Advanced Synthesis of D-A Type Triarylphosphine Compounds for Commercial Optoelectronic Applications
Advanced Synthesis of D-A Type Triarylphosphine Compounds for Commercial Optoelectronic Applications
The rapid evolution of the organic optoelectronics sector demands materials that transcend the limitations of traditional luminophores, particularly in the realm of stable free radicals. Patent CN114920774A introduces a groundbreaking class of D-A (Donor-Acceptor) type triarylphosphine compounds, specifically designed to overcome the stability and efficiency bottlenecks inherent in prior art triarylmethyl systems. This intellectual property outlines a robust synthetic methodology for generating compounds such as CP-DMA, DCP-DMA, and TCP-DMA, which exhibit exceptional paramagnetism, thermal stability, and unique photo-induced luminescent properties. By leveraging a donor-acceptor molecular architecture, these materials facilitate the generation of solid-state light-induced free radicals, a critical advancement for next-generation photoelectromagnetic devices. The strategic integration of strong electron-donating N,N-dimethyl groups with varying degrees of chlorinated acceptor units creates a versatile platform for tuning emission wavelengths and radical stability.
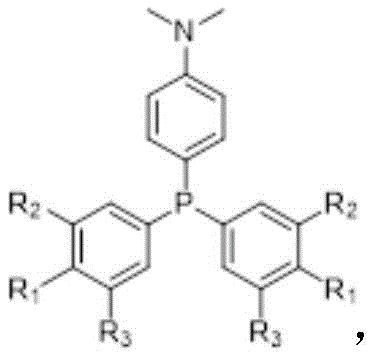
For procurement specialists and supply chain managers evaluating reliable electronic chemical suppliers, the structural versatility presented in this patent offers significant advantages. The core scaffold allows for precise modulation of electronic properties simply by adjusting the halogen substitution pattern on the phenyl rings, as illustrated in the general formula. This modularity means that a single synthetic platform can yield a family of distinct products tailored for specific emission requirements, ranging from pure blue to red light, without necessitating entirely new process development pipelines. Such flexibility is paramount for manufacturers seeking to diversify their product portfolios in the competitive display and lighting markets while maintaining streamlined production workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of stable organic luminescent radicals has been severely constrained by the dominance of the triarylmethyl radical family, including variants like PTM, TTM, and PyBTM. These conventional materials suffer from intrinsic limitations, primarily emitting in the orange to near-infrared regions, which leaves a significant gap in the visible spectrum, particularly for high-energy blue and green applications. Furthermore, the nonradiative transition rates in these traditional radicals are often two orders of magnitude higher than their radiative rates, leading to poor luminous efficiency caused by excessive intramolecular vibrational coupling. From a manufacturing perspective, the synthesis of these complex methyl-based radicals often involves harsh conditions and difficult purification steps to remove persistent impurities that quench luminescence. The inability to effectively reduce internal conversion through molecular design has stalled progress in creating efficient pure red, green, and especially blue light-emitting radical systems.
The Novel Approach
The innovative strategy disclosed in this patent pivots away from the carbon-centered triarylmethyl core to a phosphorus-centered triarylphosphine architecture, fundamentally altering the electronic landscape of the molecule. By constructing a Donor-Acceptor (D-A) system where a strong electron-donating dimethylamino phenyl group is coupled with chlorinated phenyl acceptors, the invention successfully mitigates the vibrational coupling issues plaguing earlier generations. This structural redesign not only shifts the emission capabilities into the desirable blue and red regions but also enhances the stability of the resulting photo-induced free radicals. The novel approach utilizes a stepwise synthesis that builds complexity gradually, allowing for better control over side reactions and impurity profiles compared to the one-pot or high-energy methods often required for triarylmethyl synthesis. This results in materials with superior electrochemical and photostability, making them viable candidates for long-lifetime commercial devices.
Mechanistic Insights into Palladium-Catalyzed Cross-Coupling
The heart of this synthetic breakthrough lies in the sophisticated application of transition metal catalysis to construct the sterically crowded triarylphosphine core. The process initiates with the formation of a diarylphosphine oxide intermediate via the nucleophilic attack of a Grignard reagent on diethyl phosphite, a reaction that must be meticulously controlled at low temperatures to prevent over-addition or decomposition. Following the isolation of the oxide, a chlorination step using phosphorus trichloride converts the P=O bond into a reactive P-Cl bond, activating the phosphorus center for the subsequent coupling event. The final and most critical stage involves a palladium-catalyzed cross-coupling reaction, where the chlorophosphine intermediate reacts with (4-(dimethylamino)phenyl)magnesium bromide. This step requires the precise orchestration of the Pd(0) catalytic cycle to facilitate the formation of the P-C bond between the phosphorus atom and the electron-rich aniline derivative.
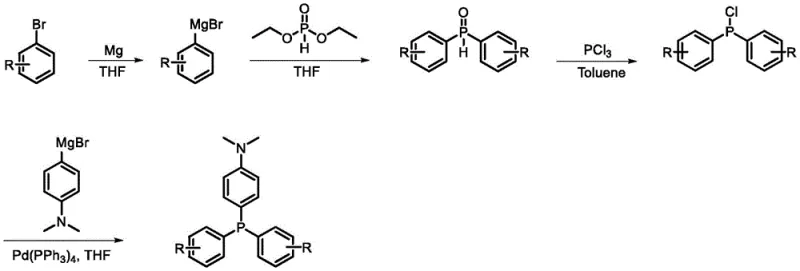
Understanding the mechanistic nuances of this coupling is essential for R&D directors focused on high-purity organic luminescent intermediates. The use of tetrakis(triphenylphosphine)palladium(0) as the catalyst ensures high turnover numbers even with the sterically demanding substrates involved. The mechanism likely proceeds through oxidative addition of the P-Cl bond to the Pd(0) center, followed by transmetallation with the Grignard reagent and reductive elimination to release the final triarylphosphine product. Crucially, the presence of the electron-donating dimethylamino group on the incoming Grignard reagent requires careful handling to avoid catalyst poisoning, yet the patented conditions demonstrate that high purity can be achieved. This mechanistic pathway effectively minimizes the formation of homocoupling byproducts and unreacted starting materials, ensuring that the final radical precursors possess the structural integrity necessary for consistent photo-induced behavior.
How to Synthesize CP-DMA Efficiently
The synthesis of the flagship compound CP-DMA serves as a prime example of the operational simplicity and robustness of this patented technology. The process begins with the preparation of bis(4-chlorophenyl)phosphine oxide, followed by its conversion to the corresponding chlorophosphine using PCl3 in toluene. The final coupling step employs a reflux condition in THF with a catalytic amount of palladium, driving the reaction to completion while maintaining the integrity of the sensitive functional groups. Detailed standardized synthetic steps for replicating this high-value intermediate are provided in the technical guide below, ensuring reproducibility for process chemists.
- Preparation of Diarylphosphine Oxide via Grignard reaction of chlorobenzene derivatives with diethyl phosphite in THF.
- Chlorination of the phosphine oxide intermediate using PCl3 in toluene to generate the reactive chlorophosphine species.
- Final Palladium-catalyzed cross-coupling with (4-(dimethylamino)phenyl)magnesium bromide to install the electron-donating group.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the transition to this D-A type triarylphosphine platform represents a strategic opportunity to optimize cost reduction in optoelectronic material manufacturing. The synthetic route relies heavily on commodity chemicals such as chlorobenzenes, diethyl phosphite, and phosphorus trichloride, all of which are available in bulk quantities from established global suppliers. This reliance on mature feedstock eliminates the supply chain volatility often associated with exotic or custom-synthesized building blocks, thereby securing a more predictable and continuous flow of raw materials. Furthermore, the reaction conditions utilize standard solvents like THF and toluene, which simplifies solvent recovery and recycling processes, contributing to both economic and environmental efficiency in large-scale production facilities.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences found in traditional radical synthesis significantly lowers the overall processing time and energy consumption. By utilizing a convergent synthesis strategy where the donor and acceptor fragments are prepared separately and joined in a final high-yielding coupling step, the process minimizes the accumulation of impurities that would otherwise require expensive chromatographic purification. The use of a low-loading palladium catalyst further reduces the cost burden associated with precious metals, while the robustness of the reaction conditions suggests that less stringent environmental controls (such as extreme cryogenic temperatures) may be sufficient for certain steps, lowering utility costs.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the decoupling of intermediate production, meaning that key precursors like the diarylphosphine oxides can be stockpiled independently of the final coupling step. This flexibility provides a buffer against demand fluctuations and ensures that reducing lead time for high-purity organic luminescent intermediates is achievable without compromising quality. Additionally, the chemical stability of the intermediates reduces the risk of degradation during storage and transport, a critical factor for maintaining supply continuity in global logistics networks where temperature control can be challenging.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, moving seamlessly from gram-scale laboratory experiments to kilogram and tonne-level commercial production without requiring fundamental changes to the reactor configuration. The waste streams generated are primarily composed of magnesium salts and aqueous phases which are well-understood and manageable within standard wastewater treatment protocols. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing process, aligning with the sustainability goals of modern electronics manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the material's capabilities and limitations.
Q: What distinguishes these D-A type triarylphosphines from traditional triarylmethyl radicals?
A: Unlike traditional triarylmethyl radicals which are limited to orange or near-infrared emission, these D-A type triarylphosphines offer tunable emission properties ranging from blue to red and exhibit superior thermal and electrochemical stability suitable for solid-state applications.
Q: Is the synthesis scalable for industrial production of organic luminescent materials?
A: Yes, the synthetic route utilizes standard industrial reagents such as PCl3 and Grignard reagents in common solvents like THF and toluene, avoiding exotic catalysts and enabling straightforward scale-up from laboratory to commercial tonnage.
Q: How does the photo-induced radical generation mechanism benefit device performance?
A: The ability to generate stable free radicals upon UV illumination allows for dynamic control of paramagnetic and luminescent properties, opening new avenues for photoelectromagnetic devices and advanced optical switching applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-A Type Triarylphosphine Supplier
As the global demand for advanced organic luminescent materials accelerates, partnering with a technically proficient manufacturer is essential for success. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of phosphine chemistry, including inert atmosphere processing and rigorous QC labs that ensure stringent purity specifications for every batch of CP-DMA, DCP-DMA, or TCP-DMA we produce. We understand that the performance of your final optoelectronic device is only as good as the purity of the intermediates used, which is why our quality assurance protocols are designed to detect and eliminate trace impurities that could affect radical stability.
We invite potential partners to engage with our technical team to discuss how this novel synthetic route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your transition to these next-generation radical materials is smooth, compliant, and commercially viable.
