Scalable Synthesis of Chiral Curcumin Analogues Using Novel Tertiary Amine-Thiourea Organocatalysts
Scalable Synthesis of Chiral Curcumin Analogues Using Novel Tertiary Amine-Thiourea Organocatalysts
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral scaffolds found in bioactive natural products, and patent CN102115446A presents a significant advancement in this domain by detailing a method for catalytically synthesizing chiral curcumin analogues. This technology leverages an asymmetric catalytic conjugate addition process, utilizing nitroalkenes and curcumin analogues as primary raw materials within a sophisticated tertiary amine-thiourea organic catalyst system. The reaction proceeds efficiently in common organic solvents over a duration of 0.5 to 15 days at temperatures ranging from -40 to 40°C, yielding conjugate addition products with remarkable stereochemical control. For R&D directors and process chemists, this represents a pivotal shift towards metal-free organocatalysis that maintains high catalytic activity and stereoselectivity, achieving enantioselectivity values up to 97% and yields up to 96%. The versatility of this approach allows for a wide range of reaction substrates, making it a compelling candidate for the development of next-generation anti-inflammatory and antioxidant therapeutic agents.
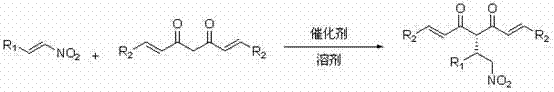
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating curcumin derivatives often rely on transition metal catalysis or stoichiometric chiral auxiliaries, which introduce significant downstream processing challenges and environmental liabilities. The presence of heavy metals necessitates rigorous purification steps to meet stringent regulatory limits for pharmaceutical ingredients, thereby increasing production costs and extending lead times for high-purity pharmaceutical intermediates. Furthermore, many conventional methods struggle to achieve high levels of enantioselectivity without employing cryogenic conditions or expensive chiral ligands that are difficult to recover and recycle. The lack of efficient protocols for the direct asymmetric functionalization of curcumin-like 1,3-diketone systems has historically limited the exploration of their full pharmacological potential, particularly regarding their interaction with biological targets like COX-1 and COX-2 inhibitors where chirality plays a critical role in efficacy.
The Novel Approach
The methodology disclosed in the patent introduces a bifunctional organocatalytic strategy that overcomes these hurdles by employing designed tertiary amine-thiourea catalysts capable of simultaneous substrate activation. This novel approach eliminates the need for transition metals entirely, addressing the growing demand for green chemistry solutions in fine chemical intermediates manufacturing. By operating under mild conditions, often at room temperature, the process reduces energy consumption and simplifies reactor requirements, facilitating easier commercial scale-up of complex pharmaceutical intermediates. The catalyst design incorporates specific aromatic and sulfonyl substituents that create a well-defined chiral environment, directing the nucleophilic attack with high precision to ensure the formation of the desired enantiomer. This strategic innovation not only improves the overall yield and optical purity but also streamlines the supply chain by reducing the dependency on scarce metal resources and complex waste treatment protocols associated with metallic catalysts.
Mechanistic Insights into Tertiary Amine-Thiourea Catalyzed Michael Addition
The core of this technological breakthrough lies in the unique structural architecture of the tertiary amine-thiourea catalyst, which functions through a dual-activation mechanism involving hydrogen bonding and Lewis base interactions. As illustrated in the general catalyst structure, the thiourea moiety acts as a hydrogen bond donor to activate the electrophilic nitroalkene, while the tertiary amine group, often derived from cinchona alkaloids, deprotonates or coordinates with the nucleophilic curcumin analogue. This synergistic effect organizes the transition state in a highly ordered spatial arrangement, effectively lowering the activation energy for the conjugate addition while imposing strict stereochemical constraints. The presence of bulky aromatic substituents on the catalyst backbone further shields one face of the reacting species, ensuring that the nucleophile attacks from the preferred trajectory to generate the chiral center with high fidelity.
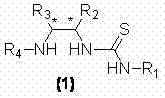
From an impurity control perspective, this mechanism offers substantial advantages by minimizing the formation of diastereomers and regioisomers that typically complicate purification. The high enantioselectivity, reported up to 97% ee in specific embodiments, implies that the crude reaction mixture is already enriched in the target isomer, significantly reducing the burden on downstream chromatographic separation. This level of control is crucial for maintaining the integrity of the impurity profile, a key concern for regulatory compliance in API manufacturing. The catalyst's ability to accommodate various substituents on both the nitroalkene and the curcumin scaffold without significant loss in selectivity suggests a robust catalytic cycle that is tolerant to electronic and steric variations. Such mechanistic stability ensures consistent batch-to-batch reproducibility, a critical factor for reliable chiral curcumin analogue supplier operations aiming for long-term commercial viability.
How to Synthesize Chiral Curcumin Analogues Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for producing these valuable compounds, starting with the preparation of the specialized organocatalyst followed by the conjugate addition reaction. The process is designed to be operationally simple, requiring standard laboratory equipment and avoiding the need for inert atmosphere techniques or extreme pressure conditions. Detailed below is the strategic workflow that integrates catalyst preparation with the final coupling step, ensuring optimal performance and yield. For comprehensive operational details and specific parameter optimization, please refer to the standardized synthesis steps provided in the guide below.
- Prepare the reaction mixture by dissolving nitroolefin and curcumin analogue substrates in a suitable organic solvent such as dichloromethane or tetrahydrofuran.
- Add the chiral tertiary amine-thiourea organocatalyst to the solution, ensuring a molar ratio of catalyst to substrate between 1% and 100% depending on desired kinetics.
- Stir the reaction at temperatures ranging from -40°C to 40°C for 0.5 to 15 days, followed by concentration and purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic technology translates into tangible operational efficiencies and risk mitigation strategies across the manufacturing value chain. The elimination of transition metals removes the necessity for expensive metal scavenging resins and extensive analytical testing for residual metals, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. Additionally, the use of readily available starting materials such as substituted nitroalkenes and curcumin derivatives ensures a stable supply base, reducing the vulnerability to raw material shortages that often plague specialty chemical sectors. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without specialized corrosion-resistant coatings, further lowering capital expenditure requirements for facility upgrades.
- Cost Reduction in Manufacturing: The metal-free nature of this catalytic system fundamentally alters the cost structure by removing expensive noble metal catalysts and the associated waste disposal fees. Since the organic catalysts can be synthesized from abundant precursors like quinine and chiral diamines, the raw material costs are significantly lower compared to rhodium or palladium-based systems. Furthermore, the simplified workup procedure, which often involves merely concentrating the reaction mixture and performing a single column chromatography, reduces solvent consumption and labor hours. These factors combine to create a leaner production model that enhances margin potential without compromising on the quality or purity of the final active ingredient.
- Enhanced Supply Chain Reliability: By relying on organocatalysis, manufacturers can decouple their production schedules from the volatile market dynamics of precious metals, which are subject to geopolitical instability and mining constraints. The robustness of the reaction across a wide temperature range (-40 to 40°C) provides flexibility in scheduling and logistics, allowing for production in facilities with varying levels of thermal control infrastructure. This adaptability ensures continuous supply continuity even during periods of utility fluctuation or equipment maintenance, making it a resilient choice for long-term contracts requiring guaranteed delivery of high-purity OLED material or pharmaceutical precursors.
- Scalability and Environmental Compliance: The homogeneous nature of the reaction system facilitates straightforward scale-up from gram-scale laboratory experiments to multi-kilogram pilot runs without encountering mass transfer limitations common in heterogeneous catalysis. The absence of toxic heavy metals aligns perfectly with increasingly stringent environmental regulations regarding effluent discharge and worker safety, simplifying the permitting process for new production lines. Moreover, the high atom economy and selectivity of the process minimize the generation of hazardous byproducts, supporting corporate sustainability goals and reducing the overall environmental footprint of the chemical manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology, derived directly from the patent's experimental data and beneficial effects. These insights are intended to clarify the practical implications of adopting this catalytic system for your specific application needs. Understanding these nuances will help stakeholders make informed decisions about integrating this methodology into their existing R&D pipelines.
Q: What are the primary advantages of using tertiary amine-thiourea catalysts for curcumin synthesis?
A: These catalysts offer metal-free conditions, eliminating toxic heavy metal residues, and provide exceptional enantioselectivity up to 97% under mild reaction temperatures.
Q: Can this synthetic method be scaled for industrial production of API intermediates?
A: Yes, the process utilizes homogeneous systems with simple workup procedures like concentration and chromatography, making it highly suitable for commercial scale-up without complex equipment.
Q: What is the substrate scope for the nitroalkene component in this reaction?
A: The method demonstrates broad substrate tolerance, successfully reacting with aromatic nitroalkenes containing various substituents like halogens, methoxy groups, and alkyl chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Curcumin Analogue Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this organocatalytic technology in advancing the development of novel therapeutic agents and functional materials. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market-ready product is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify enantiomeric excess and chemical identity. We understand that consistency is key in the pharmaceutical supply chain, and our optimized processes are designed to deliver batch-after-batch reliability that meets the exacting standards of global regulatory bodies.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next project, whether it involves agrochemical intermediates or complex drug candidates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in asymmetric organocatalysis can drive value and innovation for your organization.
