Revolutionizing 2-Iminohydantoin Production: A Technical Deep Dive into Patent CN111533694B
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access biologically active heterocyclic scaffolds, and the recent disclosure of patent CN111533694B marks a significant advancement in the synthesis of 2-iminohydantoin compounds. This specific intellectual property outlines a novel, one-step cycloaddition strategy that fundamentally alters the manufacturing landscape for this critical class of nitrogen-containing heterocycles, which are ubiquitous in natural products and drug candidates. By leveraging a direct reaction between alpha-haloacetamide derivatives and N-aryl-N-cyano-p-toluenesulfonamide compounds, the technology bypasses the tedious multi-step sequences that have historically plagued this chemical space. For R&D directors and procurement specialists alike, this patent represents not just a chemical curiosity, but a tangible opportunity to streamline supply chains and reduce the cost of goods for high-value pharmaceutical intermediates. The method's ability to operate under mild conditions while achieving exceptional yields suggests a robust platform for commercial scale-up, addressing key pain points regarding process safety and environmental compliance in modern chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2-iminohydantoin skeleton has been a challenging endeavor for synthetic chemists, often requiring convoluted routes that involve multiple protection and deprotection steps, leading to significant material loss and increased waste generation. Traditional methodologies frequently rely on harsh reaction conditions, including high temperatures and strong acidic or basic environments, which can compromise the integrity of sensitive functional groups on the substrate molecules. Furthermore, the starting materials for these legacy processes are often difficult to prepare or commercially unavailable, necessitating custom synthesis that drives up lead times and overall project costs. The cumulative effect of these inefficiencies is a low overall yield and a complex impurity profile that demands rigorous and expensive purification protocols, making the commercial production of these intermediates economically burdensome for large-scale applications.
The Novel Approach
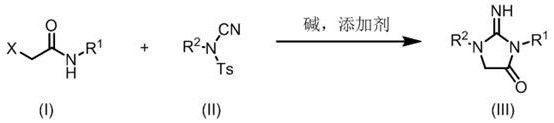
In stark contrast to these legacy issues, the methodology detailed in the patent data introduces a streamlined, atom-economical approach that utilizes readily available starting materials to construct the target heterocycle in a single operational step. This innovative route employs a base-promoted cycloaddition that proceeds smoothly at temperatures ranging from -10°C to 50°C, significantly reducing energy consumption and thermal stress on the reaction system. The use of N-aryl-N-cyano-p-toluenesulfonamide as an electrophilic cyanation reagent allows for the direct introduction of the necessary nitrogen and carbon atoms into the ring structure without the need for pre-functionalized complex precursors. This simplification of the synthetic tree not only accelerates the timeline from bench to kilogram scale but also inherently improves the purity of the crude product, thereby reducing the burden on downstream purification units and enhancing the overall sustainability of the manufacturing process.
Mechanistic Insights into CsF-Catalyzed Cycloaddition
The core of this technological breakthrough lies in the precise mechanistic interplay between the alpha-haloacetamide and the N-aryl-N-cyano-p-toluenesulfonamide under the influence of specific basic conditions. The reaction is initiated by the deprotonation of the alpha-haloacetamide by the base, generating a nucleophilic enolate or carbanion species that is stabilized by the adjacent carbonyl group. This nucleophile then attacks the electrophilic carbon of the cyano group on the sulfonamide reagent, triggering a cascade of intramolecular cyclization events that forge the new C-N bonds required to close the hydantoin ring. The presence of additives such as 18-crown-6 is critical in this mechanism, as they complex with the metal cations of the base, increasing the solubility and reactivity of the fluoride or carbonate anions in organic solvents like acetonitrile. This phase transfer catalysis effect ensures that the reaction kinetics are favorable even at lower temperatures, preventing side reactions such as hydrolysis or polymerization that often occur in more aggressive synthetic environments.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the formation of byproducts that are difficult to separate from the target molecule. The high chemoselectivity of the cycloaddition ensures that the reactive sites are engaged specifically, leaving other functional groups on the aryl rings intact, which is crucial for downstream derivatization in drug discovery campaigns. The mild conditions prevent the degradation of the sulfonamide leaving group into problematic sulfonic acid residues that could contaminate the final product. Furthermore, the stoichiometry of the reagents can be finely tuned, with the patent suggesting a molar ratio range that optimizes conversion while minimizing excess reagent waste. This level of control over the reaction pathway translates directly into a cleaner crude profile, reducing the need for extensive chromatographic purification and allowing for more efficient crystallization-based isolation methods in a commercial setting.
How to Synthesize 2-Iminohydantoin Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the specific reagent ratios and solvent choices outlined in the technical data to ensure reproducibility and safety. The process begins with the dissolution of the alpha-haloacetamide and the N-aryl-N-cyano-p-toluenesulfonamide in a polar aprotic solvent such as acetonitrile, which provides the necessary dielectric constant to support the ionic intermediates. The addition of the base and the crown ether additive must be managed to control the exotherm, although the reaction is generally mild compared to traditional methods. Detailed standardized synthesis steps see the guide below.
- Mix alpha-haloacetamide with N-aryl-N-cyano-p-toluenesulfonamide in a suitable organic solvent such as acetonitrile.
- Add a base like cesium fluoride and a phase transfer catalyst additive such as 18-crown-6 to the reaction mixture.
- Stir the reaction at mild temperatures between -10°C and 50°C, then quench with water and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology offers a compelling value proposition centered around cost reduction and supply reliability. By eliminating the need for multiple synthetic steps and expensive transition metal catalysts, the overall cost of manufacturing is significantly reduced, allowing for more competitive pricing in the global market for pharmaceutical intermediates. The use of commodity chemicals as starting materials ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialized or proprietary reagents, thereby enhancing the continuity of supply for long-term production contracts. Additionally, the mild reaction conditions reduce the engineering requirements for the production facility, lowering capital expenditure on specialized high-pressure or high-temperature reactors and simplifying the safety protocols required for operation.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction in synthetic steps directly translate to substantial cost savings by removing the need for expensive metal scavenging processes and reducing solvent consumption. The high yield reported in the patent data implies that less raw material is required to produce the same amount of product, improving the overall material efficiency and reducing the cost of goods sold. Furthermore, the simplified workup procedure reduces labor costs and utility consumption associated with prolonged heating or complex distillation processes, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as alpha-haloacetamides and sulfonamides ensures that the supply chain is robust and less susceptible to disruptions caused by the scarcity of exotic reagents. This accessibility allows for the qualification of multiple suppliers for raw materials, mitigating the risk of single-source dependency and providing greater flexibility in procurement strategies. The short reaction time and mild conditions also mean that production cycles are faster, enabling manufacturers to respond more quickly to fluctuations in market demand and reducing the inventory holding costs associated with long lead times.
- Scalability and Environmental Compliance: The process is inherently scalable due to its mild thermal profile and lack of hazardous reagents, making it suitable for transfer from gram-scale laboratory synthesis to multi-ton commercial production without significant re-engineering. The reduction in waste generation and the use of less hazardous solvents align with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations and reducing the costs associated with waste disposal. This environmental compatibility enhances the corporate social responsibility profile of the manufacturing process, which is an increasingly important factor for multinational pharmaceutical companies when selecting suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology, based on the specific data points and claims found within the patent documentation. These answers are designed to provide clarity on the operational parameters and the strategic advantages of adopting this method for the production of 2-iminohydantoin derivatives. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this new synthesis method over traditional routes?
A: The method described in patent CN111533694B offers a significantly shorter synthetic route, operating under mild reaction conditions with yields reaching up to 99%, eliminating the need for complex multi-step substrate preparation found in conventional methodologies.
Q: Which catalysts and additives are critical for this cycloaddition reaction?
A: The process relies on the synergistic effect of inorganic bases such as cesium fluoride or potassium carbonate combined with crown ether additives like 18-crown-6 to facilitate the nucleophilic attack and ring closure efficiently.
Q: Does the resulting 2-iminohydantoin scaffold demonstrate biological activity?
A: Yes, in vitro screening data indicates that compounds synthesized via this route exhibit potent antitumor activity against human breast cancer (MCF-7), prostate cancer (PC-3), and gastric cancer (MGC-803) cell lines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Iminohydantoin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in patent CN111533694B and have integrated this technology into our CDMO capabilities to serve the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-iminohydantoin intermediate meets the highest quality standards required for drug substance manufacturing, providing our partners with the confidence they need to advance their clinical programs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can optimize your supply chain and reduce your overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how our implementation of this patent can benefit your specific project requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about your sourcing strategy for this critical pharmaceutical intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
