Advanced Vilsmeier Cyclization for High-Purity 3-Formyl-4-Halopyridin-2-One Pharmaceutical Intermediates
Advanced Vilsmeier Cyclization for High-Purity 3-Formyl-4-Halopyridin-2-One Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust synthetic routes for heterocyclic building blocks, particularly pyridin-2-one derivatives which serve as critical scaffolds in numerous bioactive molecules. Patent CN101302191B introduces a groundbreaking methodology for synthesizing 3-formyl-4-halogenated pyridin-2(1H)-one compounds, addressing long-standing challenges in regioselectivity and yield. This technology leverages a modified Vilsmeier-Haack reaction condition to transform N-substituted-alpha-acetyl-beta-(N,N-dimethyl)acrylamide precursors directly into the desired heterocyclic core. For R&D directors and procurement specialists, this represents a significant opportunity to streamline the supply of high-purity pharmaceutical intermediates. The process operates under atmospheric pressure with temperatures ranging from 80 to 140°C, utilizing readily available reagents like phosphorus oxychloride or phosphorus tribromide in DMF. By eliminating the need for exotic catalysts or complex multi-step sequences, this innovation offers a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing while ensuring consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyridin-2(1H)-one compounds has relied on methodologies that present significant operational and economic drawbacks for large-scale production. Traditional approaches often involve pyridinium salt chemistry or N-alkylation reactions which can be cumbersome and generate substantial waste. Furthermore, ring-closing strategies such as the Guareschi-Thorpe reaction or intramolecular Dieckmann condensation frequently suffer from narrow substrate scope and苛刻 reaction conditions that complicate safety protocols. A critical failure point in existing literature, such as earlier Vilsmeier applications on acetoacetyl arylamines, is the lack of regiocontrol, typically resulting in a roughly 1:1 molar mixture of 3-formyl and 5-formyl isomers. This inseparable mixture drastically reduces the effective yield of the target molecule and necessitates expensive purification steps or total batch rejection, thereby inflating the cost of goods sold. Additionally, many conventional routes require expensive transition metal catalysts or high-pressure equipment, creating bottlenecks in the supply chain for reliable agrochemical intermediate suppliers and pharma partners alike.
The Novel Approach
The patented method described in CN101302191B fundamentally shifts the paradigm by utilizing specific N-substituted-enaminone substrates that undergo highly selective cyclization under Vilsmeier conditions. Instead of producing isomeric mixtures, this novel approach directs the formation of the halogen and aldehyde groups specifically to the 4 and 3 positions of the pyridin-2-one ring, respectively. This regioselectivity is achieved through the unique electronic properties of the alpha-acetyl-beta-(N,N-dimethyl)acrylamide starting material, which facilitates a clean cyclization pathway. The process is remarkably efficient, with reported yields reaching as high as 90 percent depending on the specific substituent, a stark contrast to the poor conversions seen in older methods. Operationally, the simplicity is unparalleled; the reaction proceeds at normal pressure using standard glassware, and the workup involves basic extraction and chromatography. 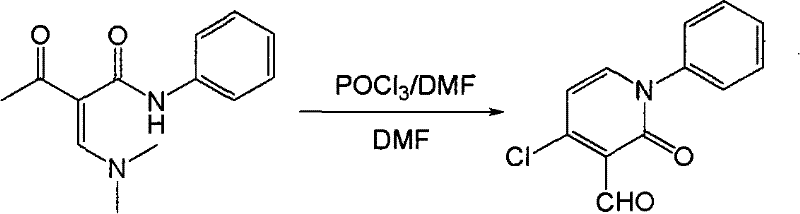 As illustrated in the reaction scheme, the transformation from the linear enaminone to the cyclic pyridone is direct and atom-economical regarding the core structure formation. This efficiency translates directly to commercial advantages, offering a streamlined path for the commercial scale-up of complex pharmaceutical intermediates without the burden of separating difficult isomers.
As illustrated in the reaction scheme, the transformation from the linear enaminone to the cyclic pyridone is direct and atom-economical regarding the core structure formation. This efficiency translates directly to commercial advantages, offering a streamlined path for the commercial scale-up of complex pharmaceutical intermediates without the burden of separating difficult isomers.
Mechanistic Insights into Vilsmeier-Mediated Cyclization and Halogenation
The core of this synthetic breakthrough lies in the dual functionality of the Vilsmeier reagent, generated in situ from phosphorus oxychloride (POCl3) or phosphorus tribromide (PBr3) and N,N-dimethylformamide (DMF). Mechanistically, the reaction initiates with the formation of the chloroiminium or bromoiminium salt, a potent electrophile that attacks the electron-rich enaminone system. Unlike standard formylation where the reagent merely adds a formyl group, here it acts as both a dehydrating agent and a halogen source. The initial attack likely occurs at the beta-carbon of the enaminone, followed by an intramolecular nucleophilic attack by the amide nitrogen onto the activated carbonyl carbon, closing the six-membered ring. The presence of the acetyl group at the alpha-position is crucial, as it provides the necessary carbon framework for the aldehyde functionality at the 3-position after hydrolysis during workup. The halogen atom (Cl or Br) is incorporated at the 4-position, derived directly from the phosphorus halide reagent. This concerted mechanism ensures that the halogen and aldehyde functionalities are installed simultaneously with the ring closure, preventing the formation of side products that plague stepwise syntheses.
From an impurity control perspective, this mechanism offers superior predictability compared to radical halogenation or electrophilic substitution on pre-formed rings. The specificity of the Vilsmeier reagent towards the enaminone double bond minimizes poly-halogenation or over-formylation. The reaction conditions, specifically the temperature range of 80 to 140°C, are optimized to drive the cyclization to completion while avoiding thermal degradation of the sensitive aldehyde moiety. Furthermore, the use of DMF as both solvent and reagent component ensures a homogeneous reaction medium, which is critical for heat transfer and mass transfer in large-scale reactors. 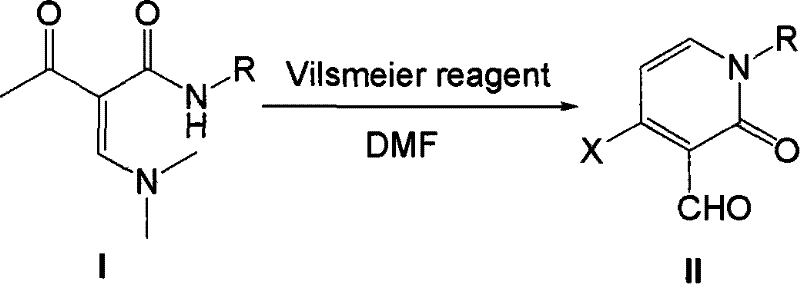 The general reaction scheme demonstrates the versatility of this mechanism across different N-substituents (R groups), ranging from simple alkyls to complex aryl groups. This mechanistic robustness means that process chemists can confidently predict the outcome for new analogs, reducing the risk of failed batches during tech transfer. The ability to switch between POCl3 and PBr3 allows for the precise installation of either chlorine or bromine, providing flexibility for downstream cross-coupling reactions often required in medicinal chemistry campaigns.
The general reaction scheme demonstrates the versatility of this mechanism across different N-substituents (R groups), ranging from simple alkyls to complex aryl groups. This mechanistic robustness means that process chemists can confidently predict the outcome for new analogs, reducing the risk of failed batches during tech transfer. The ability to switch between POCl3 and PBr3 allows for the precise installation of either chlorine or bromine, providing flexibility for downstream cross-coupling reactions often required in medicinal chemistry campaigns.
How to Synthesize 3-Formyl-4-Halopyridin-2-One Efficiently
Implementing this synthesis requires careful attention to the preparation of the Vilsmeier reagent and the addition sequence to manage exotherms effectively. The patent outlines a standardized procedure where the reagent is pre-formed at low temperatures (0-10°C) before the substrate is introduced. This controlled addition prevents localized overheating which could lead to polymerization or decomposition of the enaminone. Following the addition, the reaction mixture is heated to promote cyclization, with the exact temperature and time tuned based on the electronic nature of the N-substituent. For detailed operational parameters, stoichiometry, and specific workup procedures tailored to your facility's capabilities, please refer to the standardized guide below.
- Prepare the Vilsmeier reagent by mixing POCl3 or PBr3 with DMF at 0-10°C in a 1: 3 molar ratio.
- Add the enaminone substrate solution to the cooled reagent and heat the mixture to 80-140°C for cyclization.
- Quench the reaction in brine, extract with organic solvent, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical yield. The primary driver for cost optimization is the elimination of expensive transition metal catalysts and the reduction of unit operations. Traditional methods often require palladium or copper catalysts for halogenation, which not only add raw material costs but also necessitate rigorous metal scavenging steps to meet pharmaceutical purity standards. By utilizing commodity chemicals like POCl3 and DMF, this process significantly lowers the direct material cost. Furthermore, the high regioselectivity eliminates the need for preparative HPLC or extensive recrystallization campaigns to separate isomers, drastically reducing solvent consumption and processing time. This simplification of the downstream processing directly contributes to cost reduction in pharmaceutical intermediate manufacturing, allowing for more competitive pricing in a crowded market.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the use of bulk commodity reagents and the avoidance of precious metals. Since the reaction proceeds with high conversion and selectivity, the overall yield loss is minimized, meaning less raw material is wasted per kilogram of final product. The simplified workup, involving basic aqueous quenching and extraction, reduces the demand for specialized resins or adsorbents used in metal removal. Additionally, the atmospheric pressure operation removes the capital expenditure associated with autoclaves or high-pressure reactors, lowering the barrier to entry for contract manufacturing organizations. These factors combine to create a lean manufacturing process that maximizes output while minimizing variable costs.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available starting materials. The N-substituted-enaminone precursors and Vilsmeier reagents are standard inventory items for most fine chemical suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in utility supplies or environmental controls, ensuring consistent batch-to-batch quality. This reliability is crucial for maintaining continuous supply lines for downstream API synthesis, preventing costly production stoppages. By securing a source of these intermediates produced via this stable method, companies can mitigate the risks associated with volatile raw material markets.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the absence of hazardous high-pressure steps and the use of common solvents. The reaction generates minimal heavy metal waste, simplifying effluent treatment and aligning with increasingly stringent environmental regulations. The high atom economy of the cyclization step means less chemical waste is generated per unit of product, supporting sustainability goals. Moreover, the ability to run the reaction at concentrations up to 0.5 mol/L allows for efficient use of reactor volume, maximizing throughput in existing facilities without the need for major infrastructure upgrades.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for integrating it into your development pipeline. The following questions address common concerns regarding the practical application of this patent technology, focusing on yield consistency, substrate scope, and regulatory compliance. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy for your technical assessments.
Q: What is the primary advantage of this Vilsmeier cyclization method over traditional pyridone synthesis?
A: Unlike traditional methods that often yield mixtures of 3-formyl and 5-formyl isomers or require harsh conditions, this patented process achieves high regioselectivity for the 3-formyl-4-halo structure with yields up to 90% under mild atmospheric pressure.
Q: Can this process be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the process utilizes standard reagents like POCl3 and DMF at atmospheric pressure and temperatures between 80-140°C, making it highly suitable for large-scale commercial manufacturing without specialized high-pressure equipment.
Q: What types of substituents are compatible with this synthetic route?
A: The method demonstrates broad substrate tolerance, successfully accommodating various N-substituents including phenyl, substituted phenyl (with methyl, chloro, methoxy groups), and alkyl chains, ensuring versatility for diverse drug discovery programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Formyl-4-Halopyridin-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality heterocyclic intermediates in modern drug discovery. Our technical team has thoroughly analyzed the potential of the Vilsmeier cyclization route described in CN101302191B and is fully prepared to leverage this chemistry for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to pilot plant is seamless. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including residual solvent analysis and heavy metal testing, to guarantee that every batch meets global regulatory standards. We understand that consistency is key, and our process engineers are dedicated to optimizing this specific route to maximize yield and minimize impurities for your specific application.
We invite you to collaborate with us to unlock the full potential of this synthetic method for your pipeline. Whether you require custom synthesis of specific analogs or large-scale supply of the core scaffold, our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. Let us be your partner in driving efficiency and innovation in your supply chain.
