Advanced Magnetic Catalysis for Scalable Production of Imidazo[1,2-a]pyridine Derivatives
Introduction to Next-Generation Pharmaceutical Intermediate Synthesis
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient, and scalable synthetic methodologies. According to patent CN115286629B, a groundbreaking approach has been developed for the preparation of 3-anilinoimidazo[1,2-a]pyridine derivatives, a critical scaffold found in numerous bioactive compounds including hypnotics and anti-ulcer agents. This technology leverages a novel Fe3O4@SiO2 grafted polysulfonate material as a heterogeneous magnetic catalyst, marking a departure from traditional homogeneous acid catalysis. By integrating magnetic separation capabilities with high catalytic activity, this method addresses long-standing challenges in waste generation and catalyst recovery. For R&D directors and procurement specialists, understanding this shift is crucial as it promises not only higher purity profiles but also a streamlined supply chain for these high-value heterocyclic building blocks.
![General reaction scheme for synthesizing 3-anilinoimidazo[1,2-a]pyridine derivatives using magnetic catalyst](/insights/img/3-anilinoimidazo-pyridine-catalytic-synthesis-pharma-supplier-20260302204156-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of imidazo[1,2-a]pyridine derivatives via the Groebke-Blackburn-Bienayme (GBB) multicomponent reaction has relied heavily on homogeneous organic acid catalysts such as p-toluenesulfonic acid. While these methods offer mild reaction conditions, they suffer from severe drawbacks that hinder industrial adoption. The primary issue is the inability to recycle the catalyst, leading to the generation of substantial quantities of acidic waste that requires costly neutralization and disposal procedures. Furthermore, the separation of the product from the reaction mixture often necessitates complex work-up procedures, including extraction and column chromatography, which drastically reduce overall throughput. The low selectivity of some traditional catalysts also results in significant by-product formation, complicating the purification process and lowering the economic viability of large-scale production runs.
The Novel Approach
The innovative methodology described in the patent data introduces a robust solution by employing a Fe3O4@SiO2 grafted polysulfonate material. This heterogeneous catalyst combines the acidity required for the GBB reaction with the physical properties of a magnetic nanoparticle. The core-shell structure ensures that the active sulfonic acid sites are accessible while the magnetic core allows for instantaneous separation from the reaction mixture using an external magnetic field. This eliminates the need for filtration or centrifugation, significantly reducing processing time. Additionally, the use of an isopropanol aqueous solution as the solvent system serves a dual purpose: it acts as the reaction medium and subsequently as a recrystallization solvent. This clever integration allows the product to precipitate directly upon cooling, often achieving purities exceeding 99% without further purification, thereby revolutionizing the efficiency of the manufacturing workflow.
Mechanistic Insights into Fe3O4@SiO2 Catalyzed Cyclization
The catalytic mechanism relies on the synergistic interaction between the sulfonic acid groups grafted onto the silica surface and the reactants. The Brønsted acid sites protonate the carbonyl oxygen of the arylglyoxal hydrate, increasing its electrophilicity and facilitating nucleophilic attack by the 2-aminopyridine. This initial condensation forms an imine intermediate, which subsequently undergoes cyclization with the aromatic primary amine. The porous nature of the silica coating on the magnetite core provides a high surface area for these reactions to occur efficiently. Crucially, the magnetic property does not interfere with the chemical activity but rather enhances the process engineering aspect. The catalyst maintains its structural integrity throughout the reaction, preventing iron leaching which could otherwise contaminate the pharmaceutical product. This stability is paramount for meeting stringent regulatory requirements regarding heavy metal residues in active pharmaceutical ingredients (APIs) and their intermediates.
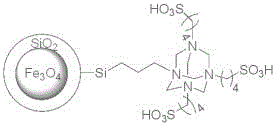
Impurity control is another critical aspect where this catalytic system excels. Traditional homogeneous acids often promote side reactions due to their high solubility and lack of steric constraints. In contrast, the heterogeneous nature of the Fe3O4@SiO2 catalyst imposes a degree of shape selectivity. The active sites are confined to the surface of the nanoparticles, which can limit the formation of bulky by-products that cannot easily access the catalytic pockets. Moreover, the high selectivity ensures that the desired 3-anilinoimidazo[1,2-a]pyridine scaffold is formed predominantly. The isopropanol-water solvent system further aids in impurity suppression; the specific polarity of this mixture favors the precipitation of the target product while keeping soluble impurities in the mother liquor. This intrinsic purification mechanism reduces the burden on downstream processing units and ensures a consistent quality profile across different batches, a key metric for supply chain reliability.
How to Synthesize 3-Anilinoimidazo[1,2-a]pyridine Derivatives Efficiently
The synthesis protocol outlined in the patent offers a straightforward, one-pot procedure that is highly amenable to standard laboratory and pilot plant equipment. The process begins with the suspension of the magnetic catalyst in an isopropanol aqueous solution, followed by the addition of the three key reactants: arylglyoxal hydrate, 2-aminopyridine, and an aromatic primary amine. The mixture is heated to reflux, typically maintaining the temperature such that solvent vapor does not exceed the second ball of the condenser, for a duration ranging from approximately 2 to 4 hours depending on the specific substrate substituents. Reaction progress is monitored via TLC until the starting materials are consumed. Upon completion, the heat and stirring are stopped, and the catalyst is immediately separated using a magnet while the solution is still hot. The filtrate is then allowed to cool naturally to room temperature, inducing crystallization of the product. The solid is collected by filtration, washed with ethanol to remove residual impurities, and dried under vacuum to yield the final high-purity derivative. For detailed standardized synthesis steps, please refer to the guide below.
- Mix arylglyoxal hydrate, 2-aminopyridine, and aromatic primary amine in isopropanol aqueous solution with the magnetic catalyst.
- Heat the suspension to reflux for approximately 2 to 4 hours until reaction completion, then separate the catalyst magnetically.
- Cool the filtrate to precipitate the product, filter, wash with ethanol, and dry to obtain high-purity derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this magnetic catalytic technology represents a strategic opportunity to optimize cost structures and enhance supply security. The elimination of homogeneous acid catalysts removes the logistical burden associated with handling, storing, and disposing of corrosive liquid acids. More importantly, the ability to recycle the catalyst multiple times without significant regeneration steps translates into substantial cost savings on raw materials. The simplified work-up procedure, which avoids energy-intensive distillation or chromatographic purification, reduces utility consumption and labor costs. These factors collectively contribute to a lower cost of goods sold (COGS), making the final intermediates more competitive in the global market. Furthermore, the robustness of the process ensures consistent supply continuity, mitigating the risks associated with batch failures or quality deviations that often plague complex synthetic routes.
- Cost Reduction in Manufacturing: The implementation of a recyclable heterogeneous catalyst fundamentally alters the cost equation of pharmaceutical intermediate manufacturing. By avoiding the purchase of fresh catalyst for every batch and eliminating the expensive waste treatment protocols required for homogeneous acids, manufacturers can achieve significant operational expenditure reductions. The one-pot nature of the reaction also minimizes solvent usage and reduces the time operators spend on reactor turnover. Additionally, the high purity of the crude product means that resources previously allocated to extensive purification processes can be redirected, further enhancing the overall economic efficiency of the production line without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of this synthetic route. The raw materials required—arylglyoxal hydrates, 2-aminopyridine, and aromatic amines—are commercially available and stable, reducing the risk of raw material shortages. The catalyst itself is durable and can be regenerated, ensuring that production is not halted due to catalyst depletion. The magnetic separation technique is easily scalable and less prone to mechanical failure compared to complex filtration systems used for fine precipitates. This reliability ensures that delivery schedules can be met consistently, fostering stronger relationships with downstream API manufacturers who depend on timely intermediate deliveries for their own production planning.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this magnetic catalytic system is inherently designed for expansion. The use of magnetic separation scales linearly from gram to tonne scales without loss of efficiency, unlike filtration which can become a bottleneck. From an environmental perspective, the process aligns with green chemistry principles by reducing waste generation and utilizing a safer solvent system. The ability to recover and reuse the isopropanol aqueous solvent further minimizes the environmental footprint. This compliance with increasingly stringent environmental regulations future-proofs the manufacturing process, avoiding potential fines or shutdowns and appealing to eco-conscious partners in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology. They are derived from the specific experimental data and beneficial effects reported in the patent literature, providing clarity on performance metrics and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific product portfolios. The answers reflect the proven capabilities of the Fe3O4@SiO2 system in maintaining high yields and purity across multiple cycles, assuring stakeholders of its industrial viability.
Q: What are the advantages of the Fe3O4@SiO2 catalyst over traditional organic acids?
A: Unlike traditional homogeneous organic acids like p-toluenesulfonic acid which generate waste acid and are difficult to recover, the Fe3O4@SiO2 grafted polysulfonate material is a heterogeneous magnetic catalyst. It allows for simple magnetic separation, can be recycled and regenerated multiple times without significant loss of activity, and eliminates the need for complex neutralization and waste treatment processes.
Q: Does this method require complex purification steps for the final product?
A: No, the method significantly simplifies purification. Due to the high selectivity of the catalytic system and the dual function of the isopropanol aqueous solution acting as both reaction and recrystallization solvent, the product often precipitates directly with high purity (often exceeding 99%) upon cooling, requiring only filtration and washing rather than column chromatography.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for industrial scalability. It utilizes a one-pot synthesis approach which reduces operational complexity, employs a robust magnetic catalyst that facilitates continuous processing, and uses environmentally benign solvents. The ability to recycle both the catalyst and the solvent system makes it economically and environmentally viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Anilinoimidazo[1,2-a]pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in modern pharmaceutical manufacturing. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the magnetic catalytic synthesis of 3-anilinoimidazo[1,2-a]pyridine derivatives are seamlessly translated into industrial reality. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the highest international standards. We understand that consistency and quality are non-negotiable in the pharma industry, and our commitment to process excellence ensures that your supply chain remains uninterrupted and compliant.
We invite you to collaborate with us to leverage these technological advancements for your specific projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this efficient route can improve your margins. Please contact us to request specific COA data for our catalog intermediates or to discuss route feasibility assessments for your custom synthesis needs. Let us help you optimize your supply chain with reliable, high-quality pharmaceutical intermediates produced through cutting-edge green chemistry.
