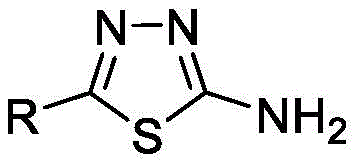
Advanced Green Synthesis of 2-Amino-5-Substituted-1,3,4-Thiadiazole Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical and agrochemical industries are constantly seeking more sustainable and efficient pathways for synthesizing critical heterocyclic scaffolds, and the technology disclosed in patent CN110724115B represents a significant leap forward in this domain. This patent details a novel method for preparing 2-amino-5-substituted-1,3,4-thiadiazole derivatives, a class of compounds renowned for their extensive biological activities ranging from antiviral and antibacterial properties to anticancer and anxiolytic effects. Traditionally, the synthesis of these valuable intermediates has been plagued by the use of harsh reagents and complex purification steps, but this new approach utilizes a Deep Eutectic Solvent (DES) system composed of choline chloride and urea. By replacing conventional volatile organic solvents and toxic catalysts with this biodegradable, recyclable medium, the process not only aligns with modern green chemistry principles but also offers a robust platform for the commercial scale-up of complex pharmaceutical intermediates. The ability to operate under mild conditions while achieving high conversion rates makes this technology particularly attractive for R&D directors focused on impurity control and process safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 1,3,4-thiadiazole derivatives has relied heavily on cyclodehydration reactions mediated by phosphorus oxychloride (POCl3), a reagent that presents severe challenges for both operational safety and environmental compliance. The use of POCl3 is associated with high toxicity and extreme corrosivity, requiring specialized equipment and rigorous safety protocols that drive up capital expenditure and operational costs. Furthermore, the reaction process involving phosphorus oxychloride is often complex, necessitating long reaction times and generating substantial amounts of acidic, phosphorous-containing wastewater that requires expensive treatment before disposal. From a supply chain perspective, the reliance on such hazardous materials introduces significant regulatory risks and potential disruptions, as the handling and transport of corrosive liquids are subject to increasingly stringent global regulations. Additionally, the post-reaction workup in conventional methods is often cumbersome, involving multiple neutralization and extraction steps that can lead to product loss and lower overall yields, thereby impacting the economic viability of large-scale manufacturing.
The Novel Approach
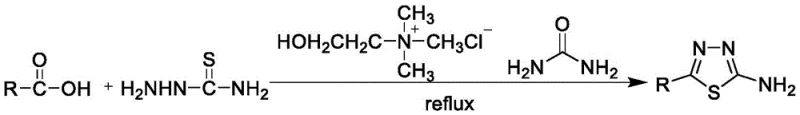
In stark contrast to these legacy methods, the innovative protocol described in CN110724115B utilizes a eutectic solvent formed by the simple mixing of choline chloride and urea, which serves a dual function as both the reaction medium and the catalyst. This approach eliminates the need for any additional organic solvents or toxic dehydrating agents, fundamentally simplifying the reaction setup and reducing the chemical footprint of the manufacturing process. The reaction proceeds efficiently at a moderate temperature of 80°C, significantly lowering energy consumption compared to high-temperature reflux conditions often required in traditional syntheses. The simplicity of the post-treatment process is another major advantage; upon completion, the product can be precipitated simply by adjusting the pH with ammonia water, allowing for easy filtration and washing with ice water. This streamlined workflow not only reduces labor intensity but also minimizes the generation of liquid waste, as the mother liquor containing the DES can be recovered and reused, creating a closed-loop system that enhances overall process sustainability.
Mechanistic Insights into DES-Catalyzed Cyclization
The efficacy of this synthetic route lies in the unique physicochemical properties of the choline chloride-urea deep eutectic solvent, which creates a highly organized hydrogen-bonding network capable of activating the reactants without the need for strong mineral acids. In this system, the urea molecules interact strongly with the chloride anions of the choline salt, resulting in a liquid mixture with a melting point much lower than that of either individual component. This hydrogen-bond donor-acceptor interaction facilitates the activation of the carboxylic acid carbonyl group, making it more susceptible to nucleophilic attack by the thiosemicarbazide. The DES environment stabilizes the transition states involved in the cyclization and dehydration steps, effectively lowering the activation energy barrier and allowing the reaction to proceed rapidly at 80°C. Furthermore, the polar nature of the solvent ensures excellent solubility for the polar intermediates, preventing premature precipitation and ensuring homogeneous reaction conditions that are critical for consistent product quality and high purity.
From an impurity control perspective, the mild acidity and specific solvation properties of the DES help suppress side reactions that are common in harsh acidic media, such as the hydrolysis of sensitive functional groups or the formation of polymeric byproducts. The absence of strong oxidizing or chlorinating agents like POCl3 means that there is no risk of introducing chlorine-containing impurities or phosphorous residues into the final API intermediate, which simplifies the downstream purification requirements. The reaction monitoring via Thin Layer Chromatography (TLC) indicates a clean conversion profile, where the disappearance of the carboxylic acid starting material correlates directly with the formation of the desired thiadiazole ring. This mechanistic clarity provides R&D teams with confidence in the reproducibility of the process, as the reaction parameters are well-defined and less prone to the variability often seen in multiphase systems utilizing volatile organic solvents.
How to Synthesize 2-Amino-5-Substituted-1,3,4-Thiadiazole Efficiently
The implementation of this green synthesis protocol requires precise control over the preparation of the eutectic solvent and the subsequent addition of reactants to ensure optimal yield and purity. The process begins with the formation of the DES by heating choline chloride and urea, followed by the controlled addition of the specific carboxylic acid derivative and thiosemicarbazide. Maintaining the reaction temperature at 80°C is crucial for balancing reaction kinetics with thermal stability, while the final isolation step relies on careful pH adjustment to maximize product recovery. For detailed operational parameters and specific stoichiometric ratios tailored to different substrates, please refer to the standardized synthesis guide below.
- Prepare the Deep Eutectic Solvent (DES) by mixing choline chloride and urea in a molar ratio of 1: 2 and stirring at 80°C until a colorless transparent solution forms.
- Cool the DES to room temperature, add the specific carboxylic acid and thiosemicarbazide, then heat the mixture to 80°C and reflux until TLC indicates reaction completion.
- Cool the reaction mixture, adjust pH to 8-9 with ammonia water under ice bath cooling to precipitate the solid product, then filter, wash with ice water, and dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this DES-based technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The elimination of hazardous reagents like phosphorus oxychloride removes a significant liability from the supply chain, reducing the costs associated with special storage, handling, and disposal of dangerous goods. Moreover, the ability to recycle the solvent system multiple times without significant loss of activity means that the consumption of raw materials per kilogram of product is drastically reduced, leading to substantial cost savings in variable manufacturing expenses. The simplified workup procedure, which avoids complex extractions and distillations, shortens the batch cycle time, thereby increasing the throughput capacity of existing manufacturing facilities without the need for major capital investment in new equipment.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dual role of the DES as both solvent and catalyst, which eliminates the need to purchase and dispose of large volumes of organic solvents and separate catalytic reagents. By recovering and reusing the choline chloride-urea mixture from the filtrate, the effective cost of the reaction medium approaches zero over multiple batches, significantly lowering the overall cost of goods sold (COGS). Additionally, the reduction in waste treatment costs, due to the absence of phosphorous waste and organic solvent emissions, further enhances the economic competitiveness of this route compared to traditional methods.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically choline chloride, urea, and various carboxylic acids, are commodity chemicals with robust and stable global supply chains, minimizing the risk of raw material shortages. Unlike specialized catalysts or reagents that may have single-source suppliers, the components of this DES system are widely available from multiple vendors, ensuring supply continuity even during market fluctuations. The simplified logistics of handling non-hazardous solids and liquids also streamline the inbound supply chain, reducing lead times and administrative burdens associated with hazardous material compliance.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward because the reaction does not involve exothermic hazards or gas evolution that typically complicate reactor design. The absence of volatile organic compounds (VOCs) ensures that the manufacturing facility remains compliant with increasingly strict environmental regulations regarding air emissions and solvent usage. This 'green' credential is increasingly valuable for securing contracts with multinational pharmaceutical companies that have aggressive sustainability targets, positioning suppliers who adopt this technology as preferred partners for long-term strategic sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this deep eutectic solvent technology for thiadiazole synthesis. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for feasibility assessments. Understanding these details is essential for evaluating the fit of this technology within your existing manufacturing portfolio.
Q: What are the primary advantages of using Deep Eutectic Solvents (DES) over traditional phosphorus oxychloride methods?
A: The DES method eliminates the use of highly toxic and corrosive phosphorus oxychloride (POCl3), significantly reducing hazardous waste generation and simplifying post-reaction workup procedures while maintaining high yields.
Q: Can the choline chloride-urea solvent system be recycled for multiple batches?
A: Yes, the patent data confirms that the filtrate containing the DES can be recovered and reused for subsequent reactions, maintaining good catalytic activity and yield efficiency over multiple cycles.
Q: What types of substituents are compatible with this synthetic route?
A: The method demonstrates broad substrate scope, successfully accommodating various carboxylic acids including alkyl, aryl, halogenated phenyl, and heterocyclic acids to produce diverse 2-amino-5-substituted-1,3,4-thiadiazole derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-Substituted-1,3,4-Thiadiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry technologies like the DES-catalyzed synthesis of thiadiazoles and are fully equipped to bring this innovation to commercial reality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are designed to handle complex heterocyclic chemistry with stringent purity specifications, supported by rigorous QC labs that utilize advanced analytical techniques to guarantee the quality of every batch. We understand that consistency and reliability are paramount in the pharmaceutical supply chain, and our commitment to process excellence ensures that we can meet the demanding requirements of global drug developers.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project, whether it involves developing a new API intermediate or optimizing an existing supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this greener methodology for your specific molecule. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive proposal that demonstrates how we can support your goals for cost reduction and sustainable manufacturing.
