Advanced Ferrocenyl Palladacycle Catalysts for Scalable Biaryl Synthesis in Pharmaceutical Manufacturing
Advanced Ferrocenyl Palladacycle Catalysts for Scalable Biaryl Synthesis in Pharmaceutical Manufacturing
The landscape of organic synthesis, particularly within the realm of pharmaceutical intermediate production, is constantly evolving to meet the dual demands of higher efficiency and stricter environmental compliance. A pivotal advancement in this field is documented in Chinese Patent CN100491387C, which discloses a novel class of ferrocenyl imidazoline ring palladium compounds designed to catalyze the synthesis of coupled products with exceptional specificity. This technology represents a significant departure from conventional methods by utilizing a non-phosphine ligand system that offers superior thermal stability and remarkable insensitivity to air and water. For R&D directors and process chemists, the introduction of such a robust catalytic system implies a potential paradigm shift in how complex biaryl structures, which are ubiquitous in active pharmaceutical ingredients (APIs), are constructed. The patent details a comprehensive preparation method involving the reaction of ferrocenyl imidic acid methyl ester hydrochlorate with vicinal diamines, followed by complexation with palladium lithium halides, resulting in a catalyst that maintains high activity under relatively mild reaction conditions. This innovation not only addresses the technical challenges of catalyst deactivation but also aligns with the global industry's push towards greener, more sustainable chemical manufacturing processes that minimize hazardous waste and energy consumption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biaryl hydrocarbon derivatives, which serve as critical scaffolds in medicinal chemistry, has relied heavily on classical methods such as the Ullmann reaction or standard palladium-catalyzed cross-couplings using phosphine ligands. The Ullmann method, while foundational, suffers from significant drawbacks including poor versatility, multi-step synthetic requirements, uncertain chemical selectivity, and the necessity for harsh reaction conditions that often degrade sensitive functional groups. Furthermore, the more modern approach of using palladium inorganic salts combined with phosphine ligands, although offering better catalytic activity, introduces its own set of operational complexities. Phosphine ligands are typically expensive, highly toxic, and extremely sensitive to oxidation and moisture, necessitating rigorous inert gas protection and anhydrous conditions throughout the entire reaction and storage lifecycle. These constraints create substantial bottlenecks in industrial settings, increasing operational costs and safety risks, particularly when scaling up for the mass production of drug molecules where consistency and safety are paramount concerns for supply chain managers.
The Novel Approach
In stark contrast to these legacy technologies, the ferrocenyl imidazoline palladium compound introduced in the patent offers a streamlined and environmentally friendly alternative that effectively circumvents the limitations of phosphine-based systems. By employing a ferrocenyl backbone coupled with an imidazoline ring, the new catalyst achieves a unique electronic and steric environment that stabilizes the palladium center without the need for air-sensitive phosphines. This structural innovation allows the catalytic synthesis of coupling products to proceed under much more forgiving conditions, often tolerating the presence of water and oxygen which would typically poison traditional catalysts.  As illustrated in the general reaction scheme, the process involves dissolving the catalyst, aryl boronic acid, alkali, and halogenated aromatic compound in an organic solvent, followed by heating to facilitate the coupling. This methodology not only simplifies the operational workflow by removing the strict requirement for inert atmospheres but also expands the scope of compatible substrates, enabling the efficient synthesis of a wide array of substituted biaryl compounds that are essential for developing next-generation therapeutics and advanced materials.
As illustrated in the general reaction scheme, the process involves dissolving the catalyst, aryl boronic acid, alkali, and halogenated aromatic compound in an organic solvent, followed by heating to facilitate the coupling. This methodology not only simplifies the operational workflow by removing the strict requirement for inert atmospheres but also expands the scope of compatible substrates, enabling the efficient synthesis of a wide array of substituted biaryl compounds that are essential for developing next-generation therapeutics and advanced materials.
Mechanistic Insights into Ferrocenyl Imidazoline Palladium Catalysis
The exceptional performance of this catalytic system can be attributed to the unique mechanistic properties imparted by the ferrocenyl imidazoline ligand architecture, which facilitates a highly efficient catalytic cycle for carbon-carbon bond formation. The ferrocenyl group acts as a robust electron donor, stabilizing the palladium center in various oxidation states throughout the oxidative addition, transmetallation, and reductive elimination steps of the Suzuki-Miyaura coupling mechanism. Unlike flexible alkyl chains, the rigid ferrocenyl-imidazoline framework prevents the aggregation of palladium species into inactive black palladium metal, a common failure mode in less stable catalytic systems. 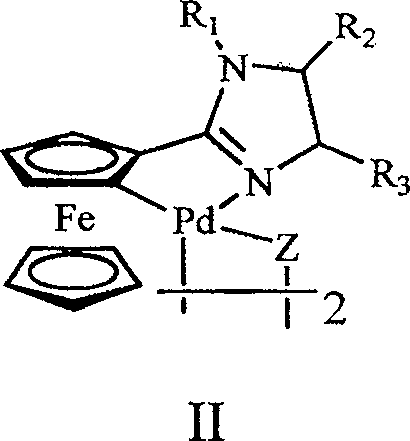 The structure shown above highlights the coordination of the palladium atom with the nitrogen atoms of the imidazoline ring and the cyclopalladated ferrocenyl moiety, creating a stable five-membered metallacycle. This specific geometry ensures that the active catalytic species remains soluble and accessible to the substrates even under prolonged heating, thereby maintaining high turnover numbers. For process chemists, understanding this stability is crucial as it directly correlates to the ability to run reactions at lower catalyst loadings, potentially as low as 0.01% to 5% mole percent relative to the halogenated aromatic compound, without sacrificing yield or reaction rate.
The structure shown above highlights the coordination of the palladium atom with the nitrogen atoms of the imidazoline ring and the cyclopalladated ferrocenyl moiety, creating a stable five-membered metallacycle. This specific geometry ensures that the active catalytic species remains soluble and accessible to the substrates even under prolonged heating, thereby maintaining high turnover numbers. For process chemists, understanding this stability is crucial as it directly correlates to the ability to run reactions at lower catalyst loadings, potentially as low as 0.01% to 5% mole percent relative to the halogenated aromatic compound, without sacrificing yield or reaction rate.
Furthermore, the mechanism inherently supports superior impurity control, a critical factor for R&D directors focused on purity profiles and regulatory compliance. The high specificity of the ferrocenyl imidazoline palladium catalyst minimizes side reactions such as homocoupling of the boronic acid or dehalogenation of the aryl halide, which are common sources of difficult-to-remove impurities in crude reaction mixtures. The thermal stability of the non-phosphine ligand ensures that the catalyst does not decompose into free palladium species that could contaminate the final product, thus reducing the burden on downstream purification processes like chromatography or crystallization. This clean reaction profile is particularly advantageous when synthesizing complex pharmaceutical intermediates where trace metal residues must be kept below stringent ppm levels. The ability to operate in mixed solvent systems, including aqueous mixtures with methanol or tetrahydrofuran, further enhances the green chemistry credentials of the process by allowing for easier workup procedures and reducing the volume of organic solvents required for extraction, ultimately leading to a more sustainable and cost-effective manufacturing route.
How to Synthesize Ferrocenyl Imidazoline Palladium Compounds Efficiently
The preparation of this high-performance catalyst is designed to be straightforward and scalable, making it accessible for both laboratory research and industrial production environments. The synthesis begins with the preparation of ferrocenyl imidic acid methyl ester hydrochloride, which is then reacted with a vicinal diamine under mild conditions to form the ferrocenyl imidazoline ligand precursor. This ligand is subsequently treated with palladium lithium halides and sodium acetate in an organic solvent at room temperature to yield the final red solid palladacycle catalyst. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and purification techniques required to achieve high purity catalyst batches, are outlined in the guide below to ensure reproducibility and quality control.
- Prepare the catalyst by reacting ferrocenyl imidic acid methyl ester hydrochloride with vicinal diamine, followed by complexation with palladium lithium halides.
- Dissolve the catalyst, aryl boronic acid, base, and halogenated aromatic compound in an organic solvent such as toluene or methanol.
- Heat the mixture to 70-150°C or stir at room temperature depending on the specific protocol, then extract and purify the resulting biaryl coupling product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ferrocenyl imidazoline palladium catalyst technology translates into tangible strategic benefits that extend far beyond simple reaction yields. The primary advantage lies in the drastic simplification of the supply chain logistics associated with catalyst handling and storage. Since the catalyst is insensitive to air and water, it eliminates the need for specialized packaging, inert gas blanketing during transport, and climate-controlled warehousing that are mandatory for traditional phosphine ligands. This robustness significantly reduces the risk of material degradation during shipping and long-term storage, ensuring that the catalyst performs consistently batch after batch, which is vital for maintaining continuous production schedules in large-scale manufacturing facilities. Moreover, the use of inexpensive bases such as potassium carbonate or sodium acetate, instead of costly and hazardous reagents, further drives down the raw material costs associated with the coupling process.
- Cost Reduction in Manufacturing: The economic impact of switching to this catalyst system is profound, primarily driven by the elimination of expensive and toxic phosphine ligands which often constitute a significant portion of the catalyst cost. By utilizing a non-phosphine ligand system that is air-stable, manufacturers can avoid the substantial overhead costs related to safety equipment, waste disposal of toxic phosphine oxides, and the complex engineering controls required to maintain anhydrous and oxygen-free environments. Additionally, the high catalytic activity allows for the use of lower catalyst loadings while still achieving high conversion rates, which directly reduces the consumption of precious palladium metal per kilogram of product produced. This efficiency gain, combined with the ability to use cheaper solvents and bases, results in a substantially lower cost of goods sold (COGS) for the final pharmaceutical intermediate, providing a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the stability and ease of synthesis of the ferrocenyl imidazoline palladium compound. Because the catalyst does not require stringent inert conditions for storage or handling, the risk of supply disruptions caused by material spoilage is virtually eliminated. The synthetic route for the catalyst itself is short and utilizes readily available starting materials like ferrocene derivatives and common diamines, ensuring a stable and reliable upstream supply of the catalyst. This reliability allows procurement teams to negotiate better terms with suppliers and maintain leaner inventory levels without fear of stockouts or quality failures. Furthermore, the broad substrate scope means that a single catalyst variant can often be used for multiple different coupling reactions, reducing the complexity of the chemical inventory and simplifying the procurement process for diverse product portfolios.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this technology offers a clear path to compliant and sustainable growth. The patent explicitly mentions that the reaction feeding scale can range from milligrams to kilograms, demonstrating proven scalability for commercial production. The process generates less hazardous waste due to the absence of toxic phosphine byproducts and the potential for using aqueous solvent mixtures, which simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only helps companies meet increasingly strict environmental regulations but also enhances their corporate social responsibility profile. The ability to scale up without encountering the heat transfer or mixing issues often associated with sensitive catalytic systems ensures that the transition from pilot plant to full commercial production is smooth and predictable.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled a set of frequently asked questions based on the specific technical details and beneficial effects described in the patent literature. These answers address common concerns regarding catalyst stability, substrate compatibility, and operational parameters, providing a clear understanding of what to expect during process development. Reviewing these insights can help R&D and procurement teams quickly assess the potential value proposition of adopting this ferrocenyl palladacycle system for their specific synthesis challenges.
Q: What are the stability advantages of this ferrocenyl catalyst compared to traditional phosphine ligands?
A: Unlike traditional phosphine ligands which are often toxic and sensitive to oxygen and water requiring inert gas protection, the ferrocenyl imidazoline palladium compound described in CN100491387C exhibits excellent thermal stability and is insensitive to air and moisture, simplifying handling and storage.
Q: Can this catalyst be used for large-scale industrial production of drug intermediates?
A: Yes, the patent explicitly states that the synthetic route is simple and the feeding scale can range from milligram levels to kilogram levels, indicating strong potential for commercial scale-up and industrial application in pharmaceutical manufacturing.
Q: What types of substrates are compatible with this catalytic system?
A: The system demonstrates broad substrate scope, successfully coupling various aryl boronic acids with halogenated aromatic compounds including those with electron-withdrawing or electron-donating groups such as cyano, nitro, acetyl, and methoxy substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ferrocenyl Imidazoline Palladium Compound Supplier
As the demand for efficient and sustainable catalytic solutions continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures that your transition to advanced technologies is seamless and successful. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to navigate the complexities of process optimization and regulatory compliance with ease. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that guarantee every batch of catalyst and intermediate meets the highest international standards. Whether you are developing a new API or optimizing an existing process, our technical expertise ensures that the potential of the ferrocenyl imidazoline palladium catalyst is fully realized in your manufacturing operations.
We invite you to engage with our technical procurement team to discuss how this innovative catalytic system can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your production volume and substrate profile. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that you have all the necessary information to make informed decisions that drive efficiency and profitability in your pharmaceutical manufacturing endeavors.
