Advanced Industrial Synthesis Of Cisatracurium Besylate For Global Pharmaceutical Manufacturing
Advanced Industrial Synthesis Of Cisatracurium Besylate For Global Pharmaceutical Manufacturing
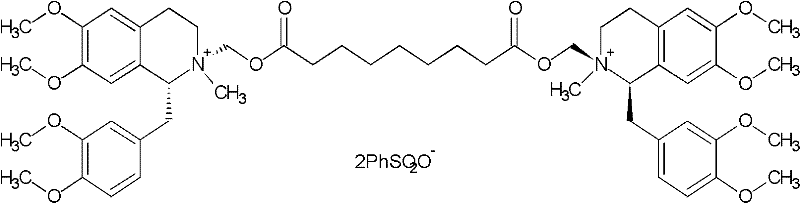
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex neuromuscular blocking agents, and patent CN102249998A presents a significant breakthrough in the preparation of Cisatracurium Besylate. This specific patent details a refined synthetic route that addresses the longstanding challenges of low yield and impurity control associated with earlier methodologies. By implementing a strategic recrystallization of the key oxalate intermediate, the process achieves a remarkable product purity exceeding 98.6% with single impurity levels maintained below 0.3%. This technical advancement is not merely a laboratory curiosity but represents a viable industrial solution that enhances the reliability of the global supply chain for this critical API intermediate. The methodology leverages precise chiral resolution starting materials and optimized reaction conditions to ensure consistent quality output suitable for stringent regulatory environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Cisatracurium Besylate has been plagued by inefficient separation processes and poor overall yields that drive up manufacturing costs significantly. Traditional approaches often involved separating the desired cis-isomer from a mixture of ten possible isomers of Atracurium Besylate, where the target compound constitutes only about 15% of the total mixture, inherently limiting the maximum theoretical yield. Furthermore, prior art methods, such as those described in patent WO2010/128518A2, suffered from critical process flaws where the intermediate oxalate salt was reacted directly without adequate purification. This lack of intermediate purification allowed impurities to carry through to the final column chromatography stage, drastically increasing the difficulty of separation and resulting in final yields as low as 5%. Additionally, the use of sodium chloride solutions in work-up procedures introduced chloride ion contamination, necessitating complex and costly removal steps that further compromised the economic viability and scalability of the process.
The Novel Approach
In stark contrast to these inefficient legacy methods, the novel approach outlined in CN102249998A introduces a pivotal purification step that fundamentally alters the process economics and product quality profile. The core innovation lies in the isolation and recrystallization of the intermediate (1R,1'R)-2,2'-(3,11-dioxo-4,10-dioxa-1,13-subtridecyl) di[1,2,3,4-tetralin-6,7-dimethoxy-1-(3,4-dimethoxyl)benzyl]isoquinoline as an oxalate salt in absolute ethanol. This specific recrystallization step elevates the purity of the intermediate to above 99.5% before it undergoes the final quaternization reaction with methyl benzenesulfonate. By ensuring the intermediate is of exceptional purity, the burden on the final silica gel column chromatography is significantly reduced, allowing for a much more efficient separation of the target cis-isomer. Consequently, this streamlined workflow boosts the overall yield from the starting material to the final product to approximately 30%, representing a six-fold improvement over the problematic 5% yield of previous industrial attempts.
Mechanistic Insights into Chiral Resolution and Selective Crystallization
The success of this synthesis relies heavily on the precise control of stereochemistry beginning with the chiral starting material, R-tetrahydropapaverine, which possesses a chiral purity of greater than 99.5%. The reaction mechanism involves a Michael-type addition where the secondary amine of the tetrahydroisoquinoline ring attacks the acrylate double bonds of 1,5-pentanediol diacrylate. This step is conducted in glacial acetic acid at a controlled temperature range of 65-75°C to facilitate the formation of the bis-quaternary precursor while minimizing side reactions. The subsequent formation of the oxalate salt is critical because the oxalate anion forms a highly ordered crystal lattice with the specific (1R,1'R) stereoisomer, effectively excluding other geometric or optical isomers during the crystallization process. This phenomenon of diastereomeric salt formation is the cornerstone of the purification strategy, leveraging solubility differences in absolute ethanol to reject impurities that would otherwise persist through the synthesis.
Following the formation of the quaternary ammonium salt (Compound V), the final separation utilizes a highly specialized silica gel column chromatography system tailored to the unique polarity of the besylate salt. The eluent system comprises a specific ratio of dichloromethane and absolute ethanol, acidified with a precise concentration of benzenesulfonic acid (0.03-0.42 g/L). The addition of benzenesulfonic acid to the mobile phase is a crucial mechanistic detail that suppresses tailing and improves peak resolution by interacting with residual silanol groups on the silica surface. This ensures that the target (1R,1'R,2R,2'R) isomer is cleanly separated from the (1R,1'R,2S,2'S) and other diastereomers. The rigorous control of the silica gel pH (3.5-5) and mesh size (200-500 mesh) further optimizes the surface area available for interaction, ensuring that the final product meets the stringent purity specifications required for parenteral anesthesia applications without the need for excessive reprocessing.
How to Synthesize Cisatracurium Besylate Efficiently
The implementation of this synthesis protocol requires strict adherence to the specified reaction parameters to replicate the high yields and purity reported in the patent documentation. Operators must ensure that the initial liberation of the free base from the leucine salt is complete before proceeding to the acrylation step, as any residual salt can interfere with the stoichiometry of the reaction. The recrystallization of the oxalate intermediate must be performed with high-purity absolute ethanol to maximize the rejection of impurities, and the temperature control during the quaternization reaction with methyl benzenesulfonate must be maintained between 20-35°C to prevent degradation. Detailed standard operating procedures regarding solvent volumes, stirring rates, and filtration techniques are essential for scaling this process from laboratory to commercial production. For a comprehensive breakdown of the specific operational steps and safety considerations, please refer to the standardized guide below.
- Dissolve R-tetrahydropapaverine-N-acetyl-L-leucine salt in water and liberate the free base using a weak base like ammonia water to obtain R-tetrahydropapaverine.
- React R-tetrahydropapaverine with 1,5-pentanediol diacrylate in acetic acid at 65-75°C to form the intermediate, then convert to oxalate salt and purify via recrystallization in absolute ethanol.
- React the purified oxalate intermediate with methyl benzenesulfonate in acetonitrile, followed by silica gel column chromatography using a specific dichloromethane-ethanol-benzenesulfonic acid eluent to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this optimized synthesis route offers tangible benefits that extend far beyond simple chemical yield improvements. The ability to produce Cisatracurium Besylate with a stable yield of 30% compared to the historical 5% benchmark translates directly into a drastic reduction in the consumption of raw materials per kilogram of finished product. This efficiency gain mitigates the volatility of raw material pricing and reduces the overall cost of goods sold, making the supply of this critical muscle relaxant more economically sustainable for generic drug manufacturers. Furthermore, the simplified purification workflow reduces the dependency on extensive chromatographic resources, which are often bottlenecks in large-scale API manufacturing facilities. By minimizing the number of processing steps and improving the robustness of the intermediate purification, the risk of batch failure is significantly lowered, ensuring a more reliable and continuous supply stream for downstream formulation partners.
- Cost Reduction in Manufacturing: The elimination of inefficient separation steps and the substantial increase in overall process yield lead to significant cost savings in raw material utilization and waste disposal. By avoiding the carry-over of impurities into the final chromatography stage, the lifespan of expensive silica gel columns is extended, and the volume of organic solvents required for elution is reduced. This streamlined approach removes the need for complex ion-exchange treatments to remove chloride contaminants, further lowering utility and chemical costs. Ultimately, the process transforms a previously marginal manufacturing route into a commercially viable operation with a much healthier profit margin.
- Enhanced Supply Chain Reliability: The robustness of the recrystallization step ensures that intermediate quality is consistent, reducing the variability that often leads to supply disruptions in complex chiral syntheses. Because the process does not rely on the low-yield separation of isomers from a racemic mixture, the throughput capacity of the manufacturing plant is effectively multiplied without requiring additional capital investment in new reactors. This increased capacity allows suppliers to build larger safety stocks and respond more agilely to fluctuations in market demand for neuromuscular blocking agents. The stability of the process also simplifies technology transfer between manufacturing sites, facilitating a more resilient global supply network.
- Scalability and Environmental Compliance: The use of standard solvents like dichloromethane, ethanol, and acetonitrile, combined with the reduction in total solvent volume due to higher yields, simplifies solvent recovery and recycling operations. The process avoids the generation of heavy metal waste or difficult-to-treat saline effluents associated with older chloride-based work-up methods. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental compliance burden on manufacturing facilities. The scalability is further supported by the use of conventional unit operations such as crystallization and filtration, which are well-understood and easily scaled from pilot plants to multi-ton commercial production lines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Cisatracurium Besylate using this patented methodology. These answers are derived directly from the experimental data and process descriptions found in the source patent to ensure accuracy and relevance for technical decision-makers. Understanding these nuances is critical for evaluating the feasibility of integrating this supply source into your existing procurement strategy. We encourage potential partners to review these details closely to appreciate the depth of process optimization achieved.
Q: What is the primary advantage of the oxalate recrystallization step in this synthesis?
A: The recrystallization of the intermediate oxalate salt in absolute ethanol significantly increases purity to over 99.5% before the final quaternization step, preventing impurity carry-over that typically complicates downstream chromatography and reduces overall yield.
Q: How does this method improve yield compared to previous industrial processes?
A: By optimizing the purification of the intermediate and utilizing a specific column chromatography eluent system, this method achieves a final yield of approximately 30%, which is a substantial improvement over the 5% yield reported in prior art methods like WO2010/128518A2.
Q: What specific chromatography conditions are required for the final separation?
A: The process requires silica gel with a pH range of 3.5-5 and an eluent composed of dichloromethane, absolute ethanol, and a precise amount of benzenesulfonic acid to effectively separate the cis-isomer from other stereoisomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cisatracurium Besylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the production of life-saving anesthetic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated purification techniques described in patent CN102249998A can be successfully translated to an industrial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced HPLC and chiral separation capabilities to verify that every batch meets the <98.6% purity and <0.3% single impurity targets. Our commitment to quality assurance means that we do not just supply chemicals; we deliver validated process reliability that supports your regulatory filings and commercial launch timelines.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this higher-yield manufacturing method. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to serve as your long-term strategic partner. Contact us today to secure a stable supply of high-quality Cisatracurium Besylate and gain a competitive edge in the pharmaceutical market.
