Advanced Synthetic Route for High-Purity Nintedanib Intermediates and Commercial Scalability
Advanced Synthetic Route for High-Purity Nintedanib Intermediates and Commercial Scalability
The pharmaceutical landscape for oncology and fibrosis treatments continues to evolve, driven by the demand for more efficient and purer active pharmaceutical ingredients (APIs). A pivotal development in this sector is detailed in patent CN114181132A, which discloses a robust preparation method for nintedanib, a multi-target protein tyrosine kinase inhibitor approved for treating idiopathic pulmonary fibrosis and non-small cell lung cancer. This patent introduces a streamlined synthetic strategy that bypasses the cumbersome deprotection steps and high impurity profiles associated with legacy manufacturing processes. By leveraging a specific sequence of catalytic reduction and condensation reactions, the disclosed method achieves yields exceeding 90% with total impurity levels dropping to as low as 0.08%. For global procurement teams and R&D directors, this represents a critical opportunity to optimize the supply chain for this high-value API intermediate, ensuring both regulatory compliance and economic efficiency in large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
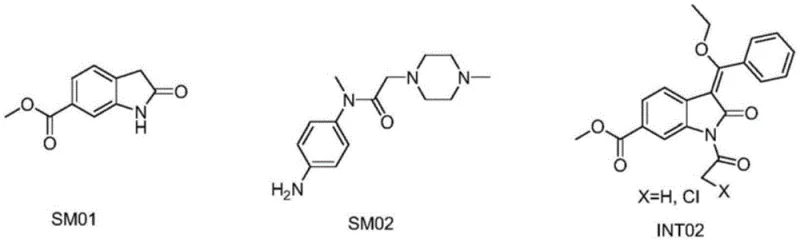
Historically, the synthesis of nintedanib has relied on intermediates such as SM01, SM02, and INT02, which necessitate complex deprotection sequences and often result in significant by-product formation. As illustrated in the structural comparison of traditional precursors, these conventional routes frequently involve harsh chemical conditions that compromise the integrity of the sensitive oxindole core. The literature indicates that attempting to simplify these operations through 'one-pot' methods often leads to inseparable mixtures, drastically lowering the final product purity and complicating downstream purification. Furthermore, the reliance on specific protecting groups that require removal in later stages adds unnecessary unit operations, increasing both the cost of goods sold (COGS) and the environmental footprint of the manufacturing process. These inefficiencies create bottlenecks for reliable nintedanib intermediate suppliers who must guarantee strict impurity specifications for regulatory filing.
The Novel Approach
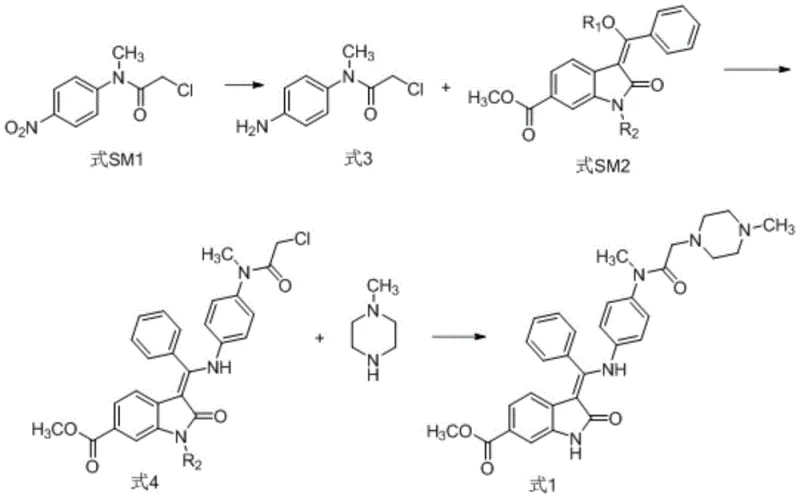
In stark contrast, the methodology outlined in CN114181132A presents a paradigm shift by utilizing a nitro-reduction strategy followed by direct condensation. The new route initiates with the reduction of compound SM1 to an amine intermediate (Formula 3), which then reacts directly with the oxindole derivative SM2. This approach elegantly circumvents the need for early-stage deprotection, thereby minimizing side reactions and preserving the stereochemical integrity of the molecule. The subsequent reaction with N-methylpiperazine proceeds under mild conditions, yielding the final target with exceptional clarity. This streamlined pathway not only enhances the overall yield but also simplifies the isolation procedure, typically requiring only recrystallization from methanol to achieve pharmaceutical-grade purity. For manufacturers seeking cost reduction in API manufacturing, this reduction in processing steps translates directly to lower operational expenditures and reduced waste generation.
Mechanistic Insights into Catalytic Hydrogenation and Condensation
The cornerstone of this improved synthesis lies in the initial catalytic hydrogenation step, where the nitro group of SM1 is selectively reduced to an amine without affecting other sensitive functional groups. The patent specifies the use of 5% Pd/C as a heterogeneous catalyst under a hydrogen pressure of 0.1-0.2 MPa, a condition that balances reaction kinetics with safety. This step is critical because incomplete reduction can lead to nitro-impurities that are difficult to remove later, while over-reduction or catalyst poisoning can stall the process. The use of methanol as a solvent facilitates the solubility of the organic substrate while maintaining a stable environment for the palladium catalyst. Following filtration to remove the catalyst, the resulting amine solution is immediately available for the next step, demonstrating a telescoped potential that further enhances process efficiency and throughput capabilities for commercial scale-up of complex pharmaceutical intermediates.
Subsequent condensation and substitution reactions are equally optimized to control the impurity profile. The reaction between the amine intermediate and the oxindole derivative (SM2) is conducted in a mixed solvent system of methanol and DMSO at elevated temperatures (70°C), promoting the formation of the Schiff base or condensation product (Formula 4). The final nucleophilic substitution with N-methylpiperazine is performed in the presence of potassium carbonate, which acts as a base to scavenge the leaving group. Analytical data from the patent examples reveals that this specific sequence results in a maximum single impurity of merely 0.03% and a total impurity content of 0.08%. Such rigorous impurity control is vital for R&D directors focusing on the purity and impurity profile of the final drug substance, as it significantly reduces the burden on analytical quality control and ensures consistent batch-to-batch reproducibility.
How to Synthesize Nintedanib Efficiently
Implementing this novel synthetic route requires precise control over reaction parameters to maximize the benefits observed in the patent examples. The process is designed to be robust, utilizing common reagents and standard equipment found in most fine chemical facilities. The key to success lies in the efficient handling of the catalytic hydrogenation step and the careful management of the exothermic condensation reaction. Operators must ensure complete removal of the palladium catalyst prior to the addition of the oxindole derivative to prevent any potential metal-catalyzed side reactions. The detailed standardized synthesis steps below outline the specific stoichiometry, temperature profiles, and workup procedures necessary to replicate the high yields and purity reported in the intellectual property documentation.
- Reduce compound SM1 (nitro precursor) to amine compound 3 using catalytic hydrogenation with Pd/C.
- React compound 3 with protected oxindole derivative SM2 to form the condensation product (compound 4).
- Perform nucleophilic substitution on compound 4 with N-methylpiperazine to yield the final nintedanib product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers tangible strategic advantages beyond mere technical elegance. The elimination of complex deprotection sequences and the reduction in unit operations directly correlate to a simplified manufacturing workflow. This simplification reduces the dependency on specialized reagents and minimizes the number of intermediate isolations, which are often the primary sources of yield loss and delay in pharmaceutical production. Consequently, this leads to substantial cost savings in raw material consumption and labor hours. Furthermore, the use of mild reaction conditions (10-30°C for the final steps) lowers the energy demand for heating and cooling, contributing to a more sustainable and economically viable production model that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The novel route eliminates the need for expensive protecting group manipulations and the associated reagents required for their removal. By streamlining the synthesis from SM1 to the final product in fewer steps, the process inherently reduces the consumption of solvents and auxiliary chemicals. This reduction in material intensity, combined with the high isolated yield of over 90% reported in the examples, drives down the cost per kilogram of the active ingredient. Additionally, the avoidance of transition metal catalysts in the later stages removes the necessity for costly and time-consuming heavy metal scavenging processes, further optimizing the overall production economics.
- Enhanced Supply Chain Reliability: The robustness of the catalytic hydrogenation and condensation steps ensures a high degree of process reliability, which is crucial for maintaining continuous supply lines. Unlike older methods that suffer from variable yields due to difficult separations, this method produces a crude product of such high purity that simple recrystallization suffices for final purification. This predictability allows supply chain planners to forecast production timelines with greater accuracy, reducing the risk of stockouts. The use of commercially available starting materials like SM1 and N-methylpiperazine also mitigates the risk of raw material shortages, ensuring a stable and resilient supply chain for high-purity nintedanib intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The moderate hydrogen pressure and ambient temperature requirements for key steps reduce the engineering controls needed for reactor design, facilitating faster technology transfer. From an environmental perspective, the reduction in solvent usage and the elimination of hazardous deprotection by-products significantly lower the waste load. This aligns with stringent environmental regulations and reduces the costs associated with waste treatment and disposal, making the process not only chemically superior but also environmentally compliant for large-scale industrial operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims presented in CN114181132A, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this new process for their own manufacturing portfolios.
Q: What are the purity levels achievable with this new nintedanib synthesis method?
A: According to patent CN114181132A, the novel route achieves exceptional purity with a maximum single impurity of 0.03% and total impurities as low as 0.08%, significantly outperforming conventional methods that struggle with by-product separation.
Q: How does this process improve supply chain stability compared to traditional routes?
A: The new method utilizes mild reaction conditions (10-30°C) and avoids complex deprotection sequences found in older routes. This simplifies the workflow, reduces the risk of batch failure, and ensures more consistent lead times for high-volume API production.
Q: Is the catalytic hydrogenation step scalable for industrial manufacturing?
A: Yes, the process employs standard 5% Pd/C catalysts under moderate hydrogen pressure (0.1-0.2 MPa), which are well-established parameters for commercial-scale reactors, ensuring safe and efficient scale-up from kilogram to tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nintedanib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively analyzed the route disclosed in CN114181132A and possesses the technical capability to implement this high-efficiency process at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our state-of-the-art facilities are equipped with the necessary hydrogenation reactors and purification systems to meet the stringent purity specifications required for oncology APIs, guaranteeing a product that consistently meets the highest quality standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this novel method. We encourage you to request specific COA data and route feasibility assessments to verify the superior impurity profile and yield advantages firsthand. Let us be your trusted partner in delivering high-quality nintedanib intermediates that drive your drug development programs forward with speed and certainty.
