw material utilization is efficient, minimizing waste generation and the associated disposal costs.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount in the pharmaceutical industry, and this method enhances reliability by diversifying the source of key starting materials. 1,1-dibromo-1-alkenes can be synthesized from readily available aldehydes or ketones, meaning the supply chain is not dependent on a single niche vendor for specialized alkynes. The stability of these dibromo-precursors allows for longer shelf lives and bulk purchasing strategies, protecting the organization from short-term market fluctuations. Moreover, the robustness of the reaction conditions implies that the process can be transferred between different manufacturing sites with minimal re-validation effort, providing flexibility in case of regional disruptions or capacity constraints.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but the simplicity of this copper-catalyzed system facilitates a smoother transition from lab to plant. The absence of pyrophoric reagents or extreme pressure requirements simplifies the engineering controls needed for large reactors. Environmentally, the process generates fewer heavy metal wastes compared to noble metal catalysis, easing the burden on wastewater treatment systems. The use of DMSO, while requiring recovery, is a well-understood solvent in the industry with established recycling protocols. This alignment with green chemistry principles not only reduces the environmental footprint but also ensures compliance with increasingly stringent global regulations regarding chemical manufacturing and waste discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and theoretical framework presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential partners and licensees.
Q: What are the primary advantages of this method over traditional Click Chemistry?
A: Unlike traditional alkyne-azide cycloadditions which often require terminal alkynes and specific regio-control, this method utilizes readily available 1,1-dibromo-1-alkenes. It offers a broader substrate scope and avoids the handling of potentially unstable terminal alkynes, providing a safer and more versatile synthetic route for diverse aryl substitutions.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process is highly amenable to scale-up. It employs inexpensive and commercially available raw materials such as sodium azide and simple dibromo-olefins. The reaction conditions are relatively mild, utilizing common solvents like DMSO and standard copper catalysts, which simplifies the engineering requirements for large-scale manufacturing compared to noble metal-catalyzed processes.
Q: What types of biological activities are associated with the resulting triazole compounds?
A: The synthesized 4-aryl-1,2,3-triazoles possess significant pharmacological potential. As highlighted in the patent background, this structural motif is found in critical drugs like Tazobactam (a beta-lactamase inhibitor) and exhibits activities ranging from antibacterial and antifungal to antitumor and anti-inflammatory properties, making them valuable intermediates for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aryl-1H-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development pipelines. Our team of expert process chemists has extensively evaluated the technology described in CN102127024A and possesses the capability to optimize this route for your specific needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 4-aryl-1H-1,2,3-triazole we deliver meets the highest standards of quality and consistency required by global regulatory bodies.
We invite you to collaborate with us to leverage this innovative synthetic strategy for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to reach out today to discuss your project needs, obtain specific COA data for our reference standards, and receive comprehensive route feasibility assessments that will empower your R&D and supply chain teams to make informed decisions. Let us be your trusted partner in bringing life-saving medicines to market faster and more efficiently.
