Scalable Synthesis of 1-(biphenyl-4-yl)-2-methyl-2-morpholinopropan-1-one for Industrial UV Curing Applications
The global demand for high-performance ultraviolet (UV) curing materials has driven significant innovation in the field of radical photoinitiators, specifically targeting compounds that offer superior reactivity without the environmental drawbacks of traditional sulfur-based systems. Patent CN103588729A introduces a groundbreaking synthetic methodology for 1-(biphenyl-4-yl)-2-methyl-2-morpholinopropan-1-one, a highly efficient alpha-aminoacetophenone derivative that serves as a critical intermediate in advanced polymer chemistry. This novel approach fundamentally restructures the production landscape by replacing hazardous and costly bromination steps with a streamlined chlorination protocol, thereby addressing long-standing issues regarding atomic economy and industrial scalability. For R&D directors and procurement specialists alike, this technology represents a pivotal shift towards greener manufacturing practices that do not compromise on yield or purity standards. The process leverages a continuous reaction sequence where intermediates are not isolated, significantly reducing processing time and solvent consumption while maintaining a total yield exceeding 90%. By integrating this robust synthetic pathway, manufacturers can achieve a reliable photoinitiator intermediate supplier status, ensuring consistent quality for downstream UV curing applications in coatings, inks, and adhesives.
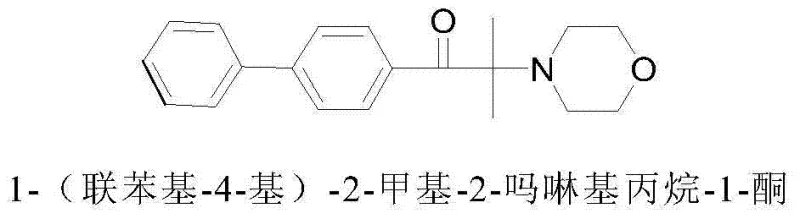
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-amino ketone photoinitiators relied heavily on bromination strategies, which, while chemically effective, imposed severe economic and environmental burdens on large-scale production facilities. Traditional routes typically involved the reaction of biphenyl derivatives with isobutyryl chloride followed by a bromination step using elemental bromine to generate an alpha-bromo ketone intermediate. This reliance on bromine introduced multiple points of failure: the reagent itself is expensive, highly corrosive to standard reactor equipment, and generates substantial quantities of acidic waste water that require complex neutralization and treatment protocols. Furthermore, the subsequent substitution reaction with morpholine often necessitated a large excess of the amine to act as both reactant and acid scavenger, leading to poor atom utilization and difficult purification processes. The cumulative effect of these inefficiencies was a prolonged production cycle, often exceeding 24 hours for the substitution step alone, and a final product purity that struggled to consistently reach the >99% threshold required for high-end optical applications. These factors collectively rendered conventional bromination methods economically unviable for modern, cost-sensitive industrial operations seeking sustainable growth.
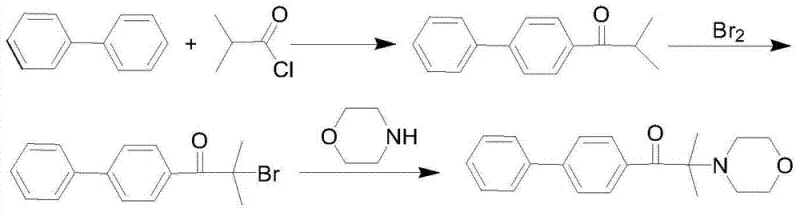
The Novel Approach
In stark contrast to the legacy bromination pathways, the innovative technique disclosed in the patent utilizes a direct chlorination strategy that dramatically simplifies the synthetic architecture while enhancing overall process economics. By substituting elemental bromine with chlorine gas, the new method capitalizes on the lower cost and higher availability of chlorine, effectively decoupling production costs from the volatile pricing of bromine markets. This strategic substitution is not merely a reagent swap but a holistic process redesign that enables a telescoped reaction sequence; the acylation, chlorination, and substitution steps can be performed sequentially in the same solvent system without the need for intermediate isolation or solvent exchange. This continuity eliminates energy-intensive distillation steps between reactions, thereby reducing the thermal load on the facility and minimizing solvent losses. The result is a streamlined workflow that significantly shortens the overall production timeline and enhances the safety profile of the operation by removing the handling risks associated with liquid bromine. For supply chain managers, this translates into a more resilient manufacturing process capable of rapid scale-up to meet fluctuating market demands for high-purity photoinitiator intermediates.
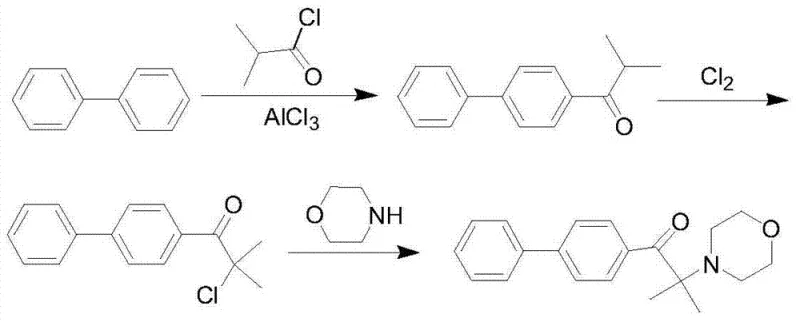
Mechanistic Insights into Chlorination-Based Substitution
The core of this technological advancement lies in the precise control of the chlorination and subsequent nucleophilic substitution mechanisms, which are optimized to maximize yield while minimizing byproduct formation. The process initiates with a classic Friedel-Crafts acylation where biphenyl reacts with isobutyryl chloride in the presence of a Lewis acid catalyst such as aluminum chloride or iron trichloride. Unlike traditional methods that might isolate the resulting ketone, the reaction mixture is directly subjected to chlorination at elevated temperatures ranging from 90°C to 120°C. Under these conditions, chlorine gas selectively substitutes the alpha-hydrogen of the ketone moiety to form the reactive alpha-chloro ketone intermediate with high specificity. The mechanistic elegance continues in the final substitution step, where the alpha-chloro ketone reacts with morpholine. Crucially, the patent describes the use of an inorganic base aqueous solution, such as sodium carbonate or sodium bicarbonate, to neutralize the hydrogen chloride generated during the reaction. This biphasic system allows the morpholine to partition effectively between the organic and aqueous phases, preventing local concentration spikes that could lead to unwanted side reactions. This careful balancing of reaction kinetics ensures that the morpholine acts primarily as a nucleophile rather than being consumed as an acid scavenger, thereby optimizing reagent usage and driving the reaction to completion with exceptional efficiency.
Furthermore, the impurity profile of the final product is rigorously controlled through the inherent selectivity of the chlorination step and the subsequent crystallization purification. The use of chlorine, being smaller and more reactive than bromine, facilitates a cleaner transformation that reduces the formation of poly-halogenated byproducts which are often difficult to separate. The final purification involves a simple recrystallization from methanol or ethanol, a step that is highly effective due to the high initial purity of the crude product obtained from the continuous reaction sequence. The patent data indicates that the filtrate from this crystallization can be recycled for subsequent batches, further enhancing the material efficiency of the process. For quality assurance teams, this means that achieving purity levels greater than 99% is not an anomaly but a reproducible standard, ensuring that the photoinitiator performs consistently in sensitive UV curing formulations without introducing color bodies or odor issues associated with sulfur-containing alternatives.
How to Synthesize 1-(biphenyl-4-yl)-2-methyl-2-morpholinopropan-1-one Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters, particularly temperature control and reagent stoichiometry, to fully realize the benefits of the continuous process design. Operators must ensure that the acylation step is conducted at low temperatures to prevent polysubstitution on the biphenyl ring, followed by a controlled ramp-up for the chlorination phase to ensure complete conversion of the ketone. The final substitution step benefits from moderate heating to accelerate kinetics without degrading the sensitive morpholine ring. Detailed standard operating procedures regarding the specific addition rates of chlorine gas and the management of the exothermic neutralization reaction are critical for safe and effective scale-up. The following guide outlines the standardized synthesis steps derived from the patent examples to assist technical teams in replicating this high-yield pathway.
- Perform Friedel-Crafts acylation of biphenyl with isobutyryl chloride using aluminum chloride or iron trichloride catalyst at 5-20°C.
- Conduct direct chlorination of the resulting ketone solution at 90-120°C by passing chlorine gas to form the alpha-chloro ketone intermediate.
- Execute nucleophilic substitution by reacting the chloro-intermediate with morpholine in the presence of an inorganic base aqueous solution at 40-80°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this chlorination-based synthesis offers profound advantages that directly impact the bottom line and operational resilience of chemical manufacturing enterprises. The elimination of bromine, a high-cost and logistically challenging reagent, immediately lowers the variable cost of goods sold, while the ability to reuse solvents across multiple reaction steps drastically reduces raw material procurement volumes. This process intensification means that facilities can produce significantly more product per unit of time and per unit of solvent purchased, creating a leaner and more responsive supply chain. Moreover, the reduction in waste generation, particularly the avoidance of heavy bromine waste streams and excessive amine salts, simplifies environmental compliance and lowers the overhead costs associated with waste disposal and treatment. These factors combine to create a manufacturing profile that is not only more profitable but also more sustainable, aligning with the increasing regulatory pressures and corporate sustainability goals prevalent in the global chemical industry today.
- Cost Reduction in Manufacturing: The substitution of expensive bromine with inexpensive chlorine gas serves as a primary driver for cost optimization, fundamentally altering the cost structure of the photoinitiator intermediate. By removing the need for stoichiometric amounts of costly brominating agents and reducing the excess morpholine required for acid scavenging through the use of inorganic bases, the direct material costs are significantly lowered. Additionally, the telescoped nature of the reaction eliminates intermediate isolation steps, which traditionally consume vast amounts of energy for distillation and drying, further compounding the savings in utility costs. This comprehensive approach to cost reduction ensures that the final product remains competitive even in price-sensitive markets, providing a distinct economic advantage over legacy production methods.
- Enhanced Supply Chain Reliability: Relying on chlorine, a commodity chemical with a robust and widespread supply infrastructure, mitigates the risk of raw material shortages that can plague specialized reagent supply chains. The simplified process flow, which requires fewer unit operations and less specialized equipment for handling corrosive liquids like bromine, enhances the overall reliability of the production line. This robustness allows for more predictable lead times and greater flexibility in scheduling production runs, enabling suppliers to respond more agilely to customer demand fluctuations. Consequently, downstream customers benefit from a more stable supply of high-purity intermediates, reducing the risk of production stoppages in their own UV curing material manufacturing lines.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scalability, with reaction conditions that are easily managed in large-scale reactors without the need for exotic materials of construction. The significant reduction in wastewater volume and the elimination of toxic bromine residues simplify the environmental permitting process and reduce the long-term liability associated with hazardous waste management. This alignment with green chemistry principles not only future-proofs the manufacturing asset against tightening environmental regulations but also enhances the brand value of the end products by offering a more eco-friendly sourcing option. The ability to recycle solvents and mother liquors further underscores the commitment to resource efficiency, making this a highly attractive proposition for environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this advanced photoinitiator intermediate. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on the practical implications of adopting this new methodology. Understanding these details is crucial for technical buyers and formulation chemists who need to validate the suitability of this material for their specific high-performance coating or ink applications.
Q: What are the primary advantages of the chlorination route over the bromination route for this photoinitiator?
A: The chlorination route utilizes chlorine gas, which is significantly cheaper and more atom-economical than bromine. It eliminates the need for expensive brominating agents, reduces equipment corrosion risks, and simplifies waste treatment by avoiding heavy bromine waste streams.
Q: How does the new synthesis method improve solvent efficiency?
A: The patented process allows for a 'one-pot' or continuous reaction sequence where the same solvent is used across all three steps. This eliminates the need for intermediate solvent recovery and distillation between steps, drastically reducing energy consumption and solvent loss.
Q: Is this intermediate suitable as a substitute for sulfur-containing photoinitiators like Irgacure 907?
A: Yes, 1-(biphenyl-4-yl)-2-methyl-2-morpholinopropan-1-one is a sulfur-free alternative that offers excellent yellowing resistance and lacks the unpleasant odor associated with sulfur-containing initiators, making it ideal for high-quality UV curing applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(biphenyl-4-yl)-2-methyl-2-morpholinopropan-1-one Supplier
As the global market for UV curing technologies continues to expand, the need for high-quality, cost-effective photoinitiator intermediates has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep technical expertise to deliver superior chemical solutions that meet the rigorous demands of modern manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your growth from pilot scale to full industrial deployment. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1-(biphenyl-4-yl)-2-methyl-2-morpholinopropan-1-one meets the highest standards of performance and consistency required for sensitive optical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this advanced synthesis technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your organization.
