Revolutionizing Heteroaryl Sulfide Production: A Catalyst-Free Green Chemistry Approach for Commercial Scale
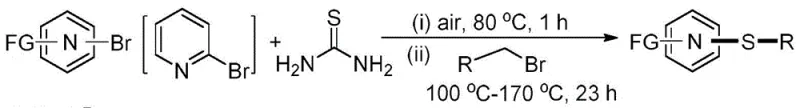
The pharmaceutical and agrochemical industries are constantly seeking more sustainable and cost-effective pathways for constructing sulfur-containing heterocycles, which are pivotal motifs in bioactive molecules. A groundbreaking development in this sector is detailed in Chinese Patent CN111559976B, which discloses a novel, green synthetic method for heteroaryl sulfides. This technology represents a paradigm shift by enabling the direct, one-pot selective synthesis of asymmetric heteroaryl thioethers using 2-bromopyridine, thiourea, and substituted benzyl bromides. Unlike conventional methods that rely on hazardous thiols and expensive transition metal catalysts, this invention operates under catalyst-free, solvent-free, and additive-free conditions. The process utilizes air as the reaction atmosphere, drastically simplifying the operational requirements and enhancing the safety profile for large-scale manufacturing. For R&D directors and procurement managers alike, this patent offers a compelling solution to reduce the environmental footprint while maintaining high selectivity for asymmetric products.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of aryl alkyl sulfides or heteroaryl sulfides has relied heavily on the nucleophilic substitution of aryl halides with thiols or thiolates. However, this classical approach suffers from severe drawbacks that hinder efficient commercial production. Thiols and thiophenols are notoriously toxic, possessing extremely unpleasant odors that require specialized containment facilities and rigorous safety protocols, thereby inflating operational costs. Moreover, many traditional cross-coupling reactions necessitate the use of precious metal catalysts such as palladium or copper, along with phosphine ligands, which are not only expensive but also difficult to remove completely from the final API intermediate. Residual heavy metals pose a significant regulatory hurdle, often requiring additional purification steps like chromatography or recrystallization, which inevitably lower the overall yield and increase waste generation. Furthermore, these reactions often demand strictly anhydrous conditions and inert gas protection, adding complexity to the reactor setup and limiting scalability.
The Novel Approach
The methodology described in patent CN111559976B elegantly circumvents these industrial pain points by employing thiourea as a stable, low-toxicity sulfur source. This innovative route avoids the direct handling of volatile and malodorous thiols entirely. By reacting 2-bromopyridine with thiourea followed by alkylation with benzyl bromides, the process achieves high selectivity for asymmetric thioethers in a streamlined fashion. The absence of external catalysts and solvents means the reaction mixture is inherently cleaner, simplifying the work-up procedure to basic column chromatography or crystallization. This "green chemistry" approach not only aligns with modern environmental regulations but also offers substantial economic benefits by reducing raw material costs and energy consumption associated with solvent recovery. The robustness of the reaction under air atmosphere further demonstrates its potential for seamless integration into existing manufacturing lines without the need for specialized inert atmosphere equipment.
Mechanistic Insights into Thiourea-Mediated Nucleophilic Substitution
The core of this synthetic breakthrough lies in the unique reactivity of thiourea as a sulfur transfer agent. In the initial stage, 2-bromopyridine reacts with thiourea at moderate temperatures (around 80°C) to likely form an isothiouronium salt intermediate. This activation step is crucial as it converts the relatively stable thiourea into a potent nucleophile capable of displacing the halide on the pyridine ring. The subsequent addition of the alkylating agent, such as substituted benzyl bromide, triggers a second nucleophilic attack. The sulfur atom, now activated within the intermediate structure, attacks the electrophilic carbon of the benzyl bromide. This two-step cascade ensures that the sulfur atom is selectively inserted between the heteroaryl ring and the alkyl group, preventing the formation of symmetric dialkyl sulfides or diaryl sulfides which are common byproducts in other methods. The reaction proceeds efficiently at temperatures ranging from 140°C to 170°C in the second step, providing the necessary thermal energy to overcome the activation barrier for the C-S bond formation without degrading the sensitive heterocyclic core.
From an impurity control perspective, this mechanism offers distinct advantages. Since the reaction does not involve radical pathways often associated with metal-catalyzed couplings, the formation of homocoupling byproducts (such as bipyridines) is minimized. The use of a slight excess of thiourea and benzyl bromide (typically 1.2 equivalents) drives the reaction to completion while keeping side reactions manageable. The absence of metal catalysts eliminates the risk of metal-induced decomposition or complexation with the product, resulting in a cleaner crude reaction profile. This purity is critical for pharmaceutical applications where strict limits on impurities are enforced. The selectivity for asymmetric sulfides is inherent to the stepwise addition of reagents, allowing chemists to precisely control the substitution pattern on the sulfur atom, thereby expanding the accessible chemical space for drug discovery and process development teams.
How to Synthesize 2-Pyridyl Methyl Sulfide Efficiently
To implement this technology for the production of key intermediates like 2-pyridyl methyl sulfide, operators follow a straightforward two-stage thermal protocol. The process begins by charging a high-pressure sealed tube with 2-bromopyridine and thiourea in a molar ratio of 1:1.2. The mixture is heated directly under air at 80°C for 1 hour to facilitate the initial coupling. Following this activation period, the alkylating agent, benzyl bromide (1.2 equivalents), is introduced into the system. The temperature is then ramped up to 140°C, and the reaction is maintained for 23 hours to ensure full conversion. This specific sequence is vital for maximizing yield, which has been demonstrated to reach up to 76% for the model substrate. The detailed standardized synthesis steps for scaling this reaction are provided in the guide below.
- Mix 2-bromopyridine and thiourea (1: 1.2 molar ratio) in a sealed tube and heat at 80°C for 1 hour under air without solvent.
- Add substituted benzyl bromide (1.2 equivalents) to the reaction mixture.
- Continue heating the mixture at 140°C to 170°C for 23 hours to complete the alkylation and form the final asymmetric thioether product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free, catalyst-free methodology translates into tangible strategic advantages. The elimination of expensive transition metal catalysts and ligands directly reduces the Bill of Materials (BOM) cost, while the removal of organic solvents cuts down on both purchase costs and the significant expenses associated with solvent recovery and waste disposal. The use of thiourea, a commodity chemical, instead of specialized thiols, ensures a stable and cost-effective supply of raw materials. Additionally, the simplified reaction conditions reduce the dependency on complex engineering controls, such as nitrogen blanketing systems, thereby lowering capital expenditure for new production lines.
- Cost Reduction in Manufacturing: The most significant financial benefit arises from the complete removal of noble metal catalysts and organic solvents. Traditional cross-coupling reactions often require palladium or copper catalysts which are subject to volatile market pricing and require costly removal steps to meet ppm-level specifications. By utilizing a catalyst-free thermal process, manufacturers can bypass these expensive purification stages entirely. Furthermore, the solvent-free nature of the reaction eliminates the need for large volumes of organic solvents, reducing both raw material procurement costs and the environmental fees associated with hazardous waste treatment. This lean manufacturing approach significantly improves the gross margin for high-volume heteroaryl sulfide production.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous raw materials like thiourea and benzyl bromides enhances supply chain resilience. Unlike thiols, which are often classified as hazardous materials with strict transportation and storage regulations, the reagents used in this process are easier to handle and source globally. The robustness of the reaction under air atmosphere means that production is less susceptible to disruptions caused by inert gas supply failures. This reliability ensures consistent lead times for customers, making it an ideal choice for long-term contracts where supply continuity is paramount for downstream API synthesis.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the absence of exothermic risks associated with rapid catalyst activation or solvent boiling. The high-temperature conditions (up to 170°C) are easily achievable in standard industrial reactors. From an environmental standpoint, the process generates minimal waste, aligning with green chemistry principles and helping companies meet increasingly stringent ESG (Environmental, Social, and Governance) targets. The reduction in hazardous waste streams simplifies regulatory compliance and reduces the administrative burden on EHS (Environment, Health, and Safety) departments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this heteroaryl sulfide synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What are the primary advantages of this thiourea-based synthesis method over traditional thiol routes?
A: This method eliminates the need for highly toxic and malodorous thiols or thiophenols, significantly improving workplace safety and environmental compliance. Furthermore, it operates without transition metal catalysts or organic solvents, reducing downstream purification costs and waste generation.
Q: Can this protocol be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable due to its simplicity. It requires only standard heating equipment under air atmosphere, avoiding the need for expensive inert gas lines or high-pressure reactors typically associated with sensitive catalytic systems.
Q: What is the substrate scope for the alkylating agent in this reaction?
A: The method demonstrates excellent versatility, successfully reacting with various substituted benzyl bromides (including electron-withdrawing and donating groups), linear alkyl bromides, cyclic alkyl bromides, and even allylic bromides to produce diverse asymmetric sulfides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heteroaryl Sulfide Supplier
The synthetic route disclosed in CN111559976B represents a significant advancement in the manufacturing of sulfur-containing pharmaceutical intermediates, offering a cleaner and more economical alternative to legacy methods. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that this innovative chemistry can be seamlessly transferred to an industrial setting. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of heteroaryl sulfide meets the highest quality standards required by global regulatory bodies. We understand the critical nature of supply chain stability and are committed to delivering consistent quality for your complex synthesis needs.
We invite you to collaborate with us to leverage this green chemistry technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply of high-purity intermediates produced via a sustainable and cost-efficient process, positioning your organization for success in a competitive market.
