Advanced Metal-Free Synthesis of 1,3-Dimethyl-3-hydroxymethylindolin-2-one Intermediates
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign pathways for constructing complex heterocyclic scaffolds, particularly those serving as core structures for bioactive molecules. Patent CN112159344A introduces a groundbreaking methodology for the synthesis of 1,3-dimethyl-3-hydroxymethylindolin-2-one compounds, a privileged structure found in numerous therapeutic agents. This innovation leverages a tandem epoxidation-intramolecular Friedel-Crafts alkylation sequence promoted solely by potassium monopersulfate (Oxone) in an acetonitrile medium. Unlike traditional approaches that rely on toxic transition metals or harsh acidic conditions, this protocol operates under mild, metal-free conditions, achieving high yields while maintaining exceptional functional group tolerance. For R&D teams and procurement specialists, this represents a paradigm shift towards greener, more cost-effective manufacturing of high-value pharmaceutical intermediates.

The limitations of conventional synthetic routes for indolin-2-one derivatives often stem from their reliance on precious metal catalysis, such as palladium or rhodium complexes, which introduce significant cost burdens and regulatory hurdles regarding residual metal limits in final drug substances. Furthermore, many existing methods require stoichiometric amounts of strong Lewis acids or Brønsted acids, necessitating rigorous quenching and neutralization steps that generate substantial chemical waste and complicate downstream processing. These traditional pathways frequently suffer from narrow substrate scope, where electron-deficient or sterically hindered substrates fail to cyclize efficiently, leading to poor atom economy and inconsistent batch-to-batch reproducibility. The necessity for extensive purification to remove metal traces not only inflates production costs but also extends lead times, creating bottlenecks in the supply chain for critical API intermediates.
In stark contrast, the novel approach detailed in the patent utilizes inexpensive and readily available potassium monopersulfate as a dual-function reagent, acting simultaneously as an oxidant and an acid promoter without the need for external additives. This metal-free strategy eliminates the risk of heavy metal contamination entirely, thereby simplifying the purification workflow and ensuring compliance with stringent ICH Q3D guidelines for elemental impurities. The reaction proceeds smoothly in acetonitrile at moderate temperatures, demonstrating remarkable chemoselectivity that preserves sensitive functional groups such as halogens and ethers which might otherwise be compromised under harsher acidic or reductive conditions. By streamlining the synthetic sequence into a single-pot tandem transformation, this method drastically reduces solvent consumption and operational complexity, offering a sustainable alternative that aligns perfectly with modern green chemistry principles and industrial scalability requirements.
Mechanistic Insights into Oxone-Promoted Epoxidation-Cyclization Cascade
The mechanistic elegance of this transformation lies in the synergistic interplay between the oxidant and the substrate, initiating with the selective epoxidation of the N-methyl-N-aryl-acrylamide alkene moiety by the peroxymonosulfate anion. This initial oxidation generates a reactive epoxy intermediate along with potassium bisulfate as a byproduct, which crucially serves as an in situ proton source to activate the epoxide ring towards nucleophilic attack. The protonation of the epoxide oxygen increases the electrophilicity of the adjacent carbon centers, facilitating an intramolecular Friedel-Crafts alkylation where the electron-rich aromatic ring attacks the activated epoxide. This cyclization step constructs the indolin-2-one core with high stereochemical fidelity, forming a new carbon-carbon bond and establishing the quaternary center at the 3-position of the heterocycle.
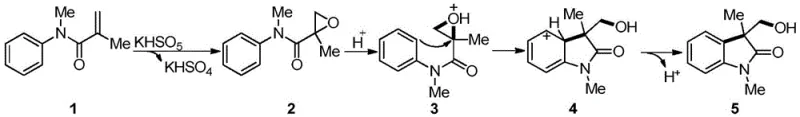
Following the cyclization event, the resulting cationic intermediate undergoes a spontaneous dehydroaromatization process to restore the aromaticity of the benzene ring, driving the reaction equilibrium towards the formation of the stable hydroxymethylindolin-2-one product. This cascade mechanism is highly efficient because it avoids the isolation of unstable intermediates and minimizes side reactions such as polymerization or over-oxidation, which are common pitfalls in stepwise synthetic approaches. The absence of exogenous acids prevents the degradation of acid-sensitive substituents, while the mild oxidative potential of Oxone ensures that the hydroxymethyl group remains intact without further oxidation to carboxylic acids. Understanding this precise mechanistic pathway allows process chemists to fine-tune reaction parameters, such as temperature and concentration, to maximize yield and minimize impurity profiles, ensuring the production of high-purity intermediates suitable for direct use in subsequent drug synthesis steps.
How to Synthesize 1,3-Dimethyl-3-hydroxymethylindolin-2-one Efficiently
Executing this synthesis requires careful attention to solvent quality and thermal control to replicate the high yields reported in the patent literature. The standard protocol involves dissolving the N-methyl-N-aryl-acrylamide precursor in dry acetonitrile to achieve a concentration between 0.1 and 0.5 mol/L, followed by the addition of 1.5 equivalents of potassium monopersulfate. The reaction mixture is then heated to an optimal temperature of 90°C and maintained under vigorous stirring for approximately 24 hours to ensure complete conversion. Monitoring the reaction progress via TLC or HPLC is recommended to determine the exact endpoint, after which the mixture is concentrated and the crude product is purified using standard silica gel chromatography techniques.
- Charge a reaction vessel with N-methyl-N-aryl-acrylamide substrate and 1.5 equivalents of potassium monopersulfate (Oxone) in acetonitrile solvent.
- Heat the reaction mixture to 90°C and maintain stirring for 24 hours to ensure complete conversion via the epoxidation-cyclization cascade.
- Upon completion, concentrate the mixture and purify the crude product using silica gel column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages by fundamentally altering the cost structure and supply reliability of producing indolin-2-one derivatives. The elimination of transition metal catalysts removes a major variable cost component, as precious metals like palladium are subject to volatile market pricing and require specialized recovery processes. Furthermore, the use of commodity chemicals like Oxone and acetonitrile ensures a stable and diversified supply base, reducing the risk of raw material shortages that can plague specialized reagent-dependent syntheses. The simplified workup procedure, devoid of complex metal scavenging steps, translates directly into reduced processing time and lower labor costs, enhancing the overall throughput of manufacturing facilities.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency in this process is the complete avoidance of expensive transition metal catalysts and ligands, which traditionally account for a significant portion of the Bill of Materials (BOM) in heterocycle synthesis. By substituting these with low-cost oxidants, manufacturers can achieve substantial savings on raw material expenditures while simultaneously reducing waste disposal costs associated with heavy metal residues. The high atom economy of the tandem reaction further contributes to cost optimization by maximizing the conversion of starting materials into the desired product, minimizing the loss of valuable intermediates.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit significantly from the use of widely available bulk chemicals rather than proprietary or niche catalysts that may have long lead times or single-source dependencies. Potassium monopersulfate and acetonitrile are produced at massive scales globally, ensuring consistent availability and price stability even during periods of market fluctuation. This robustness in the supply chain mitigates the risk of production delays, allowing for more accurate forecasting and reliable delivery schedules to downstream pharmaceutical customers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The metal-free nature of this reaction simplifies regulatory compliance and environmental permitting, as there is no need for complex effluent treatment systems designed to capture and sequester heavy metals. This facilitates easier scale-up from laboratory to pilot and commercial production scales, as the safety profile is improved by removing pyrophoric or toxic metal reagents. The reduced environmental footprint aligns with corporate sustainability goals and increasingly stringent global regulations on chemical manufacturing, positioning this technology as a future-proof solution for long-term production needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this Oxone-mediated synthesis platform. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing clarity on reaction scope, limitations, and practical handling. Understanding these nuances is essential for process development teams evaluating the feasibility of adopting this technology for their specific pipeline candidates.
Q: Why is acetonitrile critical for this Oxone-mediated cyclization?
A: According to patent optimization data, acetonitrile is the only solvent that supports high yields (up to 79%). Other common solvents like toluene, DMSO, or THF result in no reaction or trace products, likely due to specific solvation effects required for the epoxidation transition state.
Q: Does this method require transition metal catalysts?
A: No, this process is completely metal-free. It utilizes potassium monopersulfate (Oxone) which acts as both the oxidant and the acid promoter, eliminating the need for expensive palladium or copper catalysts and subsequent heavy metal removal steps.
Q: What is the functional group tolerance of this synthesis?
A: The method demonstrates excellent compatibility with various substituents on the aryl ring, including alkyl groups (methyl), alkoxy groups (methoxy), and halogens (bromo), yielding target products with efficiencies ranging from 76% to 86%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Dimethyl-3-hydroxymethylindolin-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative, metal-free synthetic routes like the one described in CN112159344A to enhance the competitiveness of your pharmaceutical supply chain. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards for identity, assay, and impurity profiles.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume needs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your development timelines with reliable, high-quality chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
